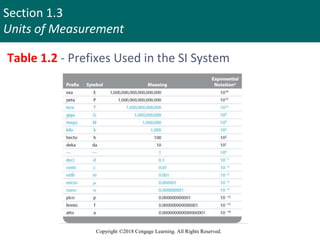
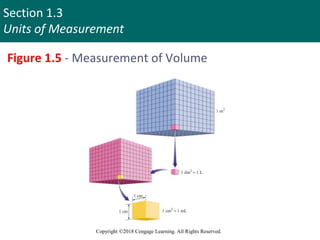
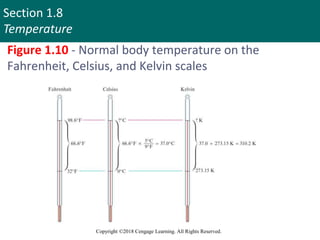
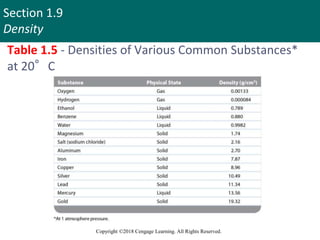
This document discusses units of measurement and significant figures in chemistry. It introduces the International System of Units (SI) as the standard system used in chemistry. The fundamental SI units for common measurements like length, mass, time and temperature are defined. The document explains the difference between accuracy and precision in measurements and discusses the impact of significant figures. Types of measurement errors like random and systematic errors are also introduced.