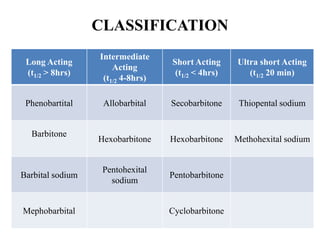
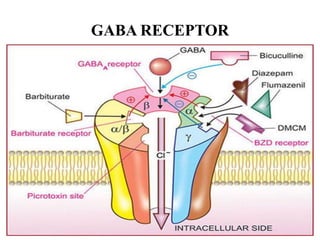
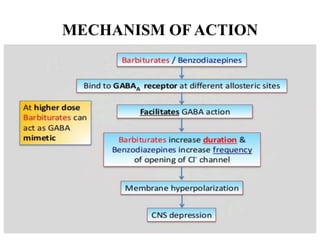
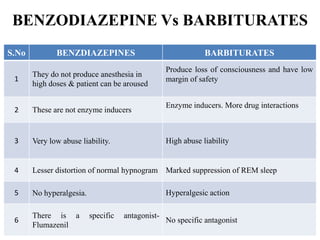
This document discusses sedative and hypnotic drugs, focusing on barbiturates. It classifies barbiturates and describes their mechanism of action, pharmacological effects, kinetics, therapeutic uses, adverse effects, interactions, and compares them to benzodiazepines. It also discusses non-benzodiazepine hypnotics including zopiclone, zolpidem, zaleplon, buspirone, and chloral hydrate.