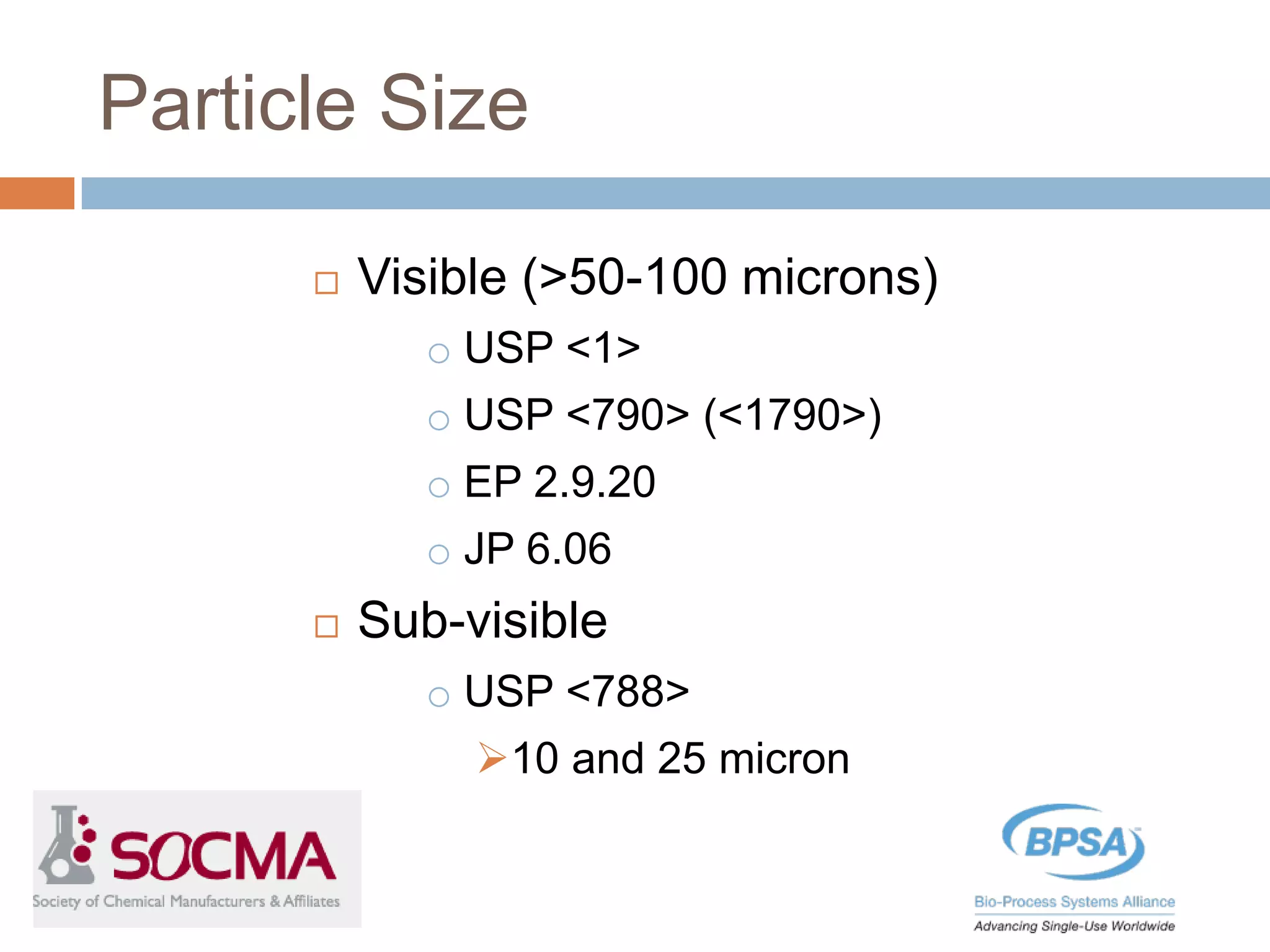
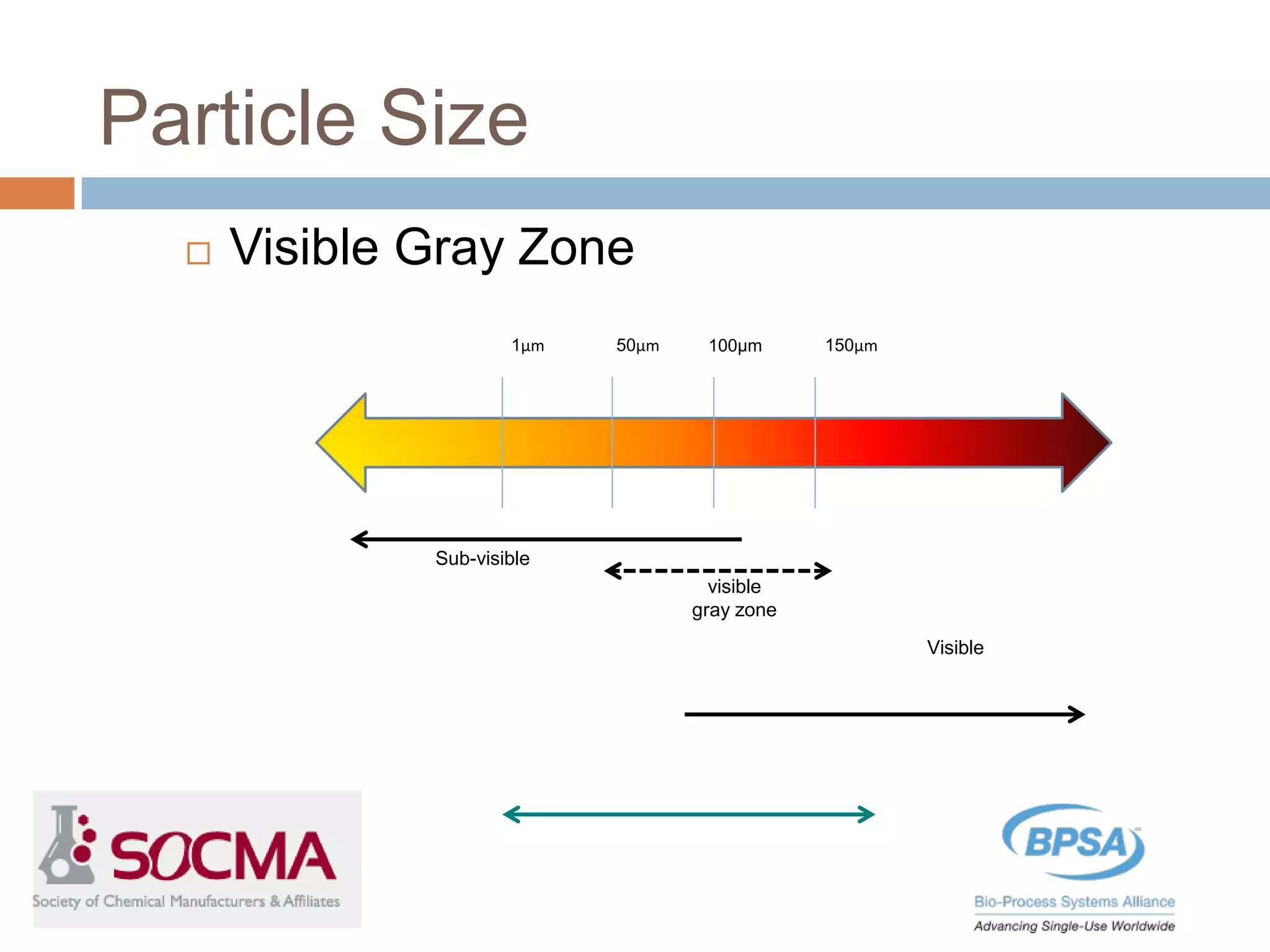
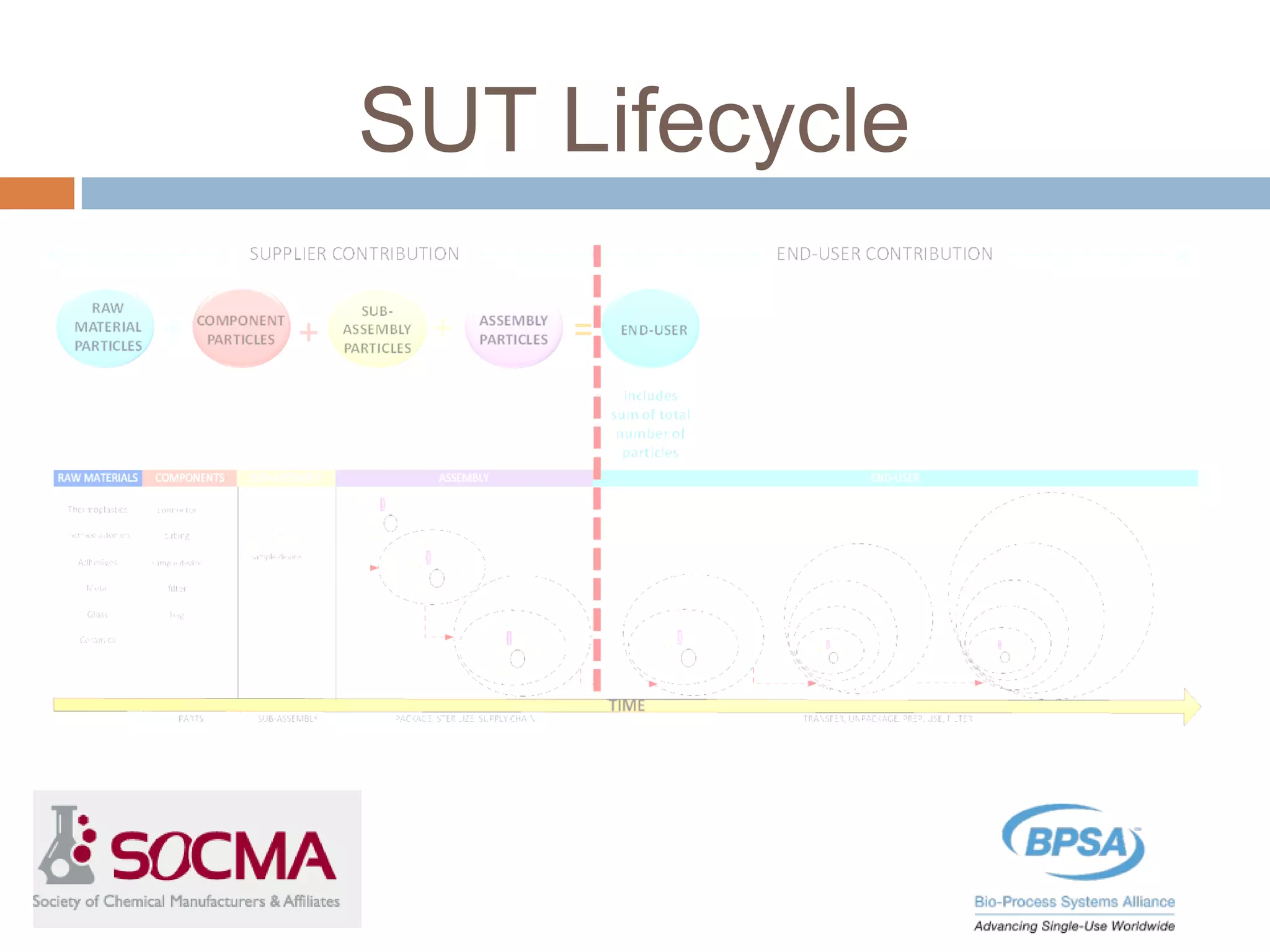
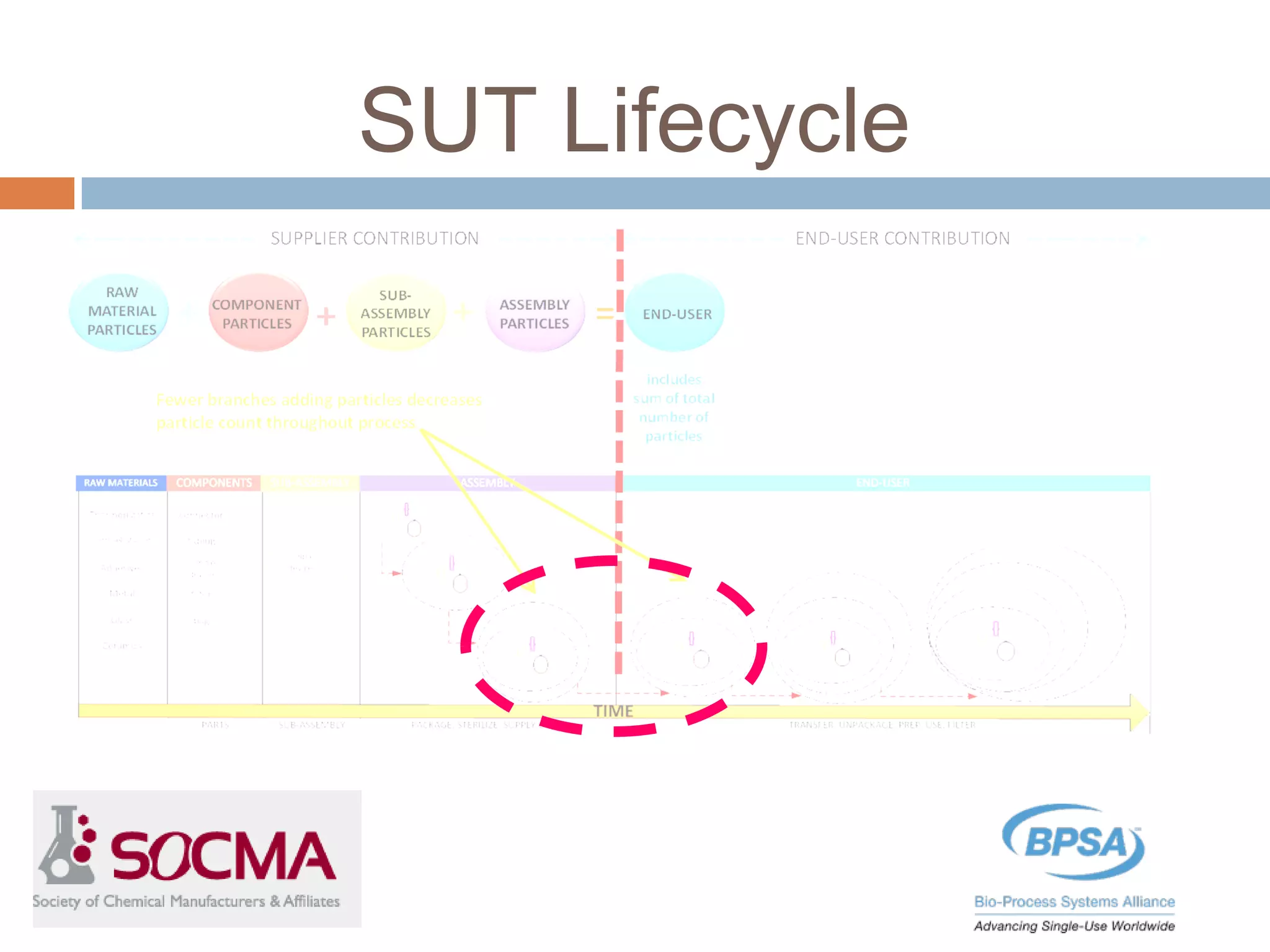
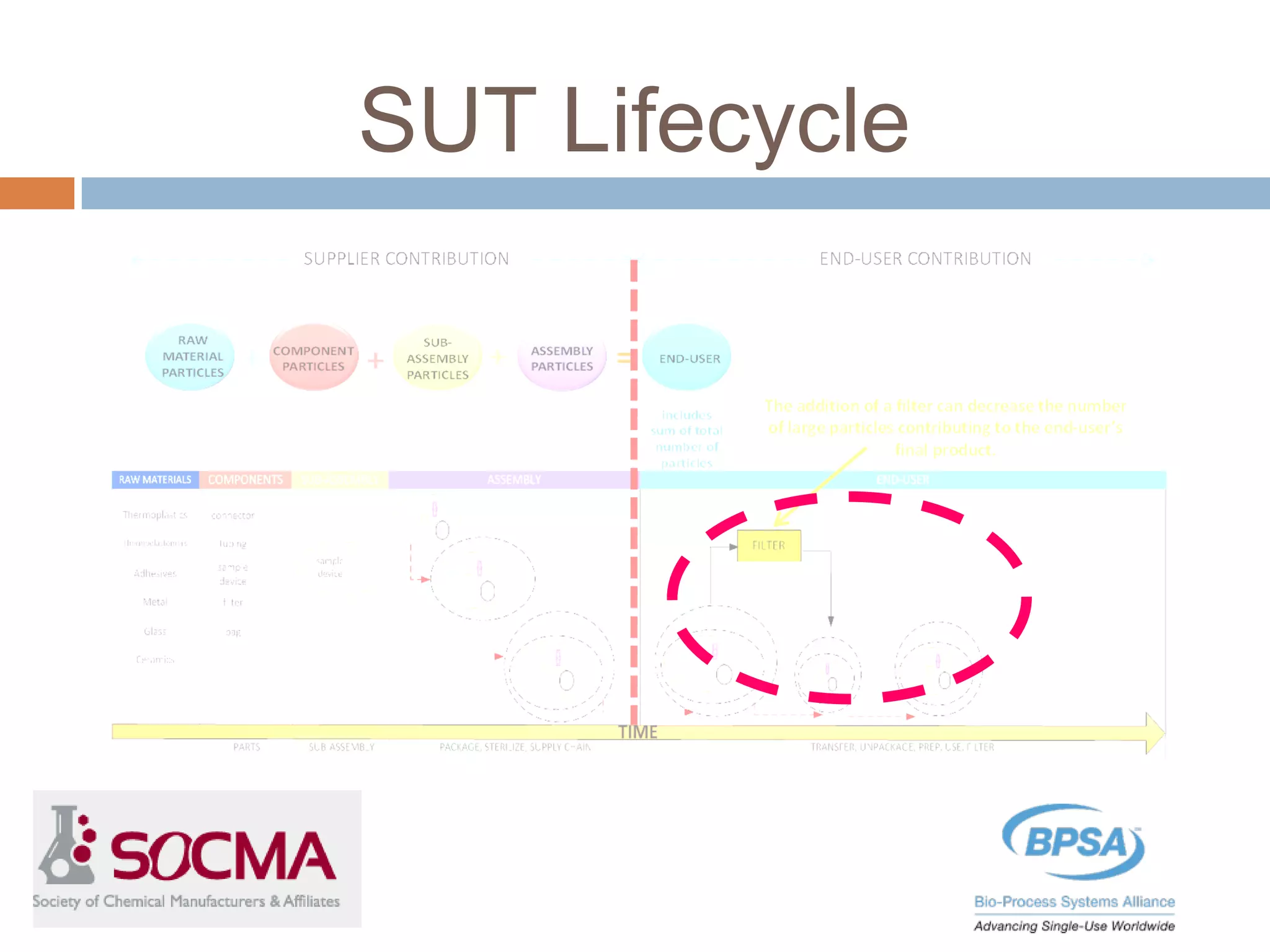
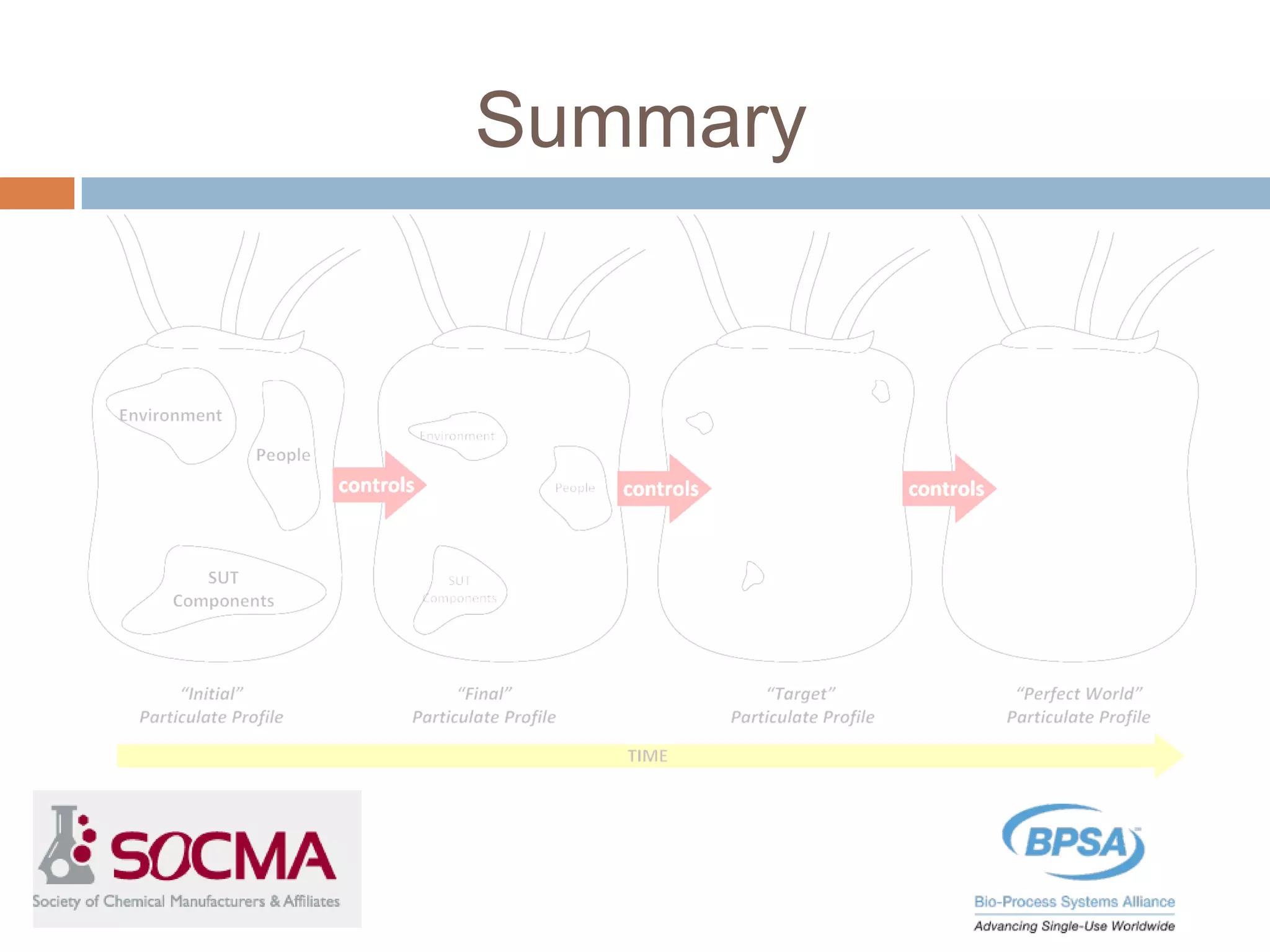
This document summarizes a report published by the Bio-Process Systems Alliance (BPSA) regarding recommendations for testing, evaluation, and control of particulates from single-use process equipment. The BPSA is a trade association that facilitates implementation of single-use technologies through various initiatives. The report was created by a working group consisting of subject matter experts from single-use technology suppliers and end users. It provides guidance on characterizing and minimizing particulate levels throughout the lifecycle of single-use technologies, including manufacturing, storage, handling and end use. It also discusses investigation and mitigation of particulate deviations. The BPSA recommends further work to develop standardized measurement methods, application-specific requirements, a catalog of particle types,