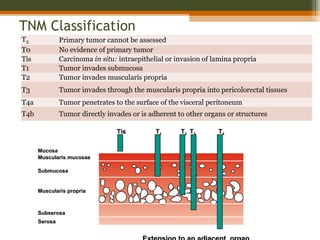
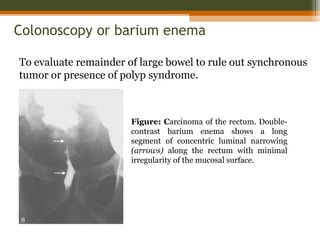
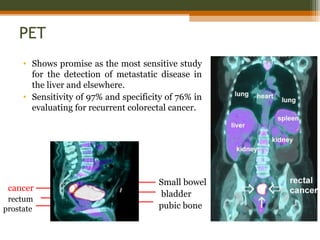
This document discusses a case of a 60-year-old male diagnosed with rectal cancer. It provides details on his medical history, including a sigmoidoscopy that revealed adenocarcinoma of the rectum. He received neoadjuvant chemoradiation therapy. The document discusses the clinical anatomy of the rectum, risk factors for rectal cancer, staging systems, diagnostic workup, and treatment options like surgery. The main treatment is surgery, with the goal of total mesorectal excision to reduce local recurrence rates.