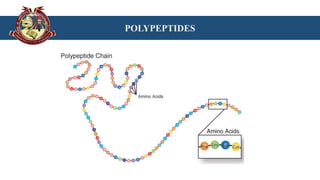
The document discusses proteins and polypeptides. It defines proteins as polymers of amino acids that perform important structural and functional roles in the body. Proteins are classified based on shape, constitution and nature. Polypeptides are continuous chains of amino acids joined by peptide bonds. They are precursors to proteins and are synthesized through a process involving transcription and translation. Common methods for synthesizing polypeptides include Fischer's method which uses protecting groups to join amino acids, and modifications of this method.