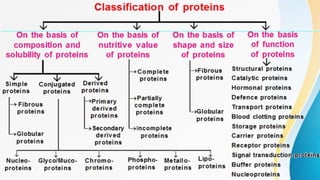
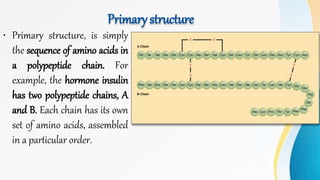
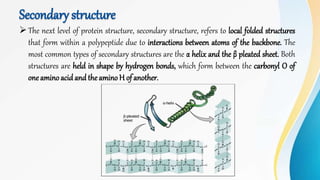
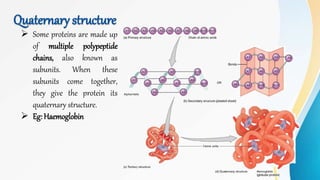
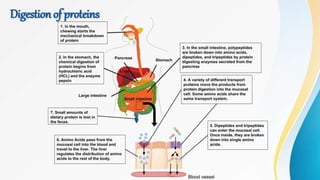
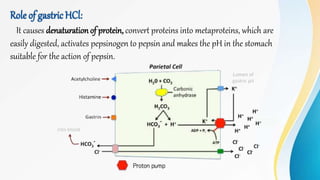
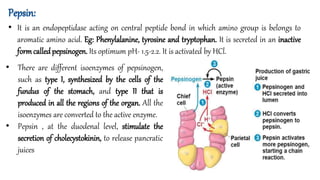
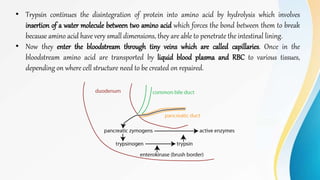
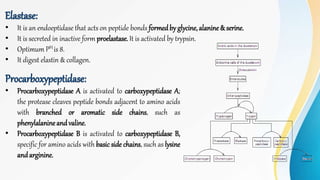
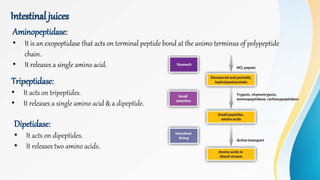
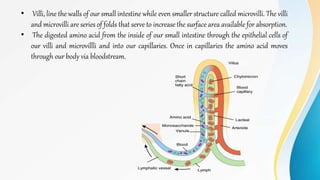
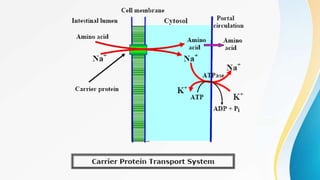
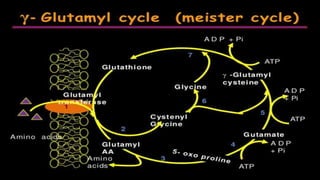
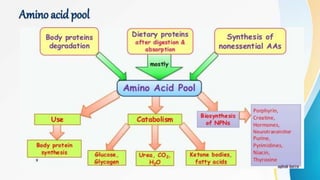
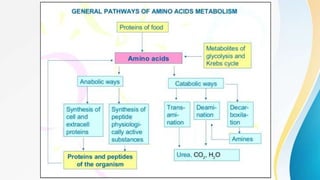
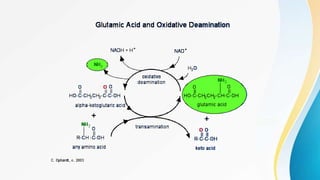
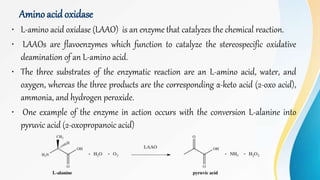
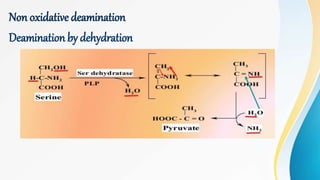
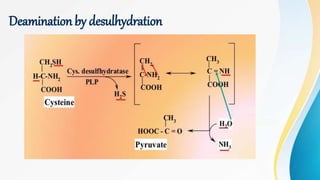
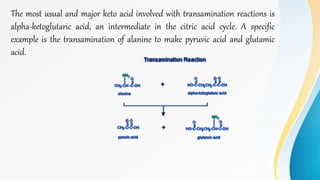
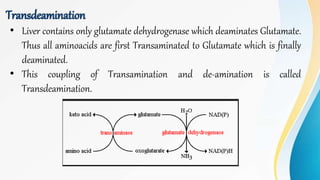
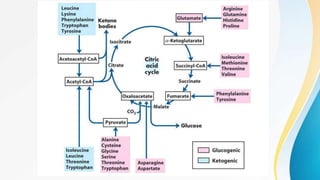
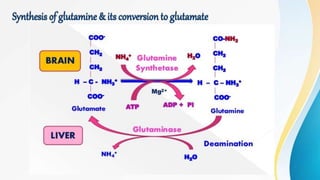
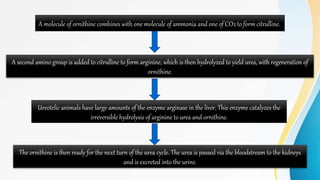
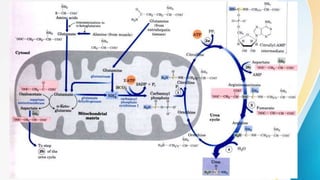
Proteins are made of amino acids and have primary, secondary, tertiary, and quaternary levels of structure. They perform many essential functions in the body. Protein digestion begins in the stomach through the actions of gastric acid and the enzyme pepsin. Pepsin breaks proteins into smaller polypeptides and proteoses. In the small intestine, pancreatic enzymes and bile further break down polypeptides into dipeptides and free amino acids which can then be absorbed.