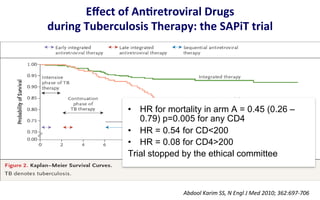
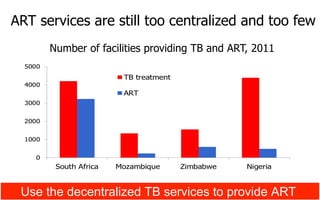
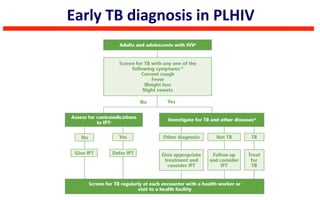
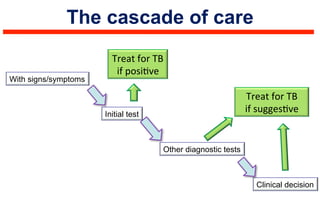
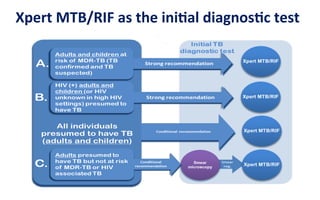
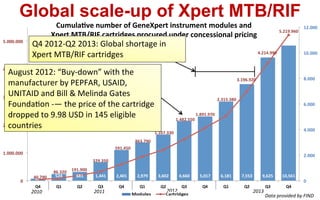
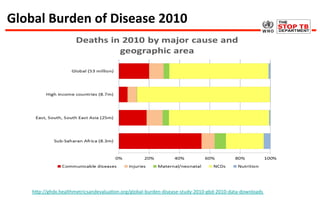
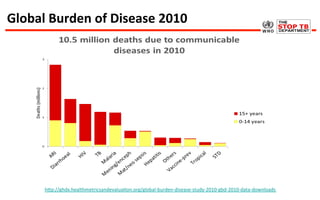
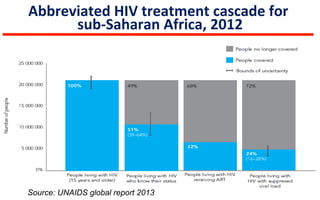
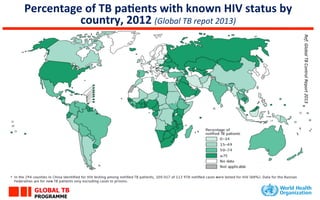
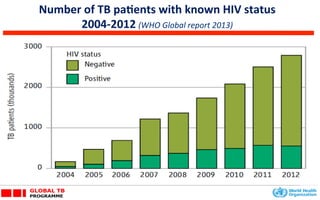
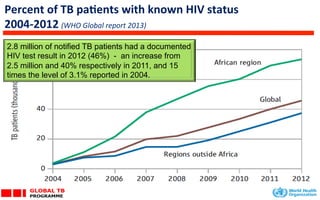
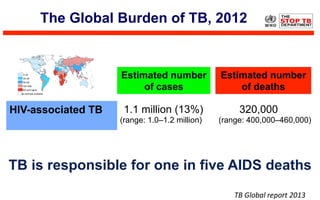
This document discusses the challenges of diagnosing HIV, AIDS, and co-infections. It notes that distinguishing between HIV infection, AIDS, and co-infections can be difficult. Point-of-care rapid tests have helped increase HIV testing, though they cannot identify acute HIV infections. The document emphasizes the importance of confirming positive rapid HIV tests with supplemental tests due to the potential for false positives in low prevalence populations.






![POC rapid tests for the diagnosis of
HIV infection
Point-‐of-‐care
rapid
tests
for
HIV
anLbody
detecLon
have
facilitated
the
scale-‐up
of
HIV
counseling
and
tesLng
throughout
resource
constraint
se_ngs
[1].
The
sensiLvity
of
these
tests
approaches
100%
and
is
equivalent
to
that
of
EIA
[2].
These
tests
cannot
idenLfy
persons
with
acute
HIV
infecLon
who
have
not
yet
developed
specific
anLbodies
[3].
1. Parekh
Clin
Pathol
2010;134:
537
2. Van
den
Berk
J
Clin
Microbiol
2003;
41:
3868
3. Stekler
JD
Clin
Infect
Dis
2009;
49:
444](https://image.slidesharecdn.com/02-140327054253-phpapp01/85/PPT-Castelli-Dall-HIV-all-AIDS-fino-alla-coinfezione-una-diagnosi-difficile-18-320.jpg)