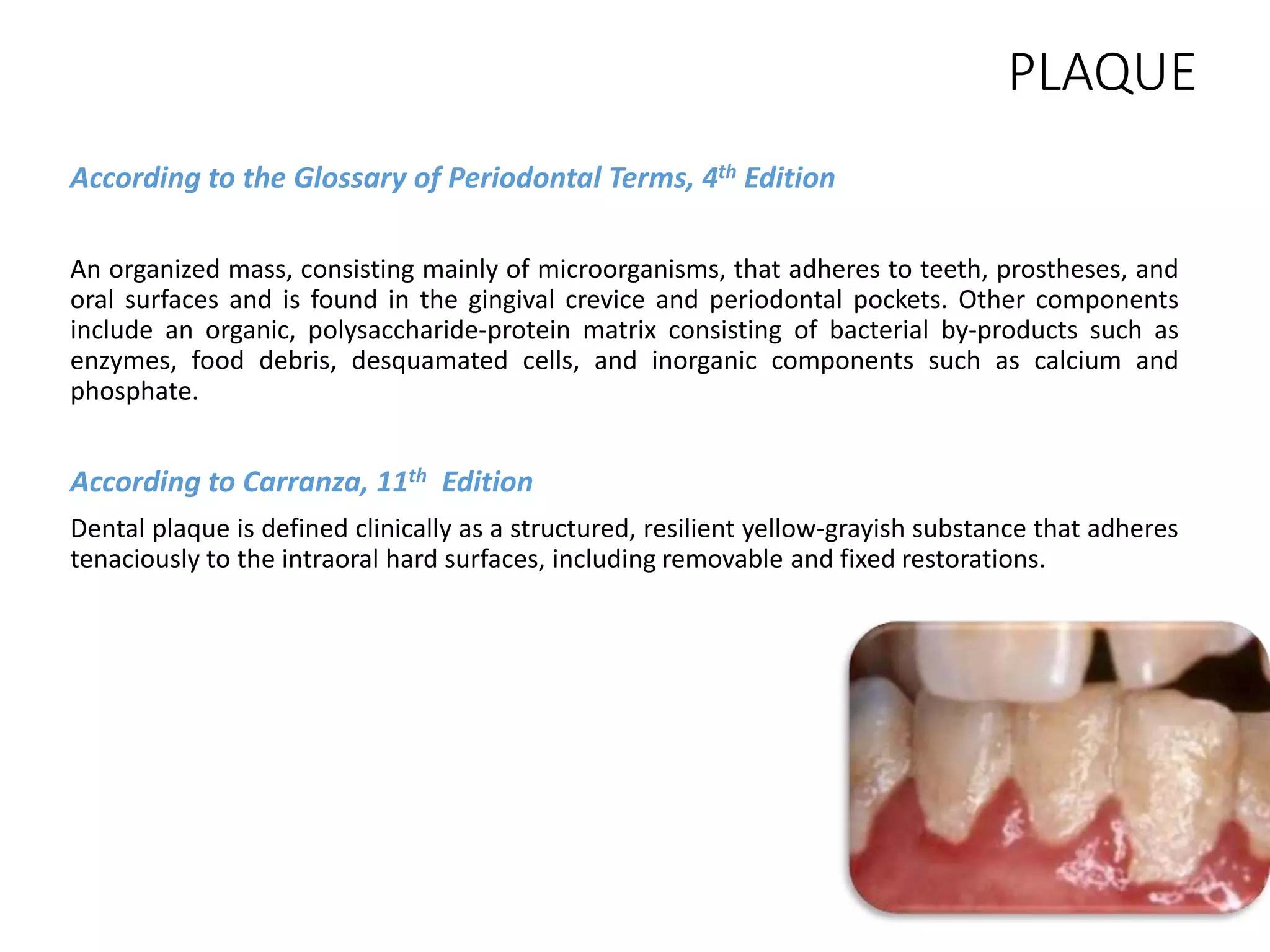
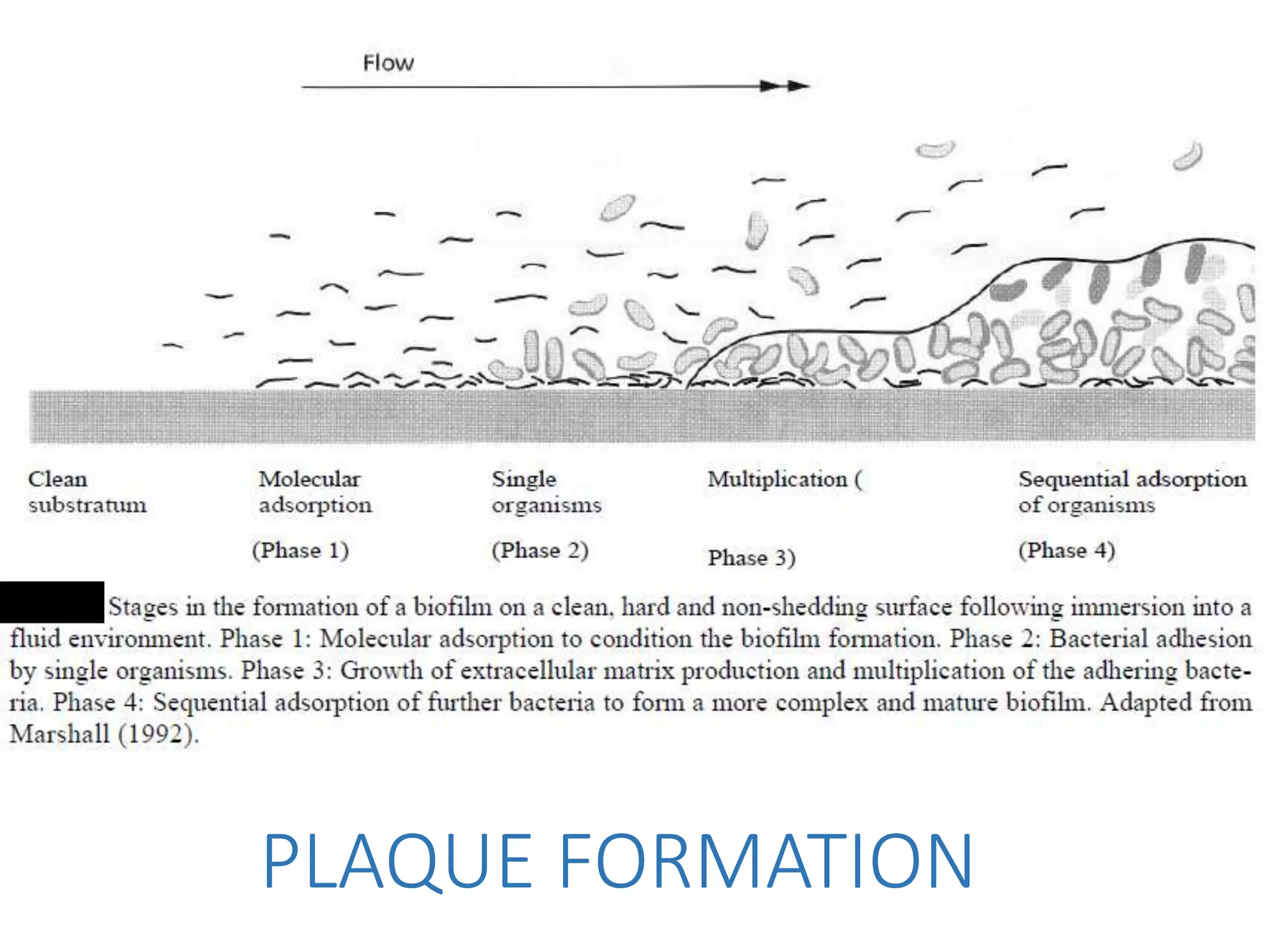
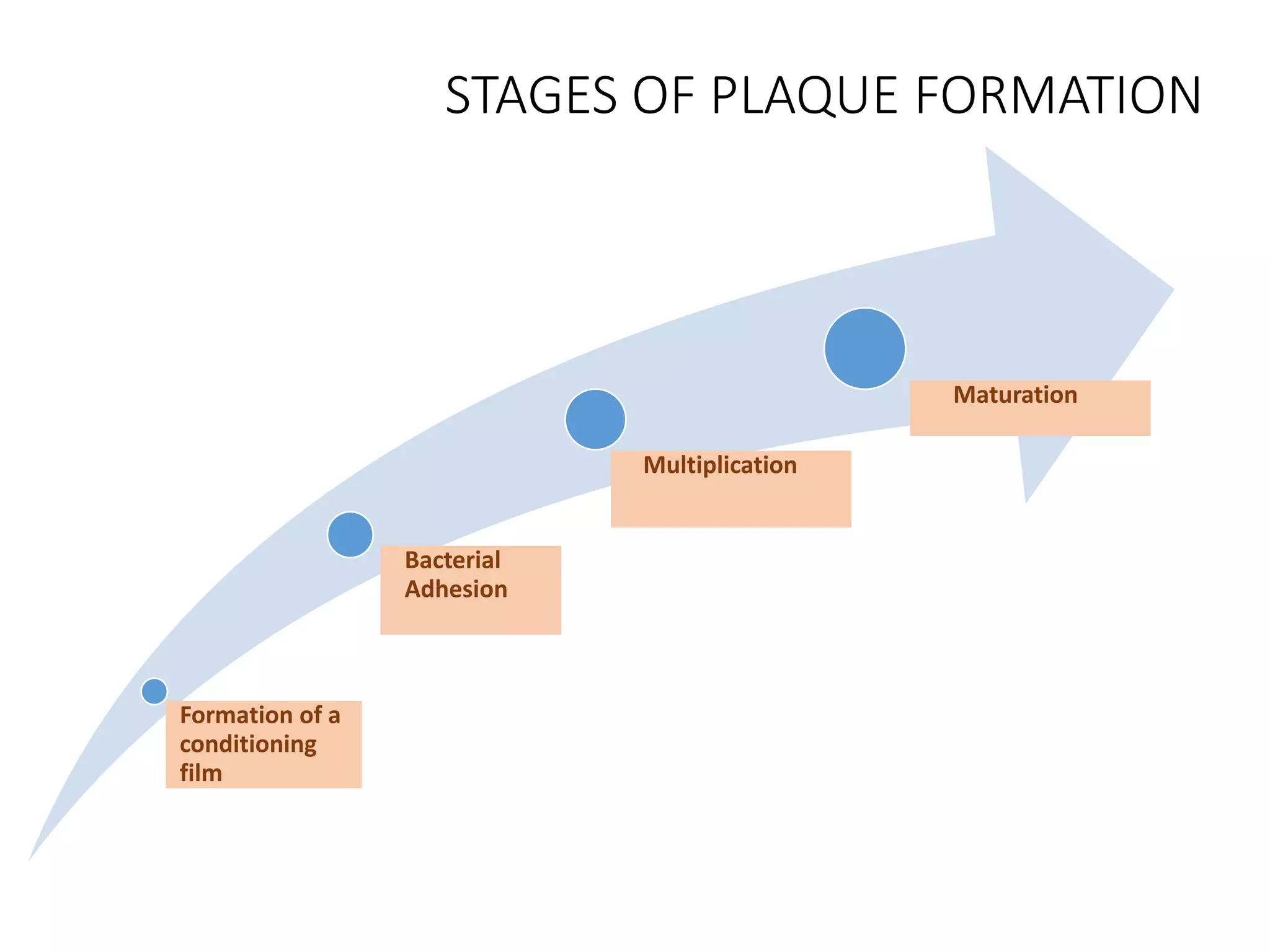
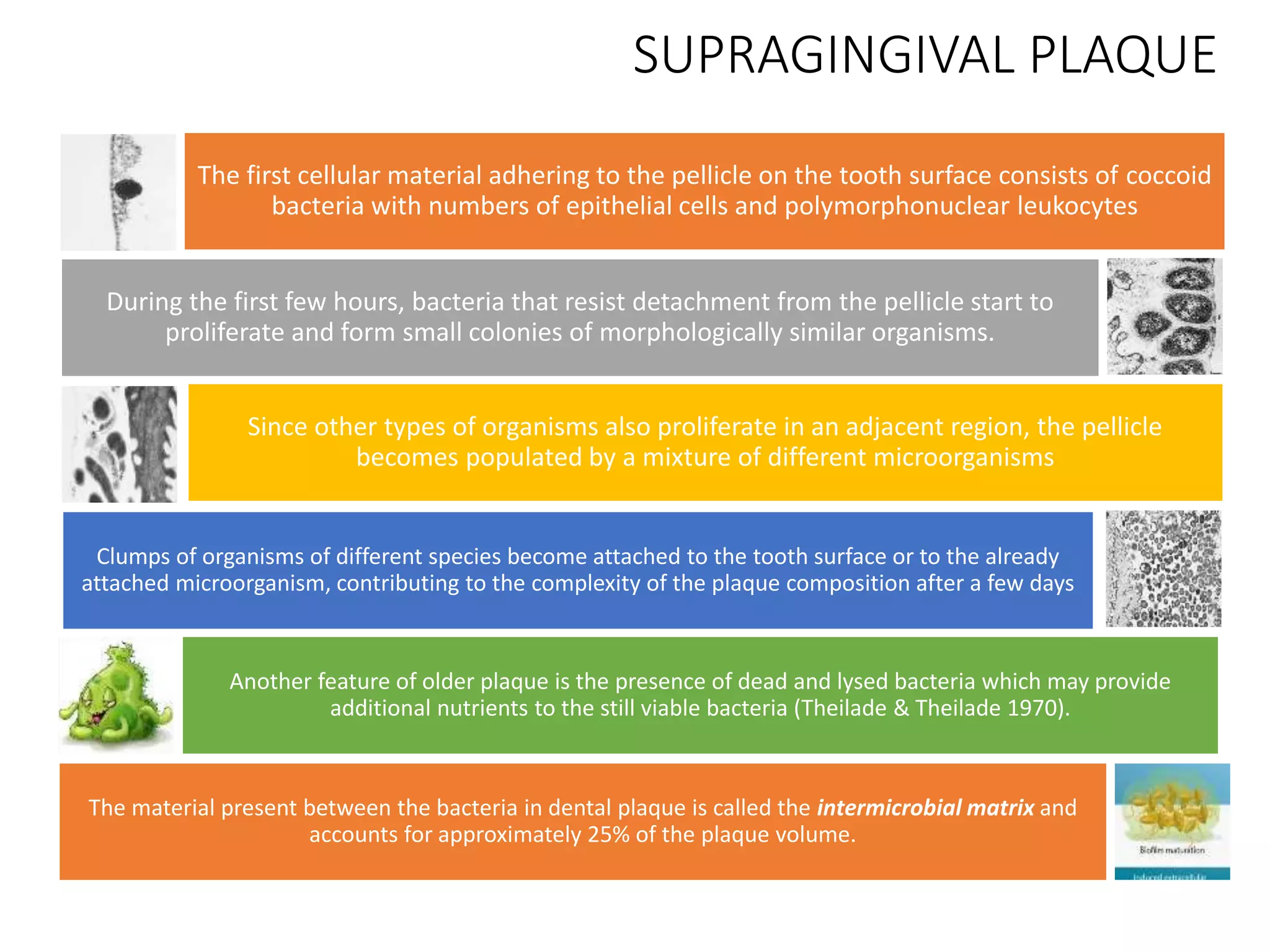
Dental plaque begins as a conditioning film that forms on teeth within minutes of cleaning. Bacteria then adhere through reversible and irreversible binding. As bacteria multiply, they synthesize extracellular polymeric substances to form a biofilm matrix. Co-adhesion and co-aggregation allow more bacteria to attach, leading to microcolony formation. Over time, this results in a mature dental plaque biofilm embedded within the matrix on the tooth surface.