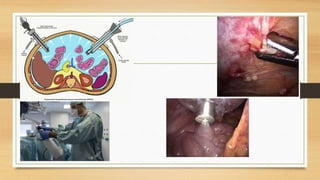
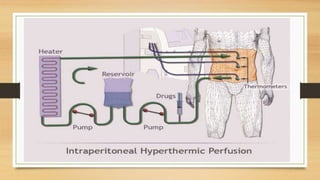
Pressurized Intra Peritoneal Aerosol Chemotherapy (PIPAC) is a minimally invasive technique that distributes chemotherapy as an aerosol into the abdominal cavity during laparoscopy. It shows superior drug distribution to peritoneal tumors compared to HIPEC. PIPAC can induce tumor regression in advanced, resistant peritoneal cancers and improve quality of life. It has few complications and can be repeated multiple times with low toxicity. PIPAC may provide an additional treatment option for symptom control in patients with stage IV cancers and peritoneal metastases.