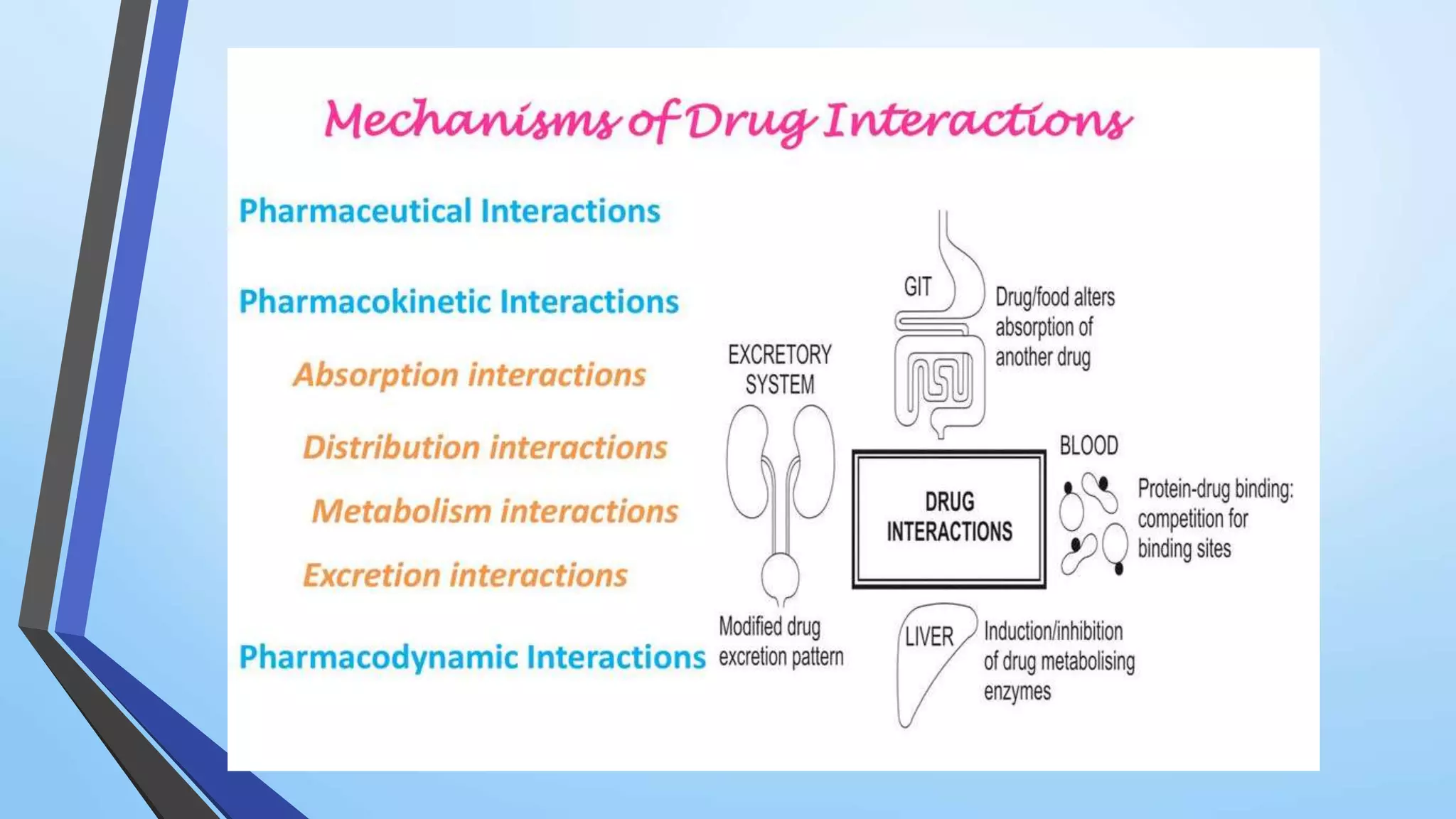
The document discusses pharmacokinetic drug interactions, defining them as alterations in the pharmacological activity of one drug due to the presence of another substance. It outlines various types of interactions, such as drug-drug and drug-food interactions, and explains mechanisms like absorption, distribution, metabolism, and elimination that can influence these interactions. Factors contributing to drug interactions include multiple drug therapy, prescribers, and patient characteristics, emphasizing that these interactions are mostly undesirable but can be beneficial in certain cases.