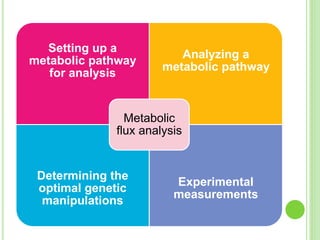
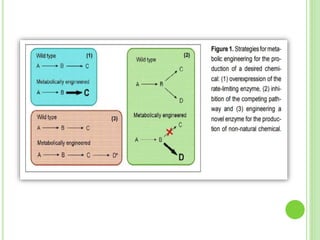
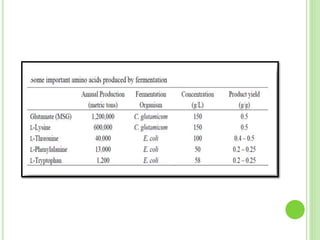
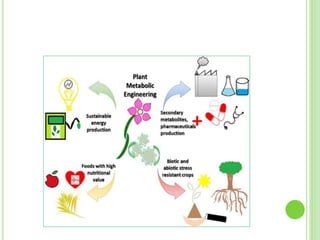
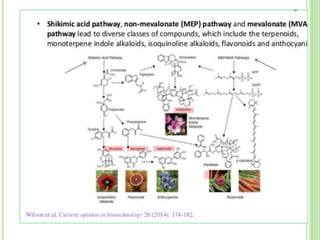
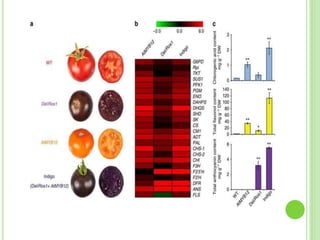
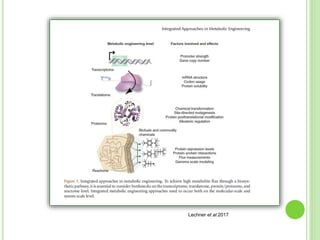
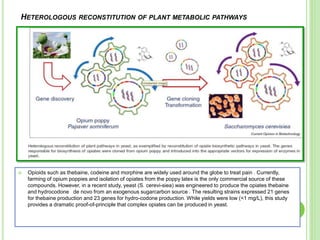
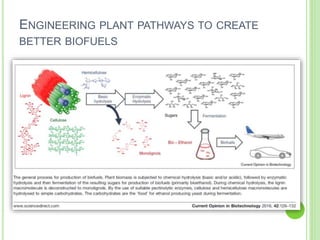
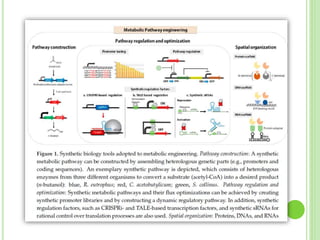
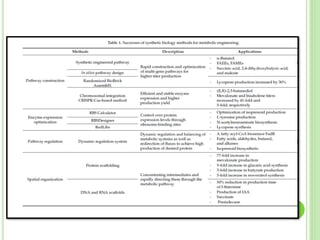
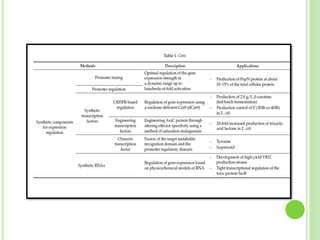
This document discusses metabolic engineering and summarizes key points about manipulating metabolic pathways in organisms. Metabolic engineering involves genetically modifying organisms to modulate their metabolism and produce desired products. It can be done by directly manipulating genes encoding enzymes in pathways or indirectly altering regulatory pathways. The document outlines several approaches to metabolic engineering, including overexpressing rate-limiting enzymes, inhibiting competing pathways, and expressing heterologous genes from other organisms. It also summarizes applications of metabolic engineering for producing amino acids, antibiotics, and manipulating plant metabolism to create foods with improved nutrients or biofuel properties.