Embed presentation
Downloaded 49 times
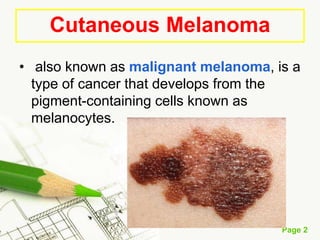









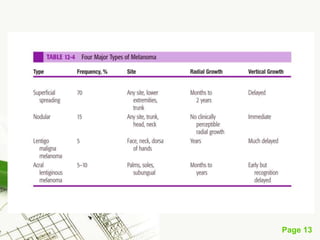

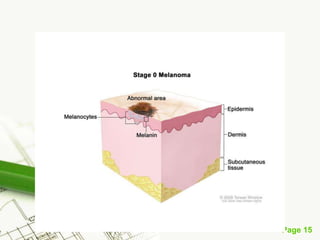
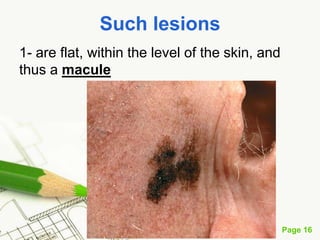
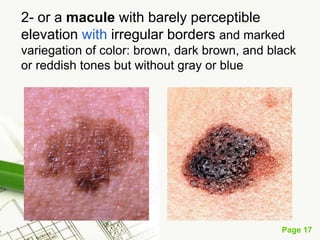






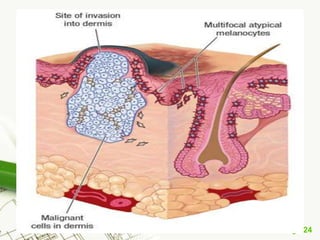

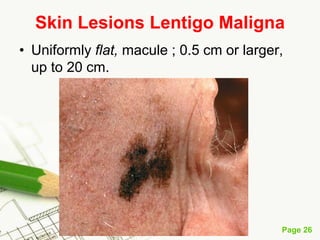
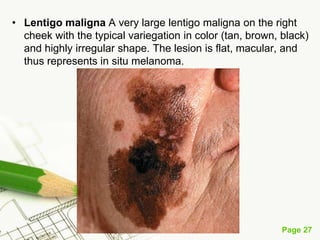
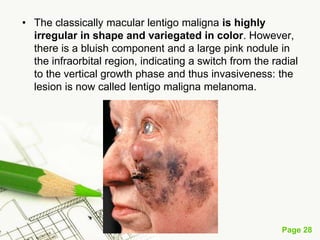
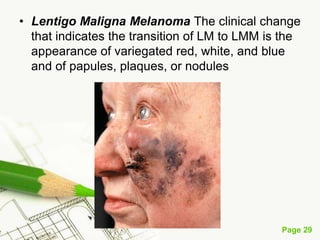



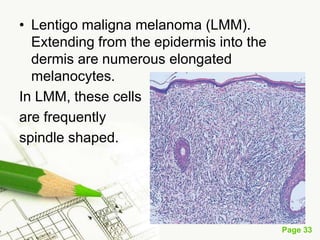

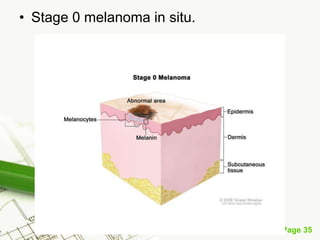
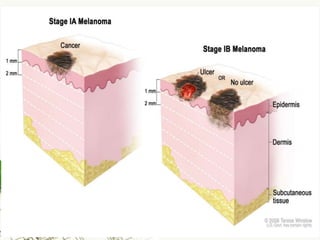

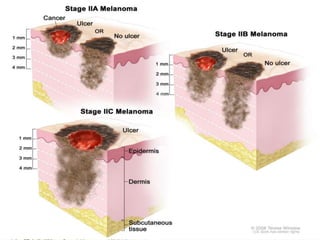
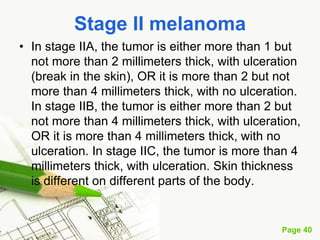
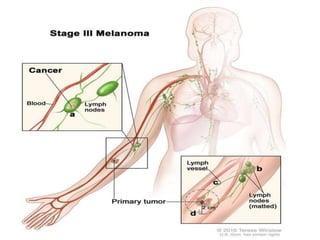
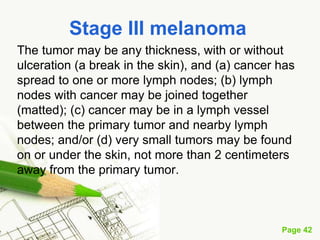
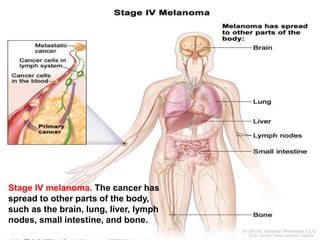
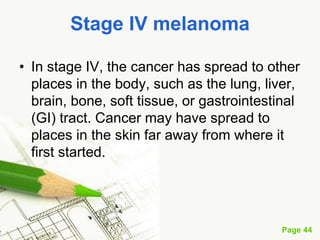




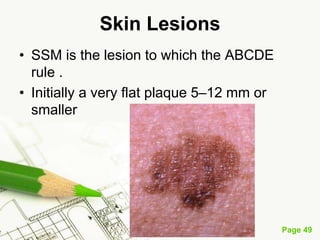


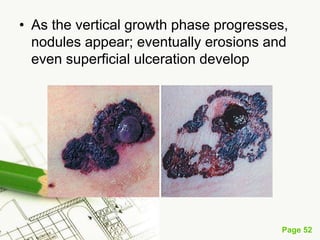







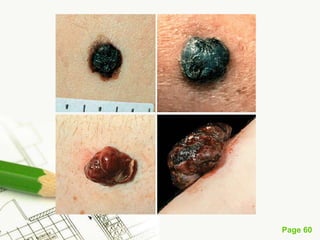


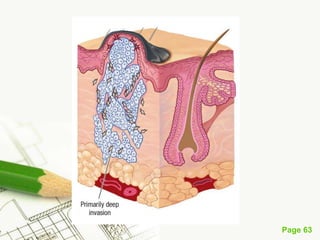






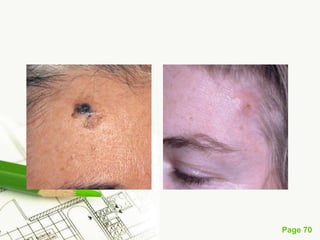


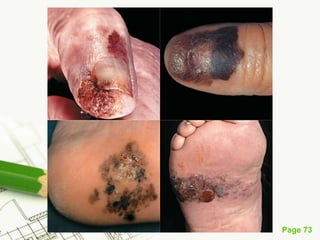




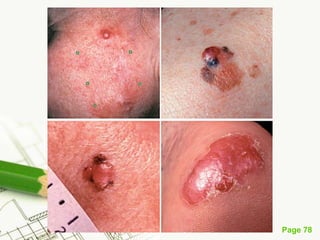




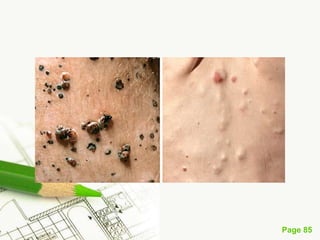






Cutaneous melanoma, a cancer originating from melanocytes, can be classified into various types, each with distinct clinical features and growth patterns. The etiology is not fully understood, but factors like genetic predisposition and sun exposure play significant roles in its development. Clinical recognition relies on the ABCDE rule, and management strategies vary based on the melanoma's stage and type.


























































































