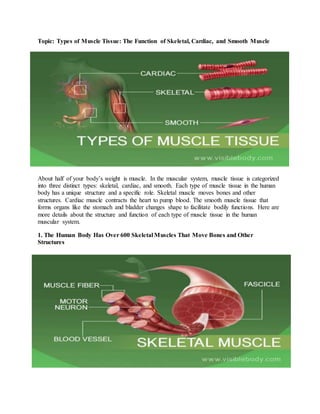
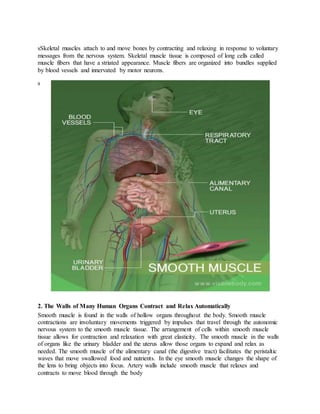
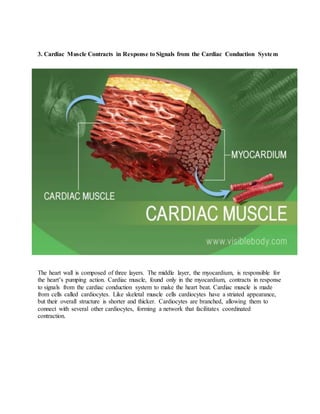
This document discusses the three main types of muscle tissue in the human body: skeletal, cardiac, and smooth muscle. It provides details on the structure and function of each type. Skeletal muscle moves bones and is striated. Cardiac muscle contracts the heart and has a branched, networked structure. Smooth muscle lines organs and contracts involuntarily. The document also covers muscle atrophy, which is a loss of mass, and hypertrophy, which is an increase in mass. Atrophy can result from disuse, disease, or aging, while hypertrophy involves growth factors, hormones, and satellite stem cells responding to exercise.