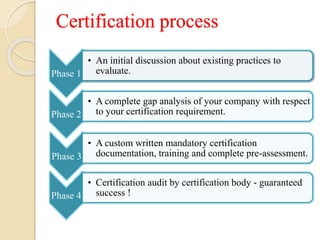
The document presents ISO 13485:2016, which outlines quality management system requirements for medical devices to ensure consistent customer satisfaction and regulatory compliance. It highlights the differences from ISO 9001, emphasizing effective implementation over continual improvement and specifics on risk management, documentation, and traceability. The certification process involves multiple phases to ensure thorough preparation and support for organizations seeking compliance.