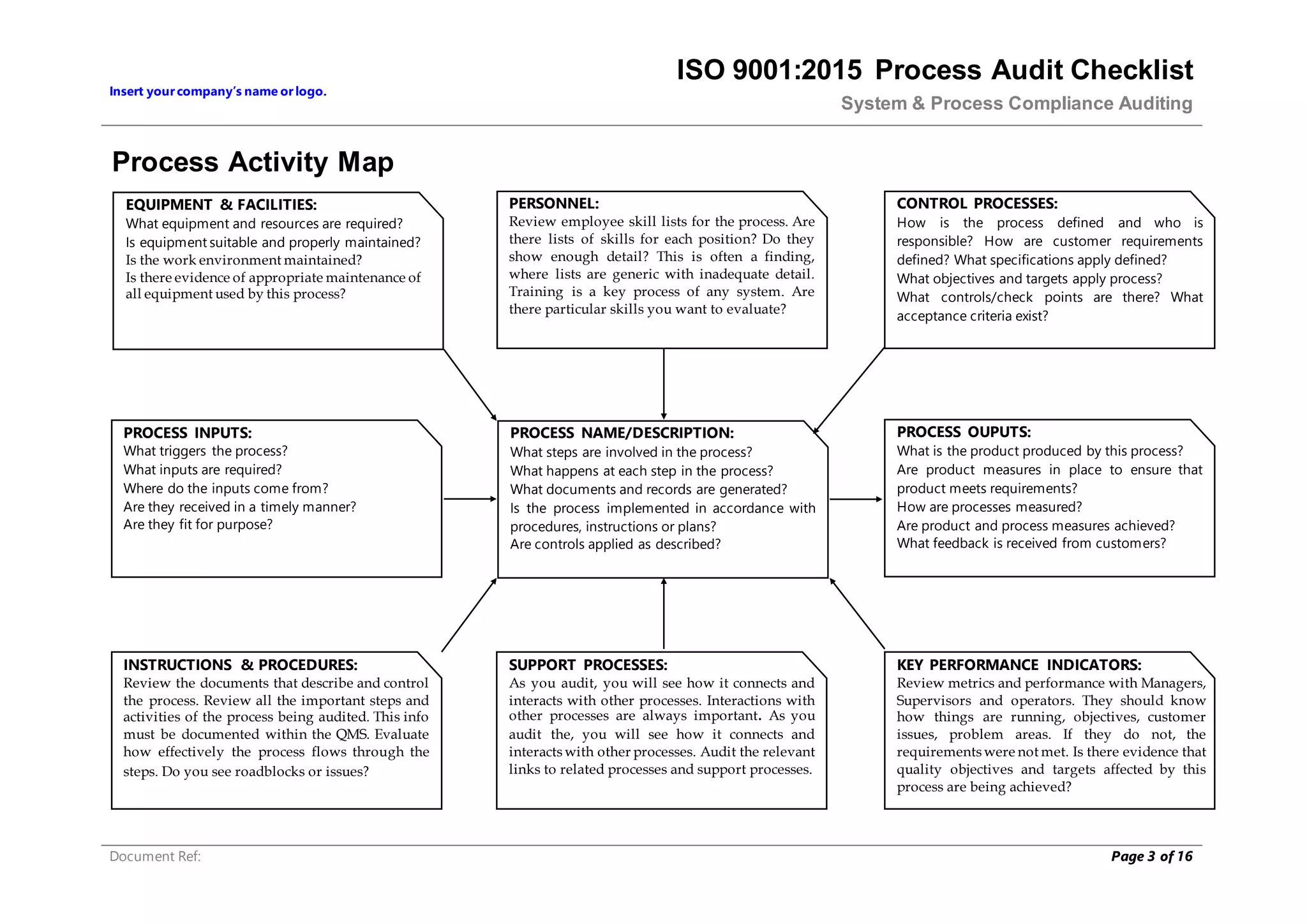
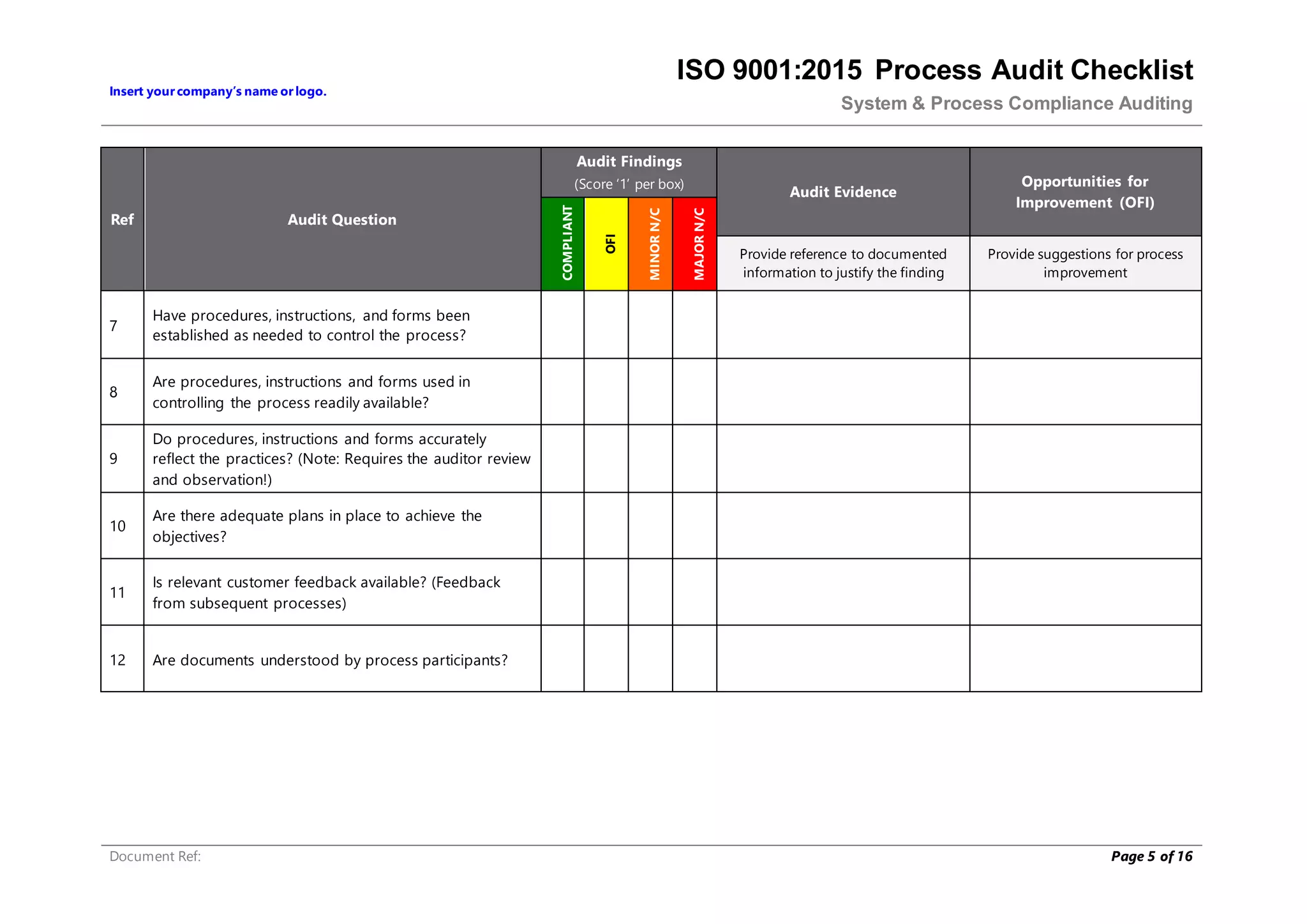
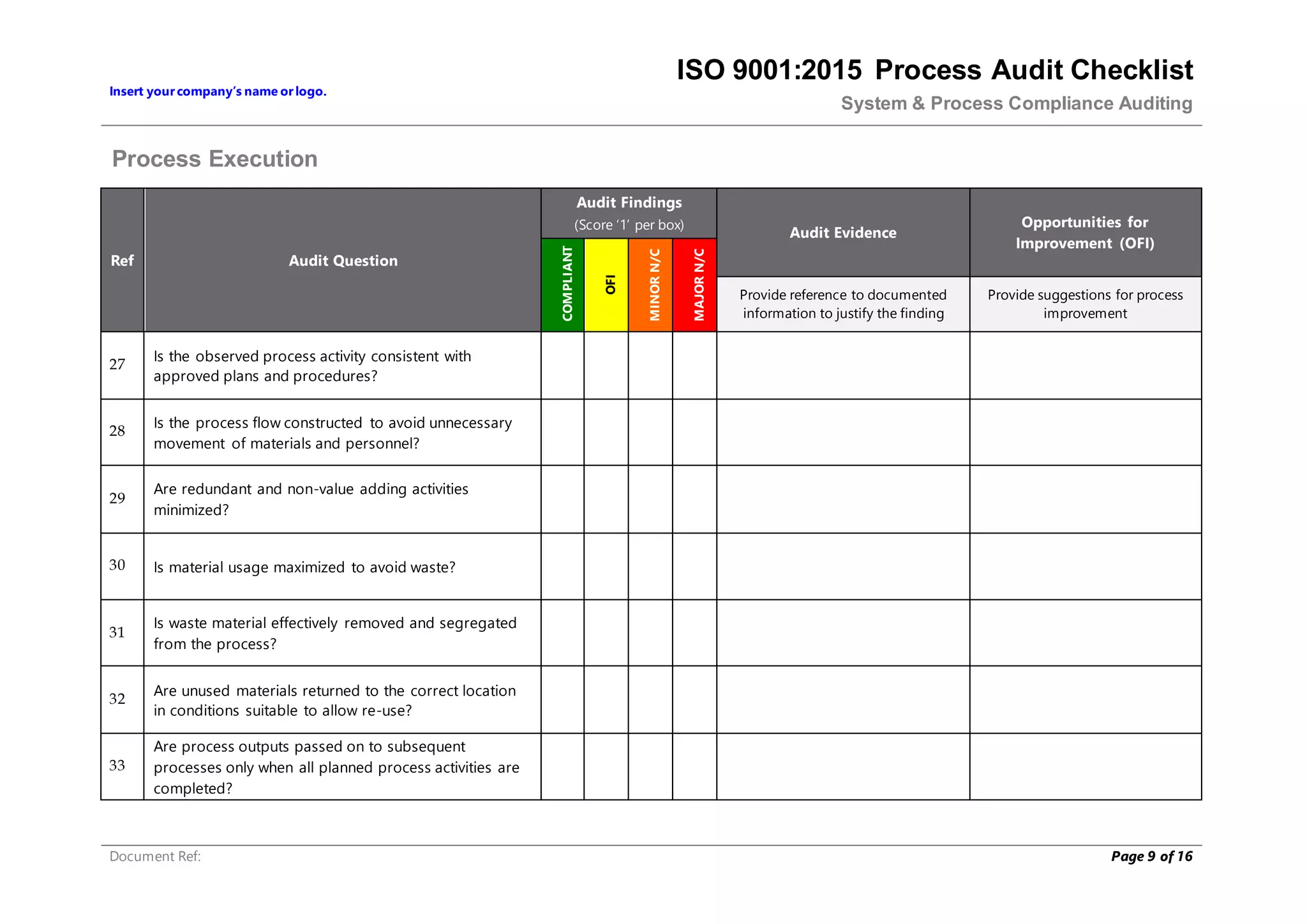
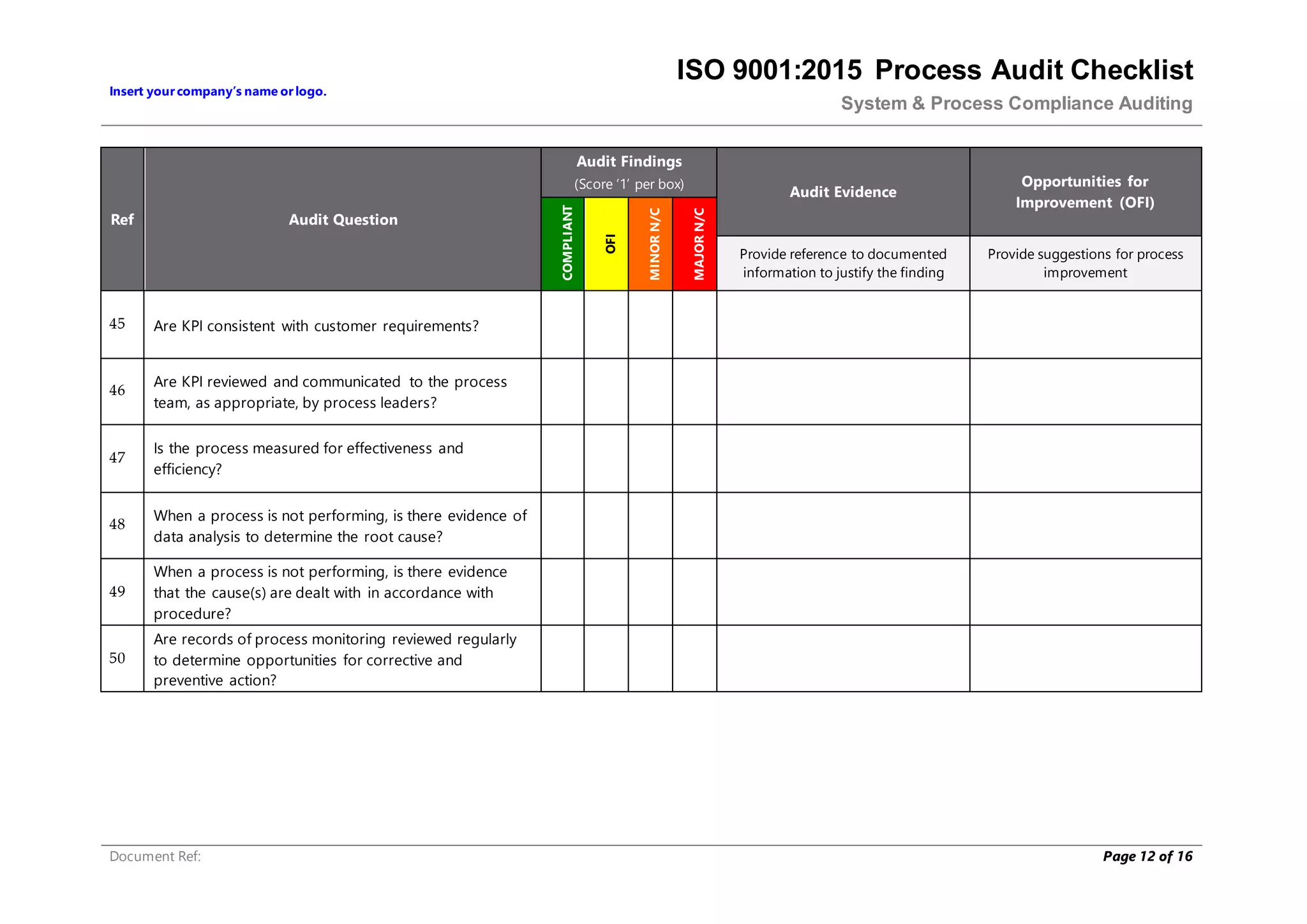
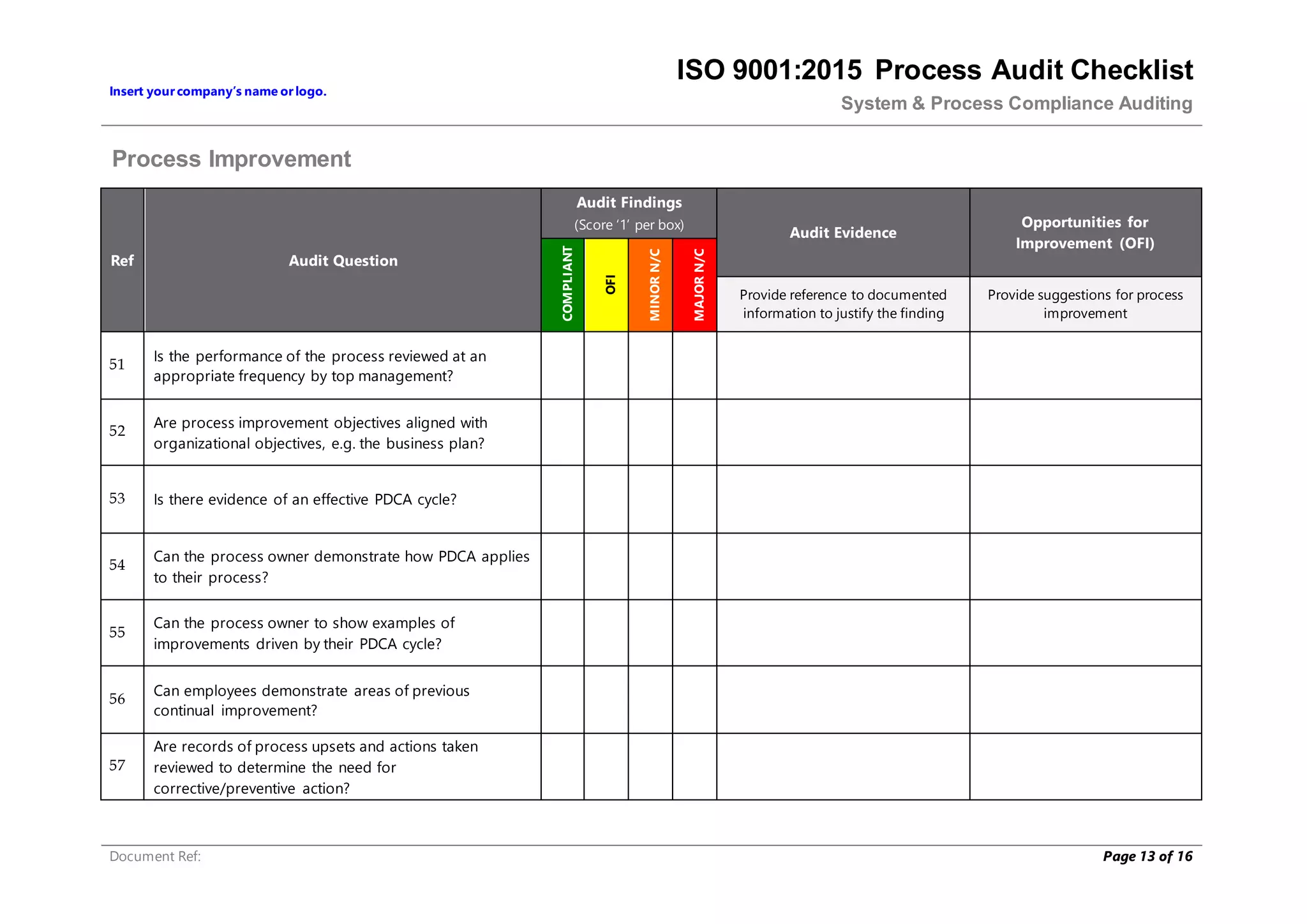
This document provides a checklist for auditing processes to assess compliance with ISO 9001:2015. The checklist includes questions addressing process definition, resources, execution, monitoring, and improvement. Scoring criteria are provided to rate audit findings as compliant, opportunity for improvement, minor nonconformance, or major nonconformance. The checklist is intended to ensure audits are conducted systematically and consistently.