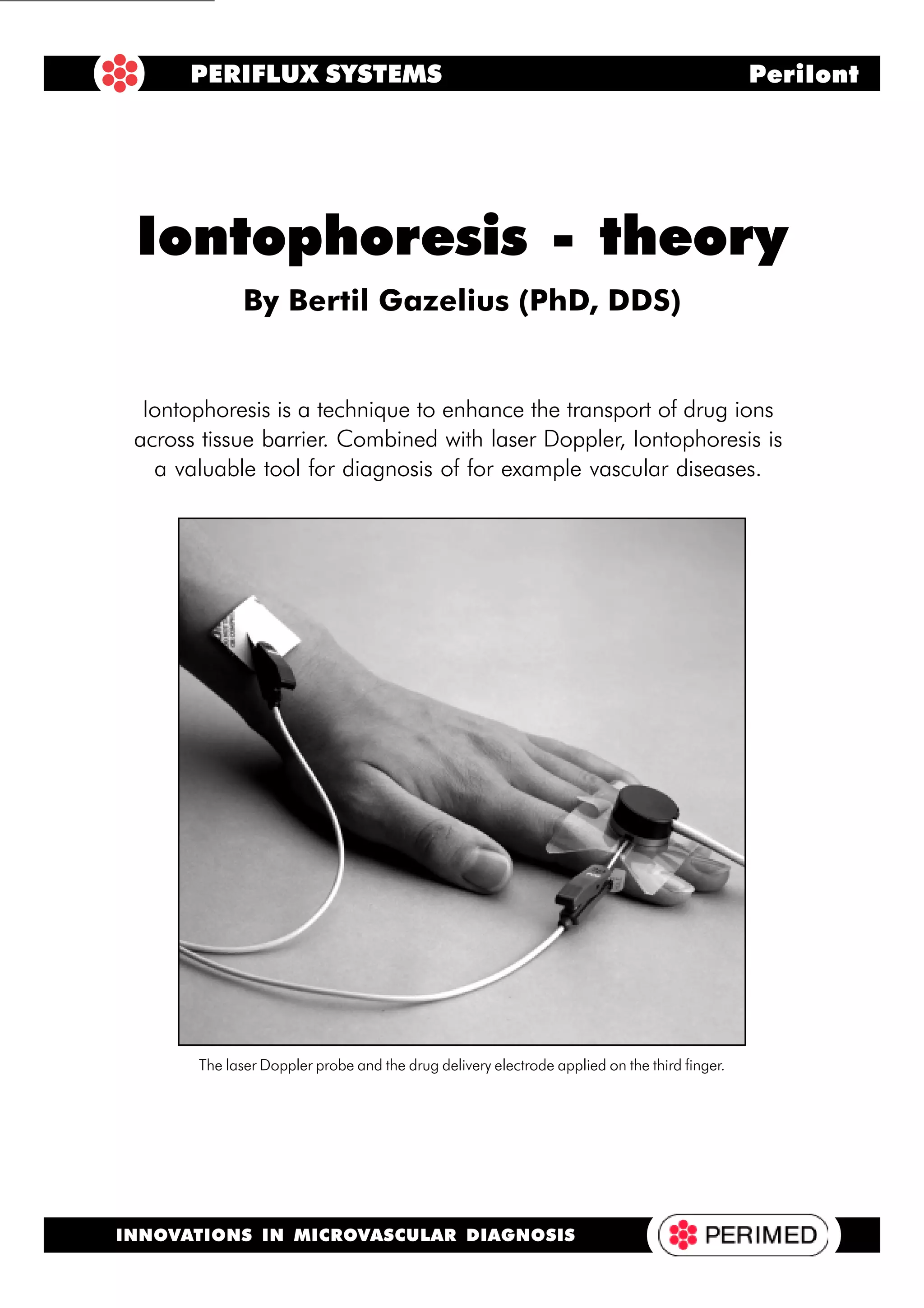
This document discusses iontophoresis, a technique used to enhance drug delivery across tissue barriers like skin using a small electric current. Iontophoresis works by repelling charged drug ions from an electrode on the skin surface, driving them into underlying tissues. The document outlines several key factors that influence iontophoresis, including drug properties, current strength, pH, concentration, molecular size, and use of continuous versus pulsed current. When combined with laser Doppler monitoring, iontophoresis can be used as a diagnostic tool to study the effects of drugs on microcirculation.