Heat of formation by substances
•
1 like•1,244 views
This document provides a table of standard enthalpies of formation (ΔHformation) in kJ/mol for various common substances. It notes that Hess's law can be used to calculate the standard enthalpy of a reaction (ΔHrx) from the standard enthalpies of formation of the products and reactants. The table lists the ΔHformation values for over 30 different elements and compounds.
Report
Share
Report
Share
Download to read offline
Recommended
Heat of formation by reactions

This document provides a standard enthalpy of formation table with the chemical reactions and corresponding enthalpy of formation values (in kJ/mol) for many common substances. The table lists the chemical formula, name of the substance, and the standard enthalpy of formation for reactions such as the formation of aluminum oxide, carbon monoxide, methane, and more. The table acts as a reference for determining enthalpy change of reactions using Hess's law by looking up the standard enthalpies of formation of products and reactants.
Tabla de potenciales redox

This document discusses three tables that are part of an electrochemical series. Table 1 provides an alphabetical list of reduction reactions and their standard reduction potentials (E° values) in volts. Table 2 lists reactions that have positive E° values relative to the standard hydrogen electrode, in order of increasing potential. Table 3 lists reactions with negative E° values relative to the standard hydrogen electrode, in order of decreasing potential. The document notes that the reliability of the E° values varies and is sometimes subject to revision.
Chemical reactions in daily life

This chemical equation shows methane gas (CH4) reacting with oxygen gas (O2) to produce carbon dioxide gas (CO2) and water vapor (H2O). The reactants methane and oxygen react in a 2:1 ratio to yield the products carbon dioxide and water.
Balancing chemical equations

This document provides a step-by-step example of balancing a chemical equation. It starts with an unbalanced equation for the combustion of ethane, C2H6, with oxygen. It then balances the atoms on both sides of the equation, showing that the balanced equation is C2H6 (g) + O2 (g) → 2CO2 (g) + 3H2O (g). Finally, it scales up the balanced equation by multiplying coefficients to get the fully balanced equation: 2C2H6 (g) + 7O2 (g) → 4CO2 (g) + 6H2O (g).
Al

This document lists chemical reactions involving aluminum and its compounds. Aluminum reacts with oxygen to form aluminum oxide, with water to form aluminum hydroxide, and with acids like hydrochloric acid and sulfuric acid to form aluminum salts. Aluminum also reacts with elements like chlorine, nitrogen, sulfur, and carbon to form various aluminum compounds. Aluminum hydroxide can be converted to aluminum oxide by removing water.
IB Chemistry on Born Haber Cycle and Lattice Enthalpy

The document describes the Born-Haber cycle process for determining the lattice enthalpy of various ionic compounds such as LiCl, NaCl, KCl, NaBr, NaF, and NaH. The Born-Haber cycle involves breaking down the ionic compound into gaseous ions and then measuring the standard enthalpy changes associated with steps such as atomization, ionization, and electron affinity which are used to calculate the unknown lattice enthalpy value. Diagrams of the Born-Haber cycle are provided for each example compound.
C20 Review Unit 02 Chemical Reactions

1) A chemical reaction can be differentiated from a phase change or nuclear change based on whether it involves a change in chemical composition, state of matter, or type of element.
2) In a chemical reaction, reactants yield products through the rearrangement of atoms. Chemical equations use symbols to represent the reactants and products.
3) Evidence that a chemical reaction occurred includes the release or absorption of energy, production of a gas, formation of a precipitate, or a color change.
Chem Unit6

Stoichiometry is the quantitative relationship between reactants and products in chemical reactions. Hess's law states that the enthalpy change of a reaction is independent of the pathway and depends only on the initial and final states. Using Hess's law, one can determine the enthalpy change of a reaction by adding the enthalpy changes of multiple steps that combine to give the overall reaction. The standard enthalpy of formation of a compound is the enthalpy change when one mole of the substance is formed from its elements in their standard states.
Recommended
Heat of formation by reactions

This document provides a standard enthalpy of formation table with the chemical reactions and corresponding enthalpy of formation values (in kJ/mol) for many common substances. The table lists the chemical formula, name of the substance, and the standard enthalpy of formation for reactions such as the formation of aluminum oxide, carbon monoxide, methane, and more. The table acts as a reference for determining enthalpy change of reactions using Hess's law by looking up the standard enthalpies of formation of products and reactants.
Tabla de potenciales redox

This document discusses three tables that are part of an electrochemical series. Table 1 provides an alphabetical list of reduction reactions and their standard reduction potentials (E° values) in volts. Table 2 lists reactions that have positive E° values relative to the standard hydrogen electrode, in order of increasing potential. Table 3 lists reactions with negative E° values relative to the standard hydrogen electrode, in order of decreasing potential. The document notes that the reliability of the E° values varies and is sometimes subject to revision.
Chemical reactions in daily life

This chemical equation shows methane gas (CH4) reacting with oxygen gas (O2) to produce carbon dioxide gas (CO2) and water vapor (H2O). The reactants methane and oxygen react in a 2:1 ratio to yield the products carbon dioxide and water.
Balancing chemical equations

This document provides a step-by-step example of balancing a chemical equation. It starts with an unbalanced equation for the combustion of ethane, C2H6, with oxygen. It then balances the atoms on both sides of the equation, showing that the balanced equation is C2H6 (g) + O2 (g) → 2CO2 (g) + 3H2O (g). Finally, it scales up the balanced equation by multiplying coefficients to get the fully balanced equation: 2C2H6 (g) + 7O2 (g) → 4CO2 (g) + 6H2O (g).
Al

This document lists chemical reactions involving aluminum and its compounds. Aluminum reacts with oxygen to form aluminum oxide, with water to form aluminum hydroxide, and with acids like hydrochloric acid and sulfuric acid to form aluminum salts. Aluminum also reacts with elements like chlorine, nitrogen, sulfur, and carbon to form various aluminum compounds. Aluminum hydroxide can be converted to aluminum oxide by removing water.
IB Chemistry on Born Haber Cycle and Lattice Enthalpy

The document describes the Born-Haber cycle process for determining the lattice enthalpy of various ionic compounds such as LiCl, NaCl, KCl, NaBr, NaF, and NaH. The Born-Haber cycle involves breaking down the ionic compound into gaseous ions and then measuring the standard enthalpy changes associated with steps such as atomization, ionization, and electron affinity which are used to calculate the unknown lattice enthalpy value. Diagrams of the Born-Haber cycle are provided for each example compound.
C20 Review Unit 02 Chemical Reactions

1) A chemical reaction can be differentiated from a phase change or nuclear change based on whether it involves a change in chemical composition, state of matter, or type of element.
2) In a chemical reaction, reactants yield products through the rearrangement of atoms. Chemical equations use symbols to represent the reactants and products.
3) Evidence that a chemical reaction occurred includes the release or absorption of energy, production of a gas, formation of a precipitate, or a color change.
Chem Unit6

Stoichiometry is the quantitative relationship between reactants and products in chemical reactions. Hess's law states that the enthalpy change of a reaction is independent of the pathway and depends only on the initial and final states. Using Hess's law, one can determine the enthalpy change of a reaction by adding the enthalpy changes of multiple steps that combine to give the overall reaction. The standard enthalpy of formation of a compound is the enthalpy change when one mole of the substance is formed from its elements in their standard states.
IB Chemistry on Born Haber Cycle and Lattice Enthalpy

The document provides information on the Born-Haber cycle and how it can be used to calculate lattice enthalpy for various ionic compounds. It gives step-by-step explanations of standard enthalpy changes used in the Born-Haber cycle calculations for ionic compounds such as LiCl, NaCl, KCl, NaBr, NaF and NaH. Diagrams illustrate the multi-stage process of determining lattice enthalpy values that cannot be measured directly through experimentation.
Extra redox problems

This document provides additional practice problems for balancing oxidation-reduction reactions in acidic and basic solutions. The problems cover reactions involving silver, zinc, chromium, phosphorus, manganese, chlorine, iron, hydrogen peroxide, and copper species. Balanced equations are provided as answers for each reaction.
013 science a-net(เคมี)

This document contains 49 multiple choice questions covering various topics in chemistry. The questions assess knowledge of topics including: chemical formulas and electron configurations; lattice energies; gas laws; enthalpy of formation; acid-base reactions; stoichiometry calculations; and organic reactions. The multiple choice options provided for each question require application of chemistry concepts to analyze and solve various problems.
Electrochemical series

The document discusses three tables that provide standard reduction potentials (E° values) for various electrochemical reactions. Table 1 lists elements alphabetically by symbol. Table 2 lists reactions with positive E° values relative to the standard hydrogen electrode, in order of increasing potential. Table 3 lists reactions with negative E° values relative to the standard hydrogen electrode, in order of decreasing potential. The document notes some reactions have more reliable E° values than others based on number of significant figures.
IB Chemistry on Redox, Oxidizing, Reducing Agents and writing half redox equa...

This document provides a tutorial on redox reactions, oxidation states, and half reactions. It begins by defining redox as involving both oxidation and reduction, which is the loss or gain of electrons. It then discusses oxidation states (also called oxidation numbers), which are assigned to atoms in compounds to keep track of electrons. Rules are provided for assigning oxidation states based on electronegativity. Redox reactions involve a change in oxidation states between reactants and products. Examples of assigning oxidation states to elements in compounds are also given.
IB Chemistry on Hess's Law, Enthalpy Formation and Combustion

1) Hess's law states that the enthalpy change of a reaction is independent of the pathway and is equal to the sum of the enthalpy changes of the steps.
2) Standard enthalpy changes of formation (ΔHf°) can be used to calculate the enthalpy change (ΔH°) of a reaction by adding the standard enthalpies of formation of products and subtracting the standard enthalpies of formation of reactants.
3) For the reaction 2H2S + SO2 → 3S + 2H2O, the calculated standard enthalpy change is -234 kJ/mol.
Inorganic Chemistry : Electrochemistry

Deep explanation on electrochemistry :)
This is actually Malaysian Higher studies syllabus :D
Good Luck
Balancing equations

The document discusses balancing chemical equations by ensuring that the same number and type of atoms appear on both sides of the reaction arrow. It provides examples of balancing a variety of chemical equations involving reactants like Mg, HCl, SO2, and products like MgCl2, SO3, NO2. Students are encouraged to practice balancing many different types of equations involving metals, nonmetals, acids, bases, and other chemicals.
Balanceo de redox, ion electro y metodo algebraico

The document contains 10 chemical equations that need to be balanced using the method of ionic half-reactions. It provides the chemical formulas and oxidation numbers for each element in each equation to allow for balancing the equations.
IB Chemistry SL Redox 

This document discusses oxidation and reduction reactions. It defines oxidation as the loss of electrons and reduction as the gain of electrons. Examples of oxidation-reduction reactions are given. Oxidation states are defined as the apparent charge of an atom in a compound. Rules for assigning oxidation states are provided. The document also discusses oxidizing and reducing agents, transition metal compounds, and the stock nomenclature system for naming transition metal ions based on oxidation states. Finally, the activity series is introduced which ranks the reactivity of metals from most reactive to least reactive.
Bab6 persamaan kimia

The document discusses equalizing reaction coefficients in chemical equations. It provides examples of balancing equations for reactions such as nitrogen and hydrogen forming ammonia, aluminum and oxygen forming aluminum oxide, and copper reacting with nitric acid to form copper nitrate. It also categorizes different types of chemical reactions such as combination, decomposition, substitution, and neutralization reactions.
Tutorial 2 part 2 answer key

This document contains 5 examples of stoichiometry calculations involving chemical reactions. Each example provides the balanced chemical equation, the relevant atomic masses, and step-by-step working to calculate the amounts of reactants and products based on the mole ratios in the balanced equations. The examples calculate amounts of unreacted iron, masses of products formed from decomposing calcium carbonate, amounts of sodium hydroxide and water formed from a reaction of sodium hydroxide and sulfuric acid, tonnes of carbon and copper produced from a copper smelting reaction, and tonnes of carbon needed to reduce iron oxide ore.
Calculations using standard enthalpies of formation

1. The document provides standard enthalpies of formation (ΔfH°) for various compounds and asks the reader to use these values to calculate the enthalpy change for 10 chemical reactions and processes.
2. It lists the standard enthalpies of formation for common compounds such as water, carbon dioxide, benzene, cyclohexane, calcium carbonate, quicklime, sulfur dioxide, sulfuric acid, ethanol, bromoethane and more.
3. The reader is to apply Hess's law and add or subtract the given standard enthalpies of formation to determine the enthalpy change for the 10 processes listed, such as the enthalpy of solution of hydrogen bromide gas and
Thermo kin ws complete

This document provides information on:
1) The solubility of various ionic compounds in water at standard temperature and pressure (SATP), categorizing them as having high solubility (≥0.1 mol/L) or low solubility (<0.1 mol/L).
2) Bond energies in kJ/mol for various bonds at SATP.
3) Specific heat capacities in J/(g∙°C) for various substances at SATP.
4) Standard enthalpy of formation (ΔH°f), standard entropy (S°), and standard Gibbs free energy of formation (ΔG°f) in kJ/mol for various inorganic and organic substances at
Are we there yet

The document describes Ishmael Masesane's research journey from classical chemistry to transfer hydrogenation of alkenes using palladium catalysis and various organic acids. It then discusses the development of enantioselective versions of this reaction using chiral organic acids like mandelic acid and tartaric acid. Finally, it outlines Masesane's approaches to synthesizing cyclic beta-amino acid derivatives using Diels-Alder reactions of furan with acrylates or maleic anhydride to form oxanorbornyl intermediates.
Chem 16 2 le answer key j4 feb 4 2011

This document contains questions about thermodynamics and electronic structure. Regarding thermodynamics, questions cover topics like exothermic and endothermic phase changes, standard enthalpies of formation, entropy, Gibbs free energy, and thermochemical equations. Regarding electronic structure, questions cover topics like electron configurations, quantum numbers, diamagnetism, isoelectronic species, and periodic trends.
Inorganic chemistry revision package solutions

The document describes several chemical reactions involving group VII elements and transition metals. It provides reaction equations to show how concentrated sulfuric acid reacts with potassium halides to produce hydrogen halides. It also describes how transition metal ions like iron and copper form colored complexes in aqueous solution due to d-d electron transitions. The reactions of cobalt ion as a catalyst in the oxidation of iodide by persulfate are also outlined.
Chem 16 2 le exam j4 feb 4 2011

This document contains 15 multiple choice questions about thermodynamics and 5 multiple choice questions about electronic structure. The thermodynamics questions cover topics like phase changes, entropy, Gibbs free energy, enthalpy of reaction, and thermochemistry calculations. The electronic structure questions cover topics like electron configuration, quantum numbers, and isoelectronic species.
Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress

This document provides thermodynamic quantities such as enthalpy (AH), Gibbs free energy (AG), and entropy (S°) for various substances at 298.15 K (25°C). It includes data for elements like aluminum, carbon, sulfur, hydrogen, oxygen, bromine, calcium, chlorine, cobalt, copper, chromium, iron, lithium, and sodium, as well as compounds including carbonates, chlorides, oxides, sulfates, and more. The values are given in units of kJ/mol, kJ/mol, and J/mol-K respectively for each substance.
สำเร๊จ

1. The document describes various chemical reactions involved in the production of ammonia, nitric acid, phosphoric acid, and other fertilizers. It outlines reaction equations and conditions.
2. Details are provided on production processes including the contact process for sulfuric acid and ammonia, as well as methods for making superphosphate from phosphate rock using sulfuric acid or phosphoric acid.
3. The document also covers reactions of hydrofluoric acid and how it is used to make hydrogen fluoride, as well as reactions of potassium compounds like potassium chloride and potassium sulfate. Blending and granulation steps in fertilizer production are briefly outlined.
อุตสาหกรรมปุ๋ย

1. The document describes several chemical reactions involved in the production of ammonia, including the Haber process and contact process.
2. It also discusses reactions for producing phosphoric acid and other phosphate compounds, including ones involving calcium fluoride and phosphorus pentoxide.
3. The production of potassium chloride and other potash fertilizers is also summarized, involving reactions between potassium chloride and sodium compounds like sodium chloride and sodium nitrate.
01

1. The document discusses various chemical reactions and processes involved in the production of fertilizers and other chemicals.
2. Key reactions include the contact process for producing ammonia and the Haber process for producing nitric acid. Sulfuric acid is also discussed as a key input.
3. Methods of blending fertilizers are described including bulk blending, granulation, and using various additives to improve properties of the fertilizer.
More Related Content
What's hot
IB Chemistry on Born Haber Cycle and Lattice Enthalpy

The document provides information on the Born-Haber cycle and how it can be used to calculate lattice enthalpy for various ionic compounds. It gives step-by-step explanations of standard enthalpy changes used in the Born-Haber cycle calculations for ionic compounds such as LiCl, NaCl, KCl, NaBr, NaF and NaH. Diagrams illustrate the multi-stage process of determining lattice enthalpy values that cannot be measured directly through experimentation.
Extra redox problems

This document provides additional practice problems for balancing oxidation-reduction reactions in acidic and basic solutions. The problems cover reactions involving silver, zinc, chromium, phosphorus, manganese, chlorine, iron, hydrogen peroxide, and copper species. Balanced equations are provided as answers for each reaction.
013 science a-net(เคมี)

This document contains 49 multiple choice questions covering various topics in chemistry. The questions assess knowledge of topics including: chemical formulas and electron configurations; lattice energies; gas laws; enthalpy of formation; acid-base reactions; stoichiometry calculations; and organic reactions. The multiple choice options provided for each question require application of chemistry concepts to analyze and solve various problems.
Electrochemical series

The document discusses three tables that provide standard reduction potentials (E° values) for various electrochemical reactions. Table 1 lists elements alphabetically by symbol. Table 2 lists reactions with positive E° values relative to the standard hydrogen electrode, in order of increasing potential. Table 3 lists reactions with negative E° values relative to the standard hydrogen electrode, in order of decreasing potential. The document notes some reactions have more reliable E° values than others based on number of significant figures.
IB Chemistry on Redox, Oxidizing, Reducing Agents and writing half redox equa...

This document provides a tutorial on redox reactions, oxidation states, and half reactions. It begins by defining redox as involving both oxidation and reduction, which is the loss or gain of electrons. It then discusses oxidation states (also called oxidation numbers), which are assigned to atoms in compounds to keep track of electrons. Rules are provided for assigning oxidation states based on electronegativity. Redox reactions involve a change in oxidation states between reactants and products. Examples of assigning oxidation states to elements in compounds are also given.
IB Chemistry on Hess's Law, Enthalpy Formation and Combustion

1) Hess's law states that the enthalpy change of a reaction is independent of the pathway and is equal to the sum of the enthalpy changes of the steps.
2) Standard enthalpy changes of formation (ΔHf°) can be used to calculate the enthalpy change (ΔH°) of a reaction by adding the standard enthalpies of formation of products and subtracting the standard enthalpies of formation of reactants.
3) For the reaction 2H2S + SO2 → 3S + 2H2O, the calculated standard enthalpy change is -234 kJ/mol.
Inorganic Chemistry : Electrochemistry

Deep explanation on electrochemistry :)
This is actually Malaysian Higher studies syllabus :D
Good Luck
Balancing equations

The document discusses balancing chemical equations by ensuring that the same number and type of atoms appear on both sides of the reaction arrow. It provides examples of balancing a variety of chemical equations involving reactants like Mg, HCl, SO2, and products like MgCl2, SO3, NO2. Students are encouraged to practice balancing many different types of equations involving metals, nonmetals, acids, bases, and other chemicals.
Balanceo de redox, ion electro y metodo algebraico

The document contains 10 chemical equations that need to be balanced using the method of ionic half-reactions. It provides the chemical formulas and oxidation numbers for each element in each equation to allow for balancing the equations.
IB Chemistry SL Redox 

This document discusses oxidation and reduction reactions. It defines oxidation as the loss of electrons and reduction as the gain of electrons. Examples of oxidation-reduction reactions are given. Oxidation states are defined as the apparent charge of an atom in a compound. Rules for assigning oxidation states are provided. The document also discusses oxidizing and reducing agents, transition metal compounds, and the stock nomenclature system for naming transition metal ions based on oxidation states. Finally, the activity series is introduced which ranks the reactivity of metals from most reactive to least reactive.
Bab6 persamaan kimia

The document discusses equalizing reaction coefficients in chemical equations. It provides examples of balancing equations for reactions such as nitrogen and hydrogen forming ammonia, aluminum and oxygen forming aluminum oxide, and copper reacting with nitric acid to form copper nitrate. It also categorizes different types of chemical reactions such as combination, decomposition, substitution, and neutralization reactions.
Tutorial 2 part 2 answer key

This document contains 5 examples of stoichiometry calculations involving chemical reactions. Each example provides the balanced chemical equation, the relevant atomic masses, and step-by-step working to calculate the amounts of reactants and products based on the mole ratios in the balanced equations. The examples calculate amounts of unreacted iron, masses of products formed from decomposing calcium carbonate, amounts of sodium hydroxide and water formed from a reaction of sodium hydroxide and sulfuric acid, tonnes of carbon and copper produced from a copper smelting reaction, and tonnes of carbon needed to reduce iron oxide ore.
What's hot (12)
IB Chemistry on Born Haber Cycle and Lattice Enthalpy

IB Chemistry on Born Haber Cycle and Lattice Enthalpy
IB Chemistry on Redox, Oxidizing, Reducing Agents and writing half redox equa...

IB Chemistry on Redox, Oxidizing, Reducing Agents and writing half redox equa...
IB Chemistry on Hess's Law, Enthalpy Formation and Combustion

IB Chemistry on Hess's Law, Enthalpy Formation and Combustion
Balanceo de redox, ion electro y metodo algebraico

Balanceo de redox, ion electro y metodo algebraico
Similar to Heat of formation by substances
Calculations using standard enthalpies of formation

1. The document provides standard enthalpies of formation (ΔfH°) for various compounds and asks the reader to use these values to calculate the enthalpy change for 10 chemical reactions and processes.
2. It lists the standard enthalpies of formation for common compounds such as water, carbon dioxide, benzene, cyclohexane, calcium carbonate, quicklime, sulfur dioxide, sulfuric acid, ethanol, bromoethane and more.
3. The reader is to apply Hess's law and add or subtract the given standard enthalpies of formation to determine the enthalpy change for the 10 processes listed, such as the enthalpy of solution of hydrogen bromide gas and
Thermo kin ws complete

This document provides information on:
1) The solubility of various ionic compounds in water at standard temperature and pressure (SATP), categorizing them as having high solubility (≥0.1 mol/L) or low solubility (<0.1 mol/L).
2) Bond energies in kJ/mol for various bonds at SATP.
3) Specific heat capacities in J/(g∙°C) for various substances at SATP.
4) Standard enthalpy of formation (ΔH°f), standard entropy (S°), and standard Gibbs free energy of formation (ΔG°f) in kJ/mol for various inorganic and organic substances at
Are we there yet

The document describes Ishmael Masesane's research journey from classical chemistry to transfer hydrogenation of alkenes using palladium catalysis and various organic acids. It then discusses the development of enantioselective versions of this reaction using chiral organic acids like mandelic acid and tartaric acid. Finally, it outlines Masesane's approaches to synthesizing cyclic beta-amino acid derivatives using Diels-Alder reactions of furan with acrylates or maleic anhydride to form oxanorbornyl intermediates.
Chem 16 2 le answer key j4 feb 4 2011

This document contains questions about thermodynamics and electronic structure. Regarding thermodynamics, questions cover topics like exothermic and endothermic phase changes, standard enthalpies of formation, entropy, Gibbs free energy, and thermochemical equations. Regarding electronic structure, questions cover topics like electron configurations, quantum numbers, diamagnetism, isoelectronic species, and periodic trends.
Inorganic chemistry revision package solutions

The document describes several chemical reactions involving group VII elements and transition metals. It provides reaction equations to show how concentrated sulfuric acid reacts with potassium halides to produce hydrogen halides. It also describes how transition metal ions like iron and copper form colored complexes in aqueous solution due to d-d electron transitions. The reactions of cobalt ion as a catalyst in the oxidation of iodide by persulfate are also outlined.
Chem 16 2 le exam j4 feb 4 2011

This document contains 15 multiple choice questions about thermodynamics and 5 multiple choice questions about electronic structure. The thermodynamics questions cover topics like phase changes, entropy, Gibbs free energy, enthalpy of reaction, and thermochemistry calculations. The electronic structure questions cover topics like electron configuration, quantum numbers, and isoelectronic species.
Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress

This document provides thermodynamic quantities such as enthalpy (AH), Gibbs free energy (AG), and entropy (S°) for various substances at 298.15 K (25°C). It includes data for elements like aluminum, carbon, sulfur, hydrogen, oxygen, bromine, calcium, chlorine, cobalt, copper, chromium, iron, lithium, and sodium, as well as compounds including carbonates, chlorides, oxides, sulfates, and more. The values are given in units of kJ/mol, kJ/mol, and J/mol-K respectively for each substance.
สำเร๊จ

1. The document describes various chemical reactions involved in the production of ammonia, nitric acid, phosphoric acid, and other fertilizers. It outlines reaction equations and conditions.
2. Details are provided on production processes including the contact process for sulfuric acid and ammonia, as well as methods for making superphosphate from phosphate rock using sulfuric acid or phosphoric acid.
3. The document also covers reactions of hydrofluoric acid and how it is used to make hydrogen fluoride, as well as reactions of potassium compounds like potassium chloride and potassium sulfate. Blending and granulation steps in fertilizer production are briefly outlined.
อุตสาหกรรมปุ๋ย

1. The document describes several chemical reactions involved in the production of ammonia, including the Haber process and contact process.
2. It also discusses reactions for producing phosphoric acid and other phosphate compounds, including ones involving calcium fluoride and phosphorus pentoxide.
3. The production of potassium chloride and other potash fertilizers is also summarized, involving reactions between potassium chloride and sodium compounds like sodium chloride and sodium nitrate.
01

1. The document discusses various chemical reactions and processes involved in the production of fertilizers and other chemicals.
2. Key reactions include the contact process for producing ammonia and the Haber process for producing nitric acid. Sulfuric acid is also discussed as a key input.
3. Methods of blending fertilizers are described including bulk blending, granulation, and using various additives to improve properties of the fertilizer.
เรื่อง แอลกอฮอล์

This document discusses alcohols and their properties. It defines primary, secondary, and tertiary alcohols. It also discusses mono-, di-, and trihydric alcohols. Finally, it covers the common and IUPAC naming conventions for alcohols.
Elektrolit dan Nonelek

This document provides an overview of chapter 4 from a chemistry textbook, which covers chemical reactions. It begins by defining electrolytes, non-electrolytes, and discussing the properties of aqueous solutions. It then covers the three main types of reactions that occur in aqueous solutions: precipitation reactions, acid-base reactions involving proton transfer, and redox reactions involving electron transfer. Specific examples of each reaction type are provided. Key concepts around oxidation, reduction, and oxidation numbers are also explained.
Manufacture of Sodium Hydroxide

The major method for producing sodium hydroxide is through the electrolysis of brine, which produces chloride gas, hydrogen gas, and sodium hydroxide. There are three main processes for this: the mercury cell process, the diaphragm cell process, and the membrane cell process. The membrane cell process is now most commonly used as it has the lowest energy requirement and produces very pure sodium hydroxide with no hazardous waste.
Similar to Heat of formation by substances (13)
Calculations using standard enthalpies of formation

Calculations using standard enthalpies of formation
Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress

Pdf tabla-de-entalpias-energia-libre-de-gibbs-y-entropia compress
More from Gary Abud Jr
Gary Abud CV 2017

Gary G. Abud Jr. is an educator who has experience as a principal, teacher, instructional coach, and educational consultant. His background includes over 15 years working in K-12 education, with experiences in both teaching and administration. He has a PhD in curriculum and instruction and provides professional development and consulting services to schools.
Abud ASN 2008-2

This study aimed to identify predictors of rapid progression of chronic kidney disease (CKD) using data from a large CKD clinic. The study found that lower initial systolic blood pressure, advanced age at the initial clinic visit, and longer duration of treatment at the clinic were associated with slower CKD progression. However, the study results contradicted previous findings that factors like proteinuria, ACE inhibitor use, diabetes, and initial anemia predict progression. The authors believe this may be due to survivor bias, as patients who died or started dialysis were not captured, and plan to re-examine the data to avoid this error.
American Society of Nephrology Presentation 2006

CKD clinics provide comprehensive care for patients with renal disease. A study of a CKD clinic found that over time in the clinic:
1) Patients experienced improvements in blood pressure, anemia, and lipid profiles. Anemia decreased from 34% to 21% and use of ESAs increased. Statin use and LDL levels improved.
2) Hyperparathyroidism was not improved, though treatment intensified.
3) Renal function decline slowed compared to historical controls, with the average GFR decreasing only 0.19 mL/min/year.
3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]![3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is a half-day workshop on implementing student-centered practices in the secondary science classroom. The topics include inquiry-based instructional design, visible thinking routines, and talk moves for productive classroom discussion.
14 Reasons to #LoveTeaching

During the week leading up to Valentine’s Day, educators around the country are sharing their love of teaching with a blog post, tweet, picture or video and uniting with the hashtag #LoveTeaching. For more information, visit: http://j.mp/loveteaching
The 2015 Nspire Talks

30 elementary, middle, and high school students came together to animate their ideas in front of a live audience in a series of short and inspiring talks. The concept of the Nspire Talks is a charity event to give kids the stage and microphone to add their voice to the collective conversation and inspire others. Speeches are no more than five minutes and cover a wide range of topics. All talks answered the question: "What's your vision for ...?"
This year's event benefitted the Michigan Eye Bank and raised awareness of the impact of the Organ Donor Registry.
An Educational Voyage Around Michigan

Gary G. Abud, Jr. was named the 2014 Michigan Teacher of the Year. He teaches science in the Grosse Pointe Public Schools and advocates for education in Michigan under the Twitter handle @mr_abud. He also runs a website that provides STEM training for Michigan teachers through an organization called mimodelinginstruction.org.
NGSS Chemistry Webinar

An introduction and overview of the NGSS, with a particular emphasis on the science and engineering practices in chemistry.
Modeling Instruction in High School Chemistry

An overview of the Modeling Method in Chemistry. This presentation was created by Larry Dukerich and Brenda Royce
Engineering Your Classroom Brand in 8 Steps

The document discusses how to engineer a "classroom brand" in eight steps: 1) Write an experience sentence to focus the brand; 2) Design a logo; 3) Create a hashtag; 4) Establish a mantra; 5) Develop student norms and values; 6) Create paraphernalia like shirts; 7) Make the brand ubiquitous in the classroom; 8) Obtain feedback from students on the brand. It argues that branding is powerful and pervasive in culture and can be applied to classrooms to develop student culture and climate.
Before, Change, After (BCA) Tables for Stoichiometry

The BCA Table method of performing stoichiometry calculations that is a cognitive approach that does not rely on algorithms, but rather it engages proportional reasoning skills.
Keeping Track of Energy in Chemical Reactions

Presentation by Larry Dukerich on how to represent energy transfer during chemical reactions. This presentation is part of the training provided by the Modeling Instruction in Chemistry Program.
Acceleration in 1D Review

This document discusses 1D kinematics and different types of graphs used to represent motion including position vs. time graphs, velocity vs. time graphs, and acceleration vs. time graphs. It provides examples of these graphs and challenges the reader to describe the motion shown in various position vs. time graphs. The document aims to help readers test, review, and reinforce their knowledge of 1D kinematics graphs and motion analysis.
10.1.1.115.7999(1)

This document analyzes the impact of homework on student achievement. It utilizes both parametric and nonparametric techniques on data from the National Educational Longitudinal Study of 1988. The results indicate:
1) Homework has a larger and more significant impact on test scores than other standard school inputs like class size.
2) The effects of homework are not uniform - it is most effective for high- and low-achieving students.
3) Parametric models overstate the impact of school inputs like homework and class size compared to nonparametric estimates.
Grosse Pointe North's Emerging Leaders Program - Overview

The Emerging Leaders Program is designed to develop leadership skills in high school students. It will involve regular meetings, mentorship, and classroom participation. Students will work on skills like communication and then apply them by modeling behaviors for peers. The program will start in the classroom and expand to the whole school and community over time. It aims to support student-centered learning and address needs of the classroom, school, and community through service.
A Standards Based Grading Case Study...Kinda

A basic overview of how one approach to Standards Based Grading (SBG) was implemented in a high school physics class using Microsoft Excel and the Pinnacle web-based gradebook.
This presentation was given as part of a larger staff professional development on SBG.
Multiple Choice Practice Questions

This document contains a 50-question chemistry semester review covering various topics including:
1) Properties of ionic compounds including their high melting points.
2) Bond types and strengths in molecules like NH3.
3) Types of bonds that would form between different sets of elements.
4) Factors that determine states of matter for different compounds at various temperatures including intermolecular forces.
The review covers concepts of bonding, molecular shapes, properties of states of matter, acid/base chemistry and chemical reactions.
Google Docs in the Classroom Overview

Google Docs is a free online word processor, spreadsheet, and presentation tool that allows multiple users to collaborate simultaneously on documents. Students can access and work on documents from any internet-connected computer, and teachers can provide feedback during the writing process. The chat feature allows experts to join lessons. Teachers can use Google Docs to collaborate with colleagues and publish materials online for students and parents.
Keratoconus Through the Eyes of a Patient

What would it be like to have a degenerative disease of the cornea? Ultimately resulting in the need for a corneal transplant, keratoconus (KC) breaks down and thins the corneal tissue until vision is impacted in major ways.
GPN Boat Regatta 2011

Students will compete in a cardboard boat regatta on April 29th at Grosse Pointe North Pool where they will demonstrate their physics knowledge by racing boats they constructed out of cardboard, with timed heats and awards judging to celebrate their accomplishments in applying physics principles to a practical challenge.
More from Gary Abud Jr (20)
3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]![3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
3 Ways to Ramp Up Your Science Instruction! [WORKSHOP]
Before, Change, After (BCA) Tables for Stoichiometry

Before, Change, After (BCA) Tables for Stoichiometry
Grosse Pointe North's Emerging Leaders Program - Overview

Grosse Pointe North's Emerging Leaders Program - Overview
Recently uploaded
BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...

BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...Nguyen Thanh Tu Collection
https://app.box.com/s/qhtvq32h4ybf9t49ku85x0n3xl4jhr15Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...Nguyen Thanh Tu Collection
https://app.box.com/s/tacvl9ekroe9hqupdnjruiypvm9rdaneA Visual Guide to 1 Samuel | A Tale of Two Hearts

These slides walk through the story of 1 Samuel. Samuel is the last judge of Israel. The people reject God and want a king. Saul is anointed as the first king, but he is not a good king. David, the shepherd boy is anointed and Saul is envious of him. David shows honor while Saul continues to self destruct.
Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...

Andreas Schleicher, Director of Education and Skills at the OECD presents at the launch of PISA 2022 Volume III - Creative Minds, Creative Schools on 18 June 2024.
Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF

The chapter Lifelines of National Economy in Class 10 Geography focuses on the various modes of transportation and communication that play a vital role in the economic development of a country. These lifelines are crucial for the movement of goods, services, and people, thereby connecting different regions and promoting economic activities.
Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt

The History of NZ 1870-1900.
Making of a Nation.
From the NZ Wars to Liberals,
Richard Seddon, George Grey,
Social Laboratory, New Zealand,
Confiscations, Kotahitanga, Kingitanga, Parliament, Suffrage, Repudiation, Economic Change, Agriculture, Gold Mining, Timber, Flax, Sheep, Dairying,
How to deliver Powerpoint Presentations.pptx

"How to make and deliver dynamic presentations by making it more interactive to captivate your audience attention"
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
Stack Memory Organization of 8086 Microprocessor

The stack memory organization of 8086 microprocessor.
RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students

Physical pharmaceutics notes for B.pharm students
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

Denis is a dynamic and results-driven Chief Information Officer (CIO) with a distinguished career spanning information systems analysis and technical project management. With a proven track record of spearheading the design and delivery of cutting-edge Information Management solutions, he has consistently elevated business operations, streamlined reporting functions, and maximized process efficiency.
Certified as an ISO/IEC 27001: Information Security Management Systems (ISMS) Lead Implementer, Data Protection Officer, and Cyber Risks Analyst, Denis brings a heightened focus on data security, privacy, and cyber resilience to every endeavor.
His expertise extends across a diverse spectrum of reporting, database, and web development applications, underpinned by an exceptional grasp of data storage and virtualization technologies. His proficiency in application testing, database administration, and data cleansing ensures seamless execution of complex projects.
What sets Denis apart is his comprehensive understanding of Business and Systems Analysis technologies, honed through involvement in all phases of the Software Development Lifecycle (SDLC). From meticulous requirements gathering to precise analysis, innovative design, rigorous development, thorough testing, and successful implementation, he has consistently delivered exceptional results.
Throughout his career, he has taken on multifaceted roles, from leading technical project management teams to owning solutions that drive operational excellence. His conscientious and proactive approach is unwavering, whether he is working independently or collaboratively within a team. His ability to connect with colleagues on a personal level underscores his commitment to fostering a harmonious and productive workplace environment.
Date: May 29, 2024
Tags: Information Security, ISO/IEC 27001, ISO/IEC 42001, Artificial Intelligence, GDPR
-------------------------------------------------------------------------------
Find out more about ISO training and certification services
Training: ISO/IEC 27001 Information Security Management System - EN | PECB
ISO/IEC 42001 Artificial Intelligence Management System - EN | PECB
General Data Protection Regulation (GDPR) - Training Courses - EN | PECB
Webinars: https://pecb.com/webinars
Article: https://pecb.com/article
-------------------------------------------------------------------------------
For more information about PECB:
Website: https://pecb.com/
LinkedIn: https://www.linkedin.com/company/pecb/
Facebook: https://www.facebook.com/PECBInternational/
Slideshare: http://www.slideshare.net/PECBCERTIFICATION
Wound healing PPT

This document provides an overview of wound healing, its functions, stages, mechanisms, factors affecting it, and complications.
A wound is a break in the integrity of the skin or tissues, which may be associated with disruption of the structure and function.
Healing is the body’s response to injury in an attempt to restore normal structure and functions.
Healing can occur in two ways: Regeneration and Repair
There are 4 phases of wound healing: hemostasis, inflammation, proliferation, and remodeling. This document also describes the mechanism of wound healing. Factors that affect healing include infection, uncontrolled diabetes, poor nutrition, age, anemia, the presence of foreign bodies, etc.
Complications of wound healing like infection, hyperpigmentation of scar, contractures, and keloid formation.
Recently uploaded (20)
BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...

BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...
Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...
Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...

Andreas Schleicher presents PISA 2022 Volume III - Creative Thinking - 18 Jun...
Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF

Lifelines of National Economy chapter for Class 10 STUDY MATERIAL PDF
Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt

Level 3 NCEA - NZ: A Nation In the Making 1872 - 1900 SML.ppt
NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx

NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx
spot a liar (Haiqa 146).pptx Technical writhing and presentation skills

spot a liar (Haiqa 146).pptx Technical writhing and presentation skills
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students

RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...
Juneteenth Freedom Day 2024 David Douglas School District

Juneteenth Freedom Day 2024 David Douglas School District
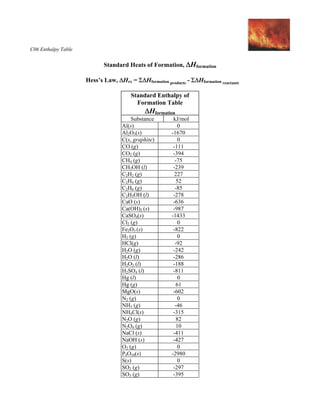
Heat of formation by substances
- 1. C06 Enthalpy Table Standard Heats of Formation, ΔHformation Hess’s Law, ΔHrx = ΣΔHformation products - ΣΔHformation reactants Standard Enthalpy of Formation Table ΔHformation Substance kJ/mol Al(s) 0 Al2O3(s) -1670 C(s, graphite) 0 CO (g) -111 CO2 (g) -394 CH4 (g) -75 CH3OH (l) -239 C2H2 (g) 227 C2H4 (g) 52 C2H6 (g) -85 C2H5OH (l) -278 CaO (s) -636 Ca(OH)2 (s) -987 CaSO4(s) -1433 Cl2 (g) 0 Fe2O3 (s) -822 H2 (g) 0 HCl(g) -92 H2O (g) -242 H2O (l) -286 H2O2 (l) -188 H2SO4 (l) -811 Hg (l) 0 Hg (g) 61 MgO(s) -602 N2 (g) 0 NH3 (g) -46 NH4Cl(s) -315 N2O (g) 82 N2O4 (g) 10 NaCl (s) -411 NaOH (s) -427 O2 (g) 0 P4O10(s) -2980 S(s) 0 SO2 (g) -297 SO3 (g) -395
