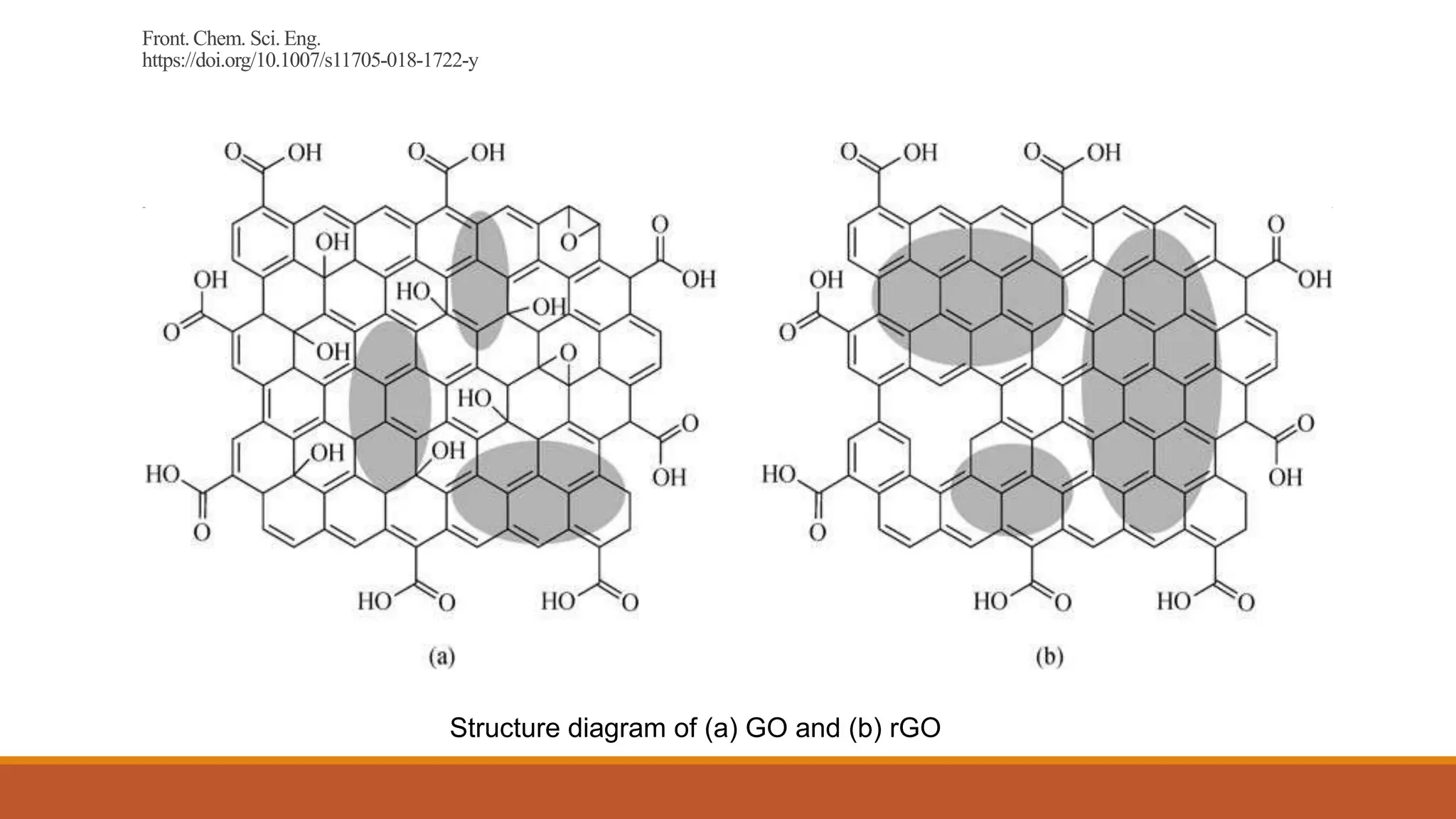
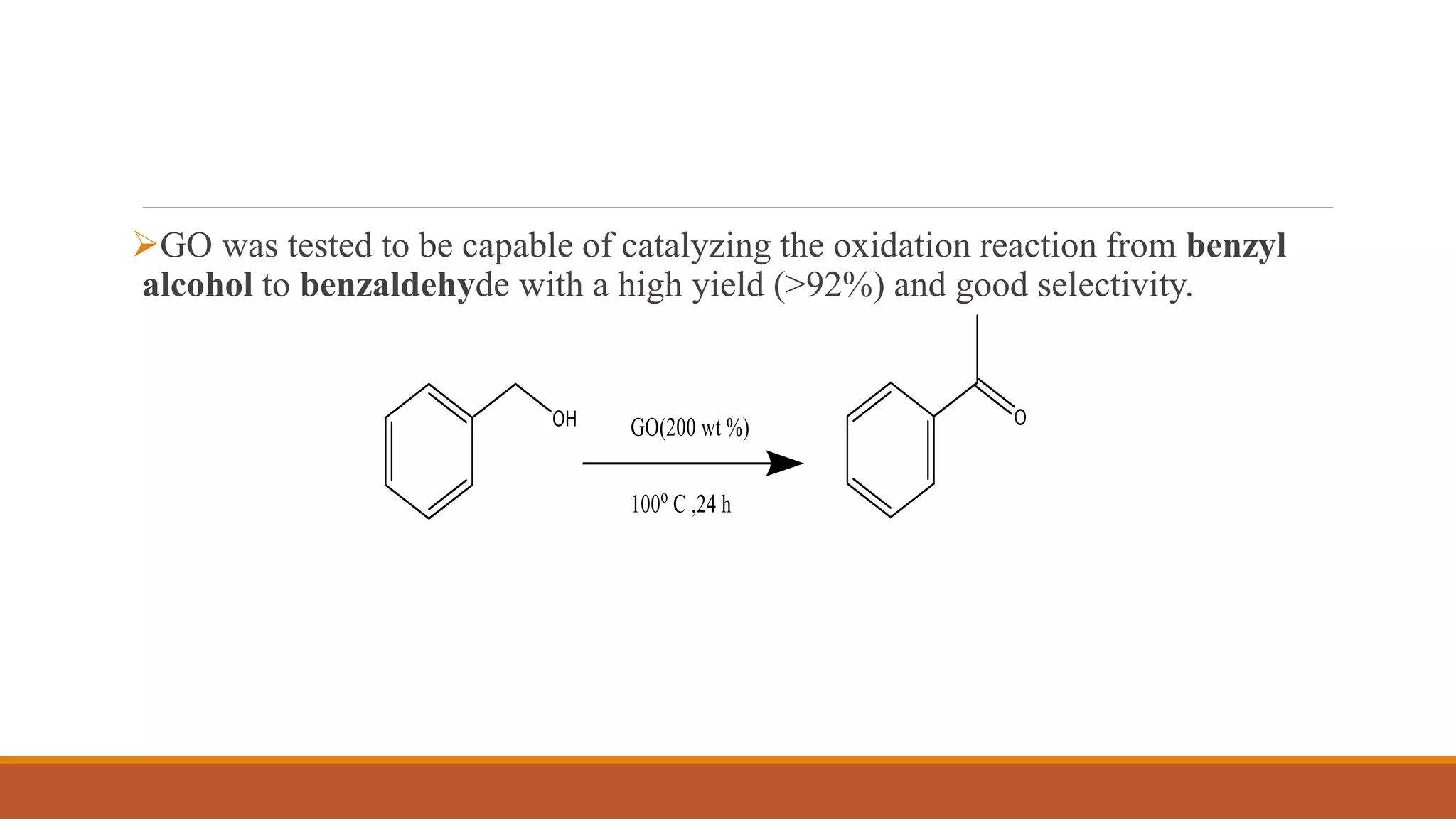
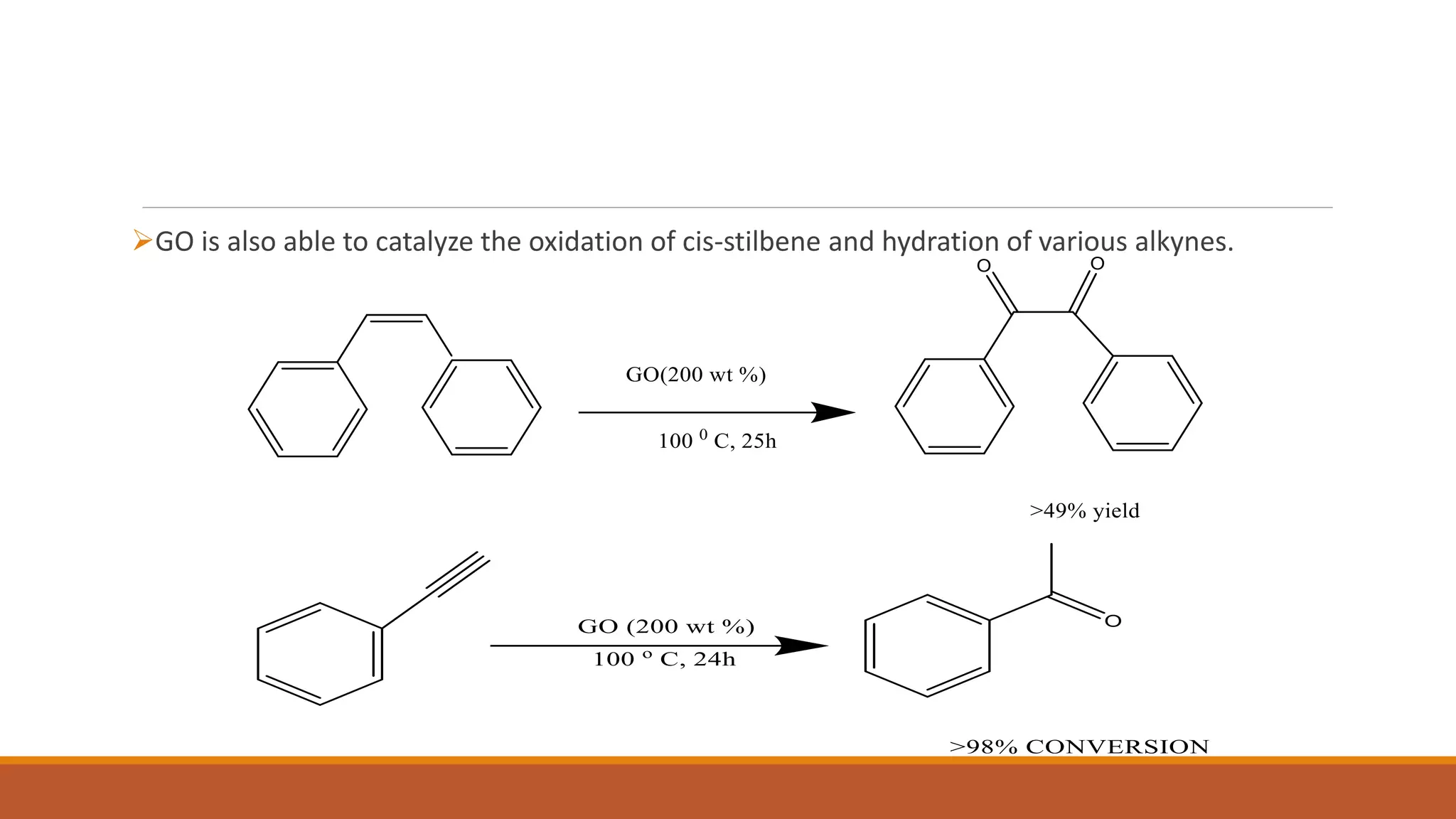
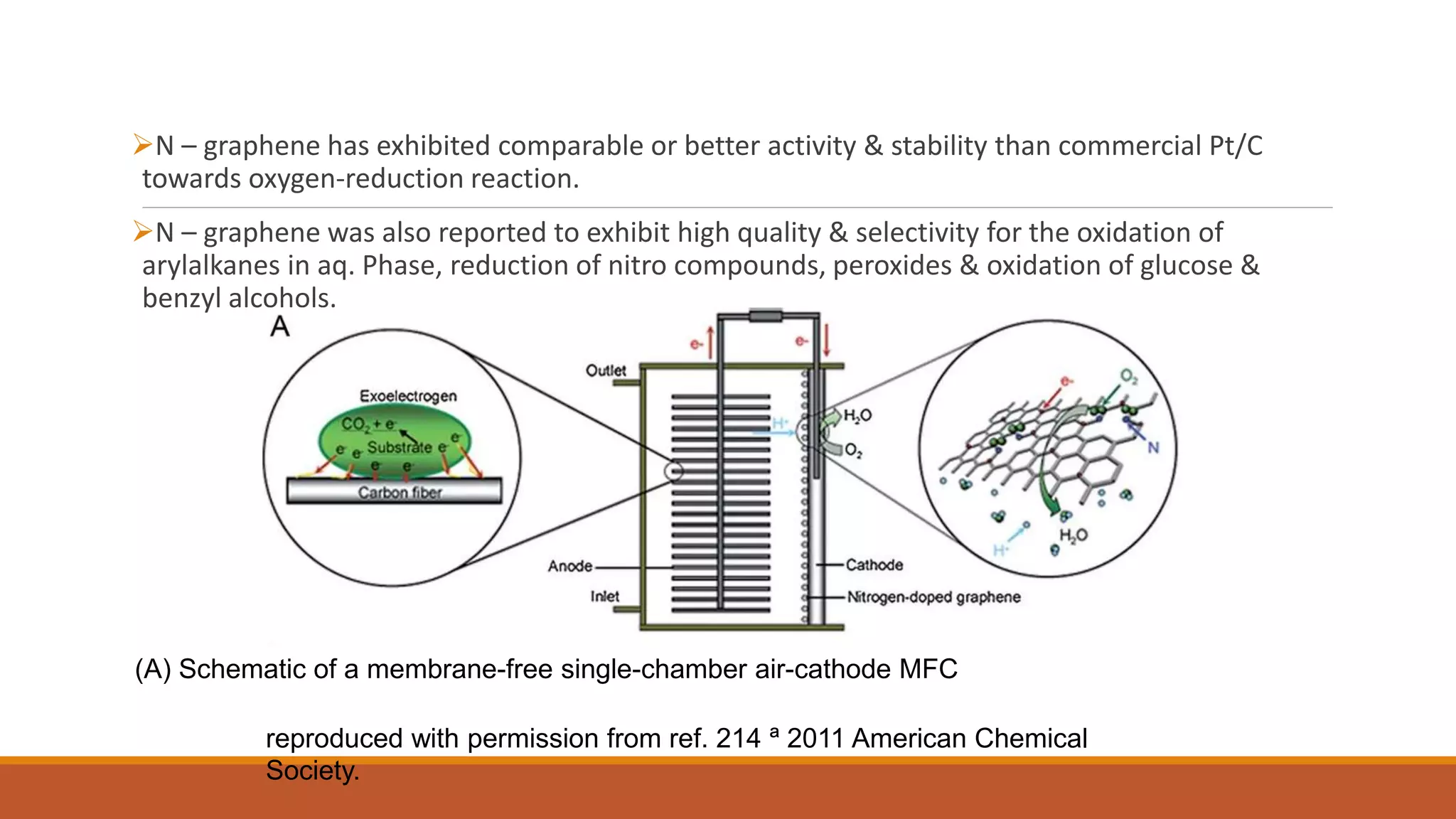
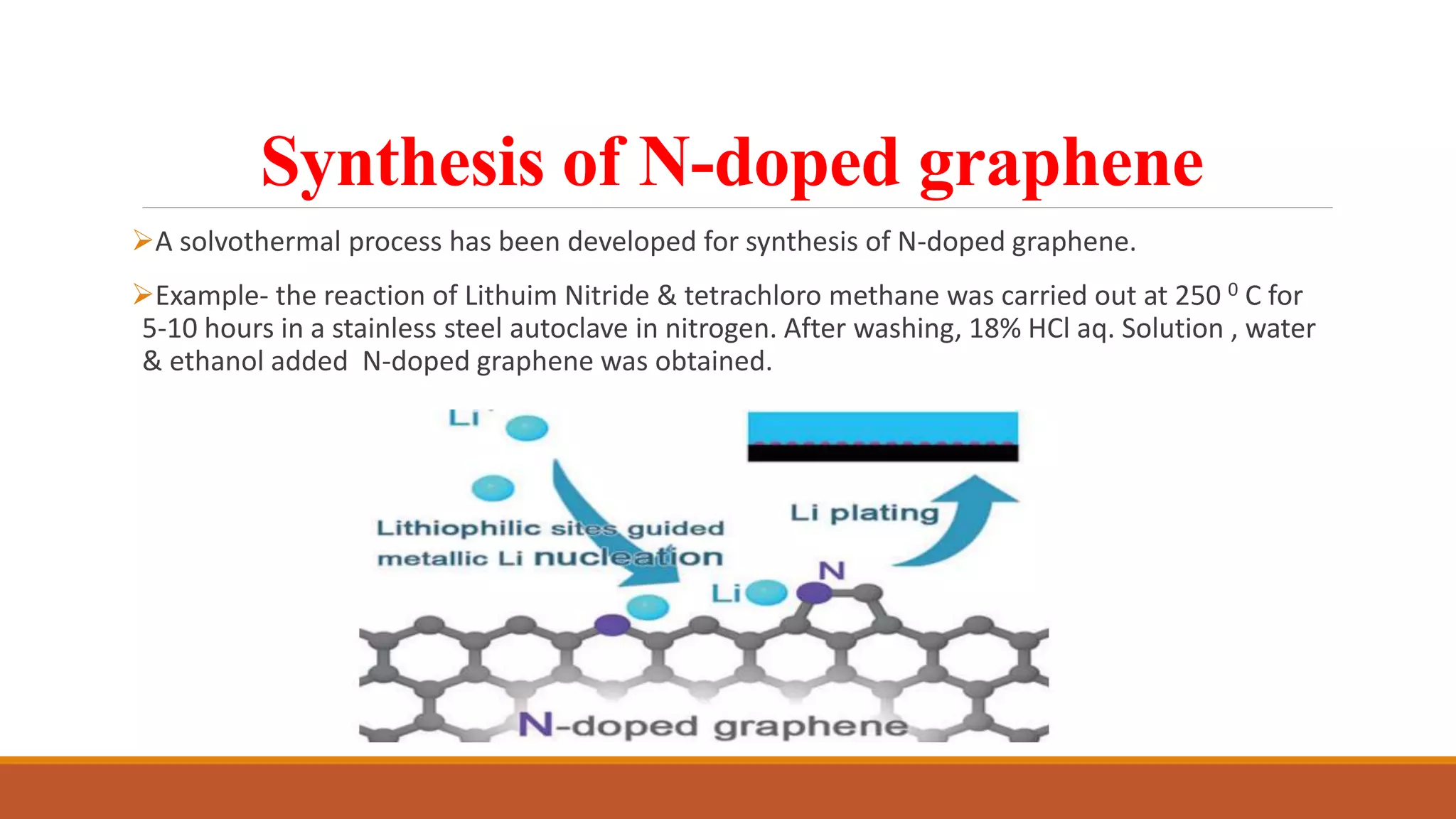
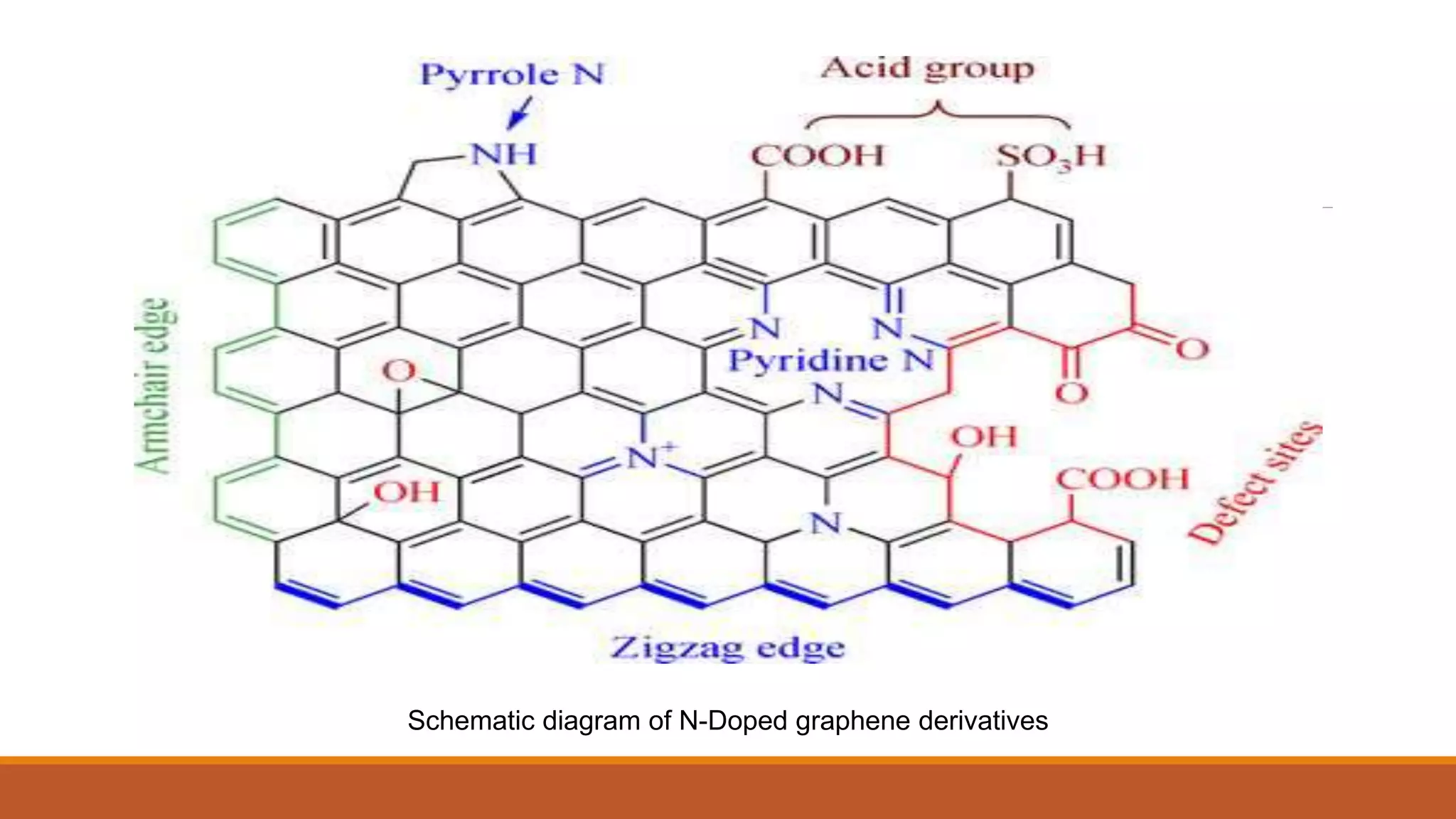
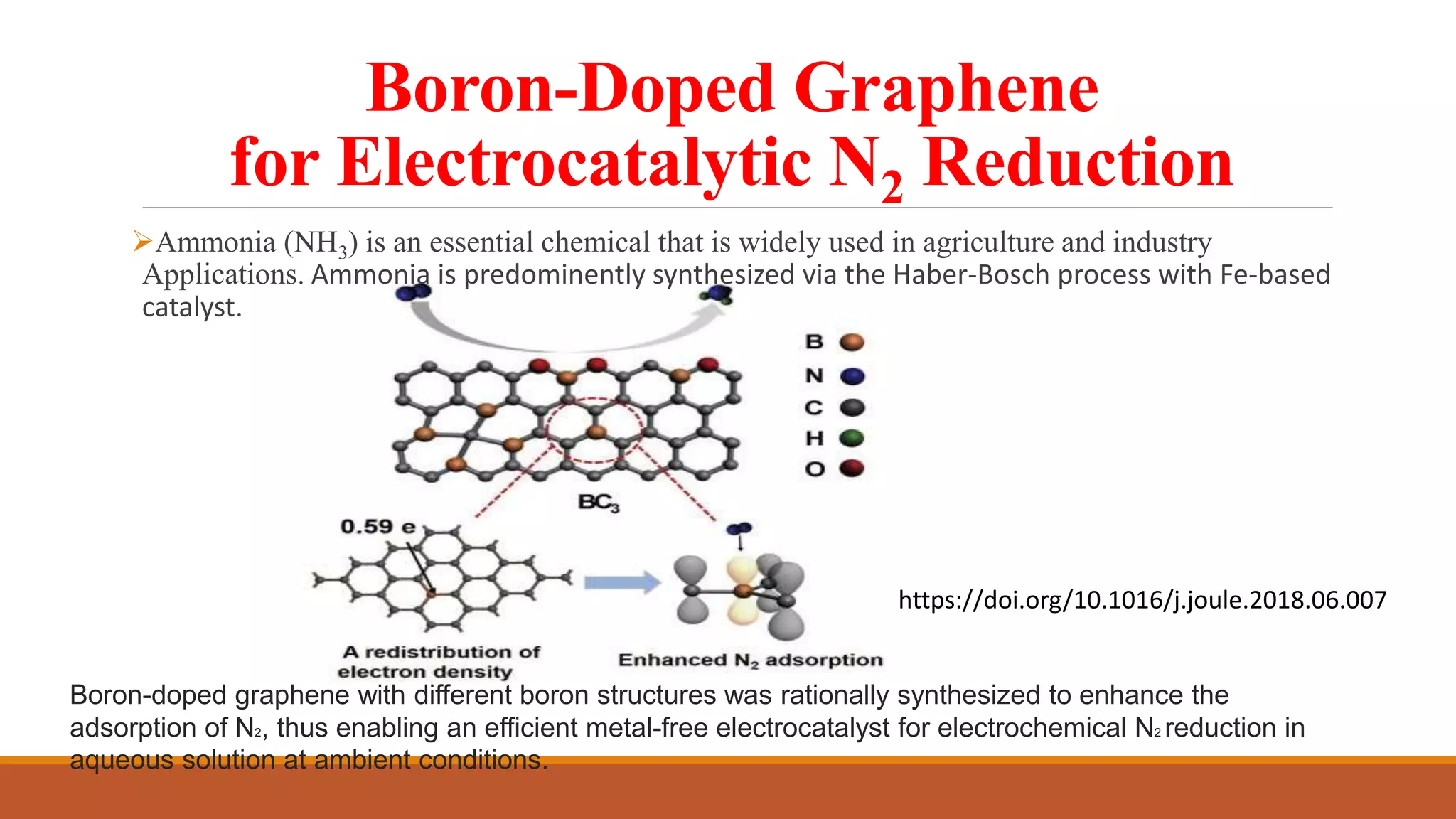
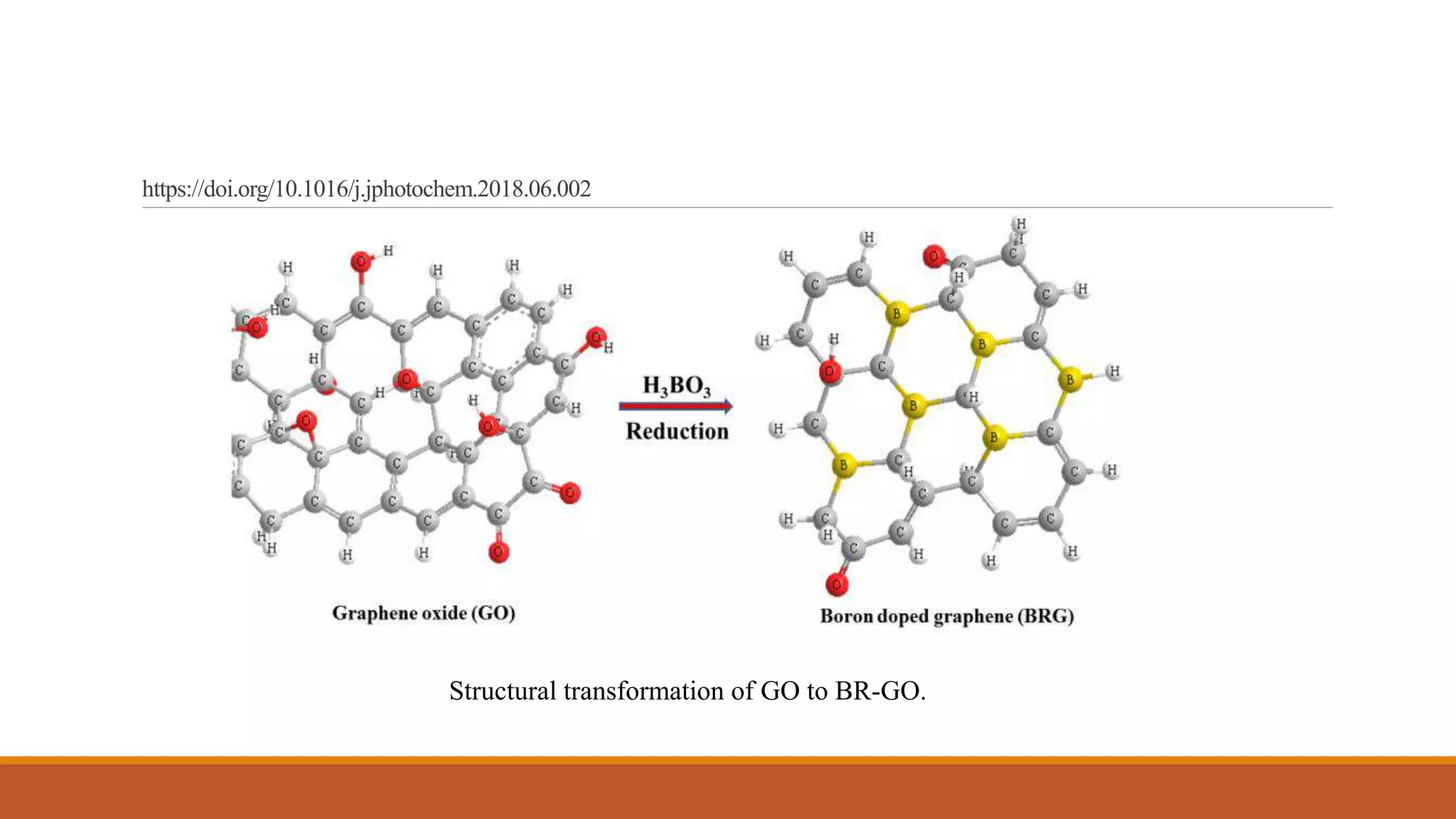
Graphene is a one atom thick layer of carbon atoms arranged in a honeycomb lattice. It has excellent mechanical and electrical properties. The document discusses the use of graphene and chemically modified graphene as catalysts. Graphene can be modified through doping with nitrogen or boron to introduce a band gap and alter its conductivity. These doped graphene materials show potential as metal-free catalysts for organic reactions, fuel cells through oxygen reduction, and nitrogen fixation through electrochemical nitrogen reduction. Doped graphene catalysts offer advantages over traditional metal catalysts including lower cost and stability.