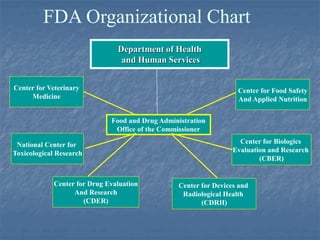
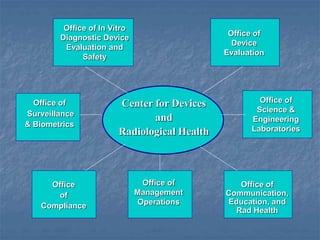
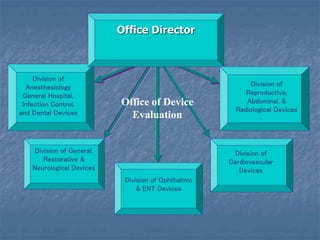
This document discusses the use of outside experts in the FDA's premarket evaluation of medical devices. It describes the FDA's goals in establishing the Medical Device Fellowship Program (MDFP), which aims to increase collaboration with external scientific experts. The MDFP offers short and long-term fellowship opportunities. The document also outlines the process of an action learning project examining how the FDA can most effectively utilize outside resources. It discusses the team's questioning and reflection approach, identification of core issues, and commitment to learning and taking action to address challenges in implementing the MDFP.