EPrints Analytics - Forensic Chemistry OER Course site, Feb/March 2011
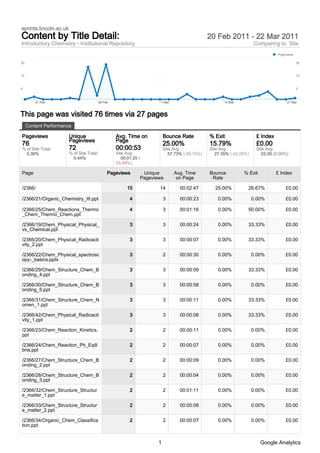
- 1. eprints.lincoln.ac.uk Content by Title Detail: 20 Feb 2011 - 22 Mar 2011 Introductory Chemistry - Institutional Repository Comparing to: Site Pageviews 30 30 15 15 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar This page was visited 76 times via 27 pages Content Performance Pageviews Unique Avg. Time on Bounce Rate % Exit £ Index Pageviews Page 76 25.00% 15.79% £0.00 % of Site Total: 72 00:00:53 Site Avg: Site Avg: Site Avg: 0.36% % of Site Total: Site Avg: 57.73% (-56.70%) 27.35% (-42.28%) £0.00 (0.00%) 0.44% 00:01:20 (- 33.49%) Page Pageviews Unique Avg. Time Bounce % Exit £ Index Pageviews on Page Rate /2366/ 15 14 00:02:47 25.00% 26.67% £0.00 /2366/21/Organic_Chemistry_III.ppt 4 3 00:00:23 0.00% 0.00% £0.00 /2366/25/Chem_Reactions_Thermo 4 3 00:01:16 0.00% 50.00% £0.00 _Chem_Thermo_Chem.ppt /2366/19/Chem_Physical_Physical_ 3 3 00:00:24 0.00% 33.33% £0.00 vs_Chemical.ppt /2366/20/Chem_Physical_Radioacti 3 3 00:00:07 0.00% 33.33% £0.00 vity_2.ppt /2366/22/Chem_Physical_spectrosc 3 2 00:00:30 0.00% 0.00% £0.00 opy-_basics.pptx /2366/29/Chem_Structure_Chem_B 3 3 00:00:09 0.00% 33.33% £0.00 onding_4.ppt /2366/30/Chem_Structure_Chem_B 3 3 00:00:58 0.00% 0.00% £0.00 onding_5.ppt /2366/31/Chem_Structure_Chem_N 3 3 00:00:11 0.00% 33.33% £0.00 omen_1.ppt /2366/42/Chem_Physical_Radioacti 3 3 00:00:08 0.00% 33.33% £0.00 vity_1.ppt /2366/23/Chem_Reaction_Kinetics. 2 2 00:00:11 0.00% 0.00% £0.00 ppt /2366/24/Chem_Reaction_Ph_Eqili 2 2 00:00:07 0.00% 0.00% £0.00 bria.ppt /2366/27/Chem_Structure_Chem_B 2 2 00:00:09 0.00% 0.00% £0.00 onding_2.ppt /2366/28/Chem_Structure_Chem_B 2 2 00:00:04 0.00% 0.00% £0.00 onding_3.ppt /2366/32/Chem_Structure_Structur 2 2 00:01:11 0.00% 0.00% £0.00 e_matter_1.ppt /2366/33/Chem_Structure_Structur 2 2 00:00:08 0.00% 0.00% £0.00 e_matter_2.ppt /2366/34/Organic_Chem_Classifica 2 2 00:00:07 0.00% 0.00% £0.00 tion.ppt 1 Google Analytics
- 2. /2366/35/Organic_Chemistry_Struct 2 2 00:00:07 0.00% 0.00% £0.00 ure.ppt /2366/41/Chemical_Reaction_- 2 2 00:00:09 0.00% 0.00% £0.00 _Redox_Reactions.ppt /2366/43/pH_equilibria_seminar.ppt 2 2 00:00:08 0.00% 0.00% £0.00 /2366/44/Organic_Chemistry_Class 2 2 00:00:08 0.00% 0.00% £0.00 ification_of_Organic_Compounds_ Seminar.ppt /2366/45/Chemical_and_Physical_ 2 2 00:00:05 0.00% 0.00% £0.00 Properties_Practical_Session.ppt /2366/46/Chemical_Structure_Struc 2 2 00:00:09 0.00% 0.00% £0.00 ture_of_Matter_Seminar.ppt /2366/47/structureofthemattersemin 2 2 00:00:17 0.00% 50.00% £0.00 ar-problems.pdf /2366/48/calculationsusingstandard 2 2 00:04:47 0.00% 0.00% £0.00 enthalpiesofformation.pdf /2366/18/Chem_Physical_Isomeris 1 1 00:00:16 0.00% 0.00% £0.00 m.ppt /2366/26/Chem_Structure_Chem_B 1 1 00:00:12 0.00% 0.00% £0.00 onding_1.ppt 1 - 27 of 27 2 Google Analytics