Analytics for http://forensicchemistry.lincoln.ac.uk, Feb/March 2011
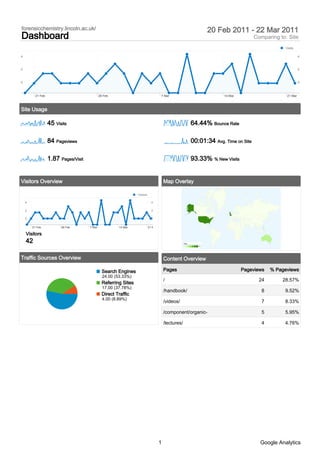
- 1. forensicchemistry.lincoln.ac.uk/ 20 Feb 2011 - 22 Mar 2011 Dashboard Comparing to: Site Visits 4 4 2 2 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar Site Usage 45 Visits 64.44% Bounce Rate 84 Pageviews 00:01:34 Avg. Time on Site 1.87 Pages/Visit 93.33% % New Visits Visitors Overview Map Overlay Visitors 4 4 2 2 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar Visitors 42 Visits 1 14 Traffic Sources Overview Content Overview Search Engines Pages Pageviews % Pageviews 24.00 (53.33%) / 24 28.57% Referring Sites 17.00 (37.78%) /handbook/ 8 9.52% Direct Traffic 4.00 (8.89%) /videos/ 7 8.33% /component/organic- 5 5.95% /lectures/ 4 4.76% 1 Google Analytics
- 2. forensicchemistry.lincoln.ac.uk/ 20 Feb 2011 - 22 Mar 2011 Visitors Overview Comparing to: Site Visitors 4 4 2 2 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar 42 people visited this site 45 Visits 42 Absolute Unique Visitors 84 Pageviews 1.87 Average Pageviews 00:01:34 Time on Site 64.44% Bounce Rate 93.33% New Visits Technical Profile Browser Visits % visits Connection Speed Visits % visits Internet Explorer 16 35.56% Unknown 43 95.56% Firefox 14 31.11% DSL 1 2.22% Chrome 11 24.44% T1 1 2.22% Safari 3 6.67% Mozilla Compatible Agent 1 2.22% 2 Google Analytics
- 3. forensicchemistry.lincoln.ac.uk/ 20 Feb 2011 - 22 Mar 2011 Traffic Sources Overview Comparing to: Site Visits 4 4 2 2 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar All traffic sources sent a total of 45 visits 8.89% Direct Traffic Search Engines 24.00 (53.33%) Referring Sites 37.78% Referring Sites 17.00 (37.78%) Direct Traffic 4.00 (8.89%) 53.33% Search Engines Top Traffic Sources Sources Visits % visits Keywords Visits % visits google (organic) 14 31.11% redox reaction in forensics 2 8.33% chemistryfm.blogs.lincoln.ac.u 9 20.00% biological molecules 1 4.17% yahoo (organic) 7 15.56% chemical and physical 1 4.17% blackboard.lincoln.ac.uk 5 11.11% chemical properties in crime 1 4.17% (direct) ((none)) 4 8.89% classification of matter 1 4.17% 3 Google Analytics
- 4. forensicchemistry.lincoln.ac.uk/ 20 Feb 2011 - 22 Mar 2011 Map Overlay Comparing to: Site Visits 1 14 45 visits came from 11 countries/territories Site Usage Visits Pages/Visit Avg. Time on Site % New Visits Bounce Rate 45 1.87 00:01:34 93.33% 64.44% % of Site Total: Site Avg: Site Avg: Site Avg: Site Avg: 100.00% 1.87 (0.00%) 00:01:34 (0.00%) 93.33% (0.00%) 64.44% (0.00%) Country/Territory Visits Pages/Visit Avg. Time on % New Visits Bounce Rate Site United Kingdom 14 2.14 00:01:44 100.00% 57.14% United States 13 1.23 00:00:21 100.00% 76.92% Australia 4 1.50 00:02:35 50.00% 50.00% Philippines 4 1.50 00:03:50 100.00% 75.00% United Arab Emirates 3 5.67 00:04:07 66.67% 0.00% Chile 2 1.00 00:00:00 100.00% 100.00% Poland 1 1.00 00:00:00 100.00% 100.00% Bangladesh 1 1.00 00:00:00 100.00% 100.00% Canada 1 1.00 00:00:00 100.00% 100.00% Malaysia 1 3.00 00:03:49 100.00% 0.00% 1 - 10 of 11 4 Google Analytics
- 5. forensicchemistry.lincoln.ac.uk/ 20 Feb 2011 - 22 Mar 2011 Content Overview Comparing to: Site Pageviews 20 20 10 10 0 0 21 Feb 28 Feb 7 Mar 14 Mar 21 Mar Pages on this site were viewed a total of 84 times 84 Pageviews 69 Unique Views 64.44% Bounce Rate Top Content Pages Pageviews % Pageviews / 24 28.57% /handbook/ 8 9.52% /videos/ 7 8.33% /component/organic-chemistry/forensic-science-use/ 5 5.95% /lectures/ 4 4.76% 5 Google Analytics