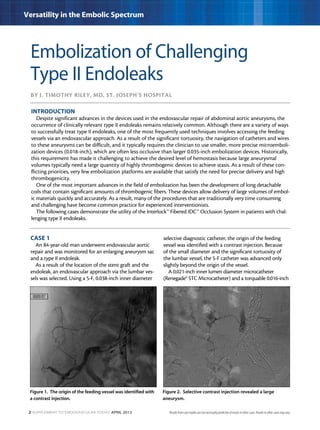
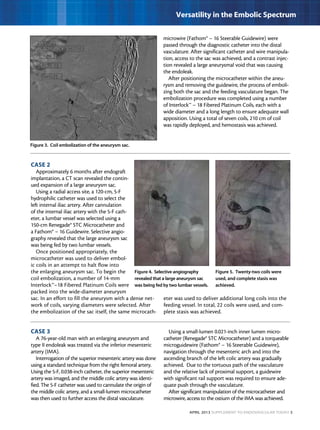
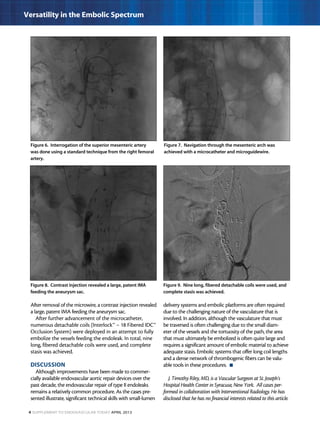
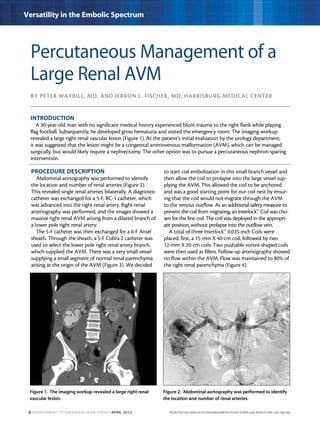
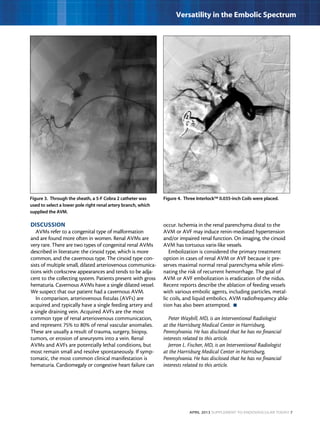
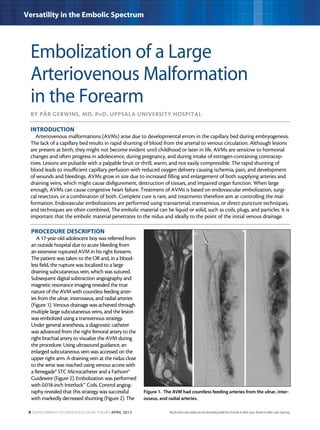
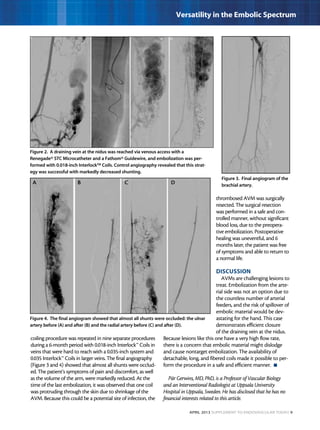
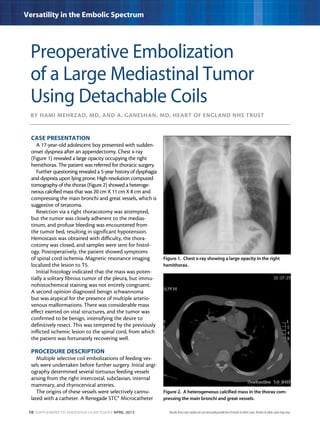
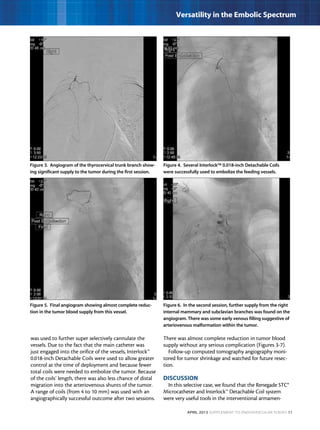
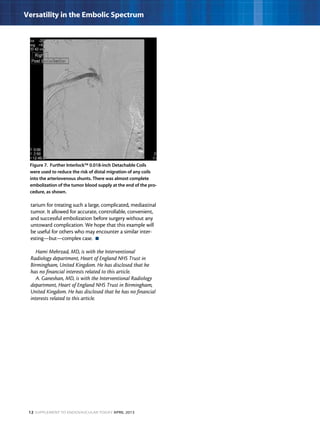
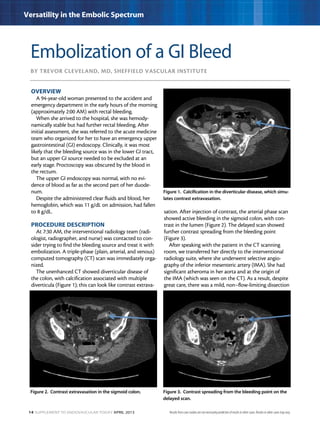
This document describes three case studies of endovascular embolization procedures to treat type II endoleaks. In the first case, a type II endoleak was accessed through tortuous lumbar vessels and embolized using fibered platinum coils. The second case also involved embolizing an aneurysm sac and feeding vessels through difficult lumbar anatomy using microcatheters and coils. In the third case, coils were deployed through the inferior mesenteric artery to treat an endoleak. The document discusses the technical challenges of these procedures and how fibered coil designs can help achieve stasis in large volumes.