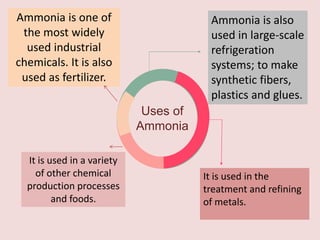
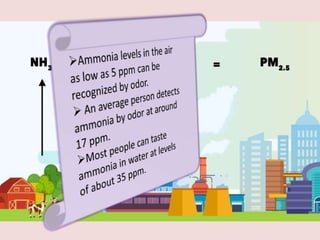
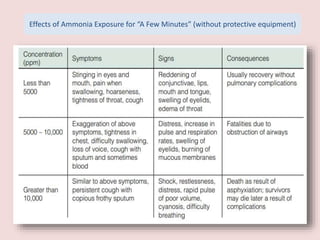
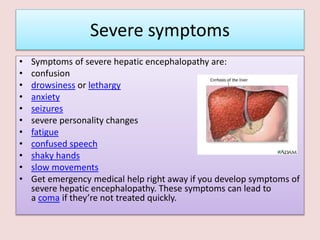
The document discusses the health effects of ammonia exposure, highlighting its industrial uses and the risk of occupational diseases. It details the acute health impacts of ammonia, including irritations and potential severe conditions like hepatic encephalopathy, which can cause significant neurological symptoms. Recommendations for protective measures when working with ammonia are also included, emphasizing the importance of safety equipment and guidelines.