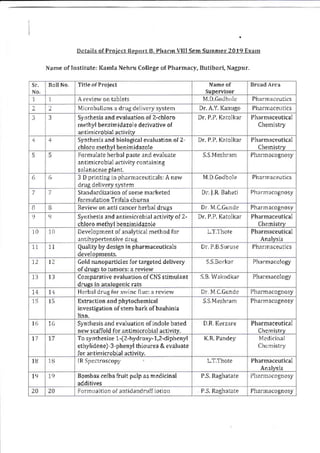
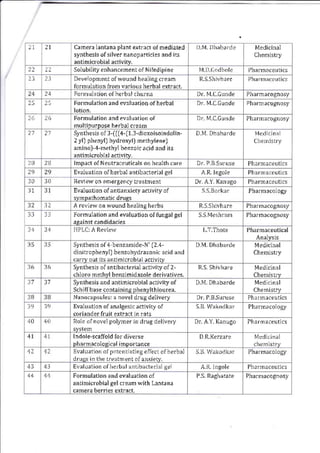
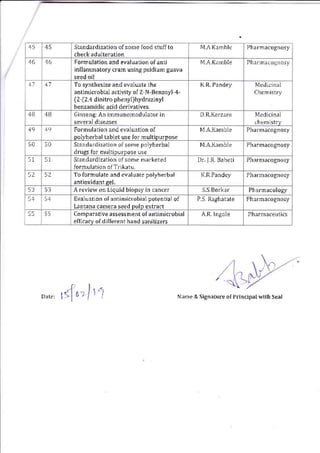
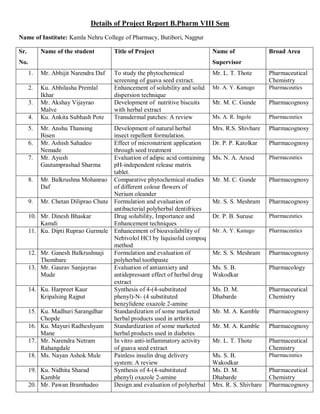
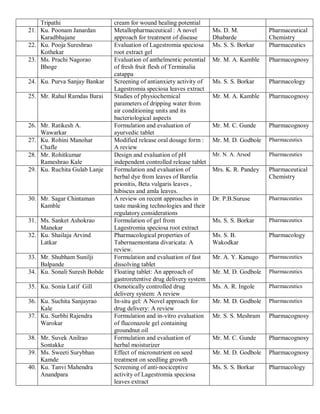
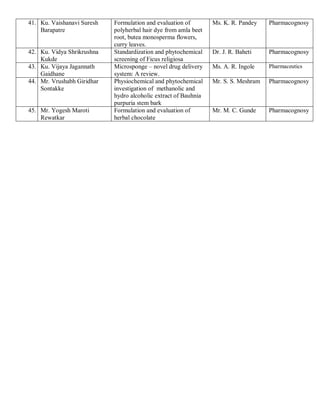
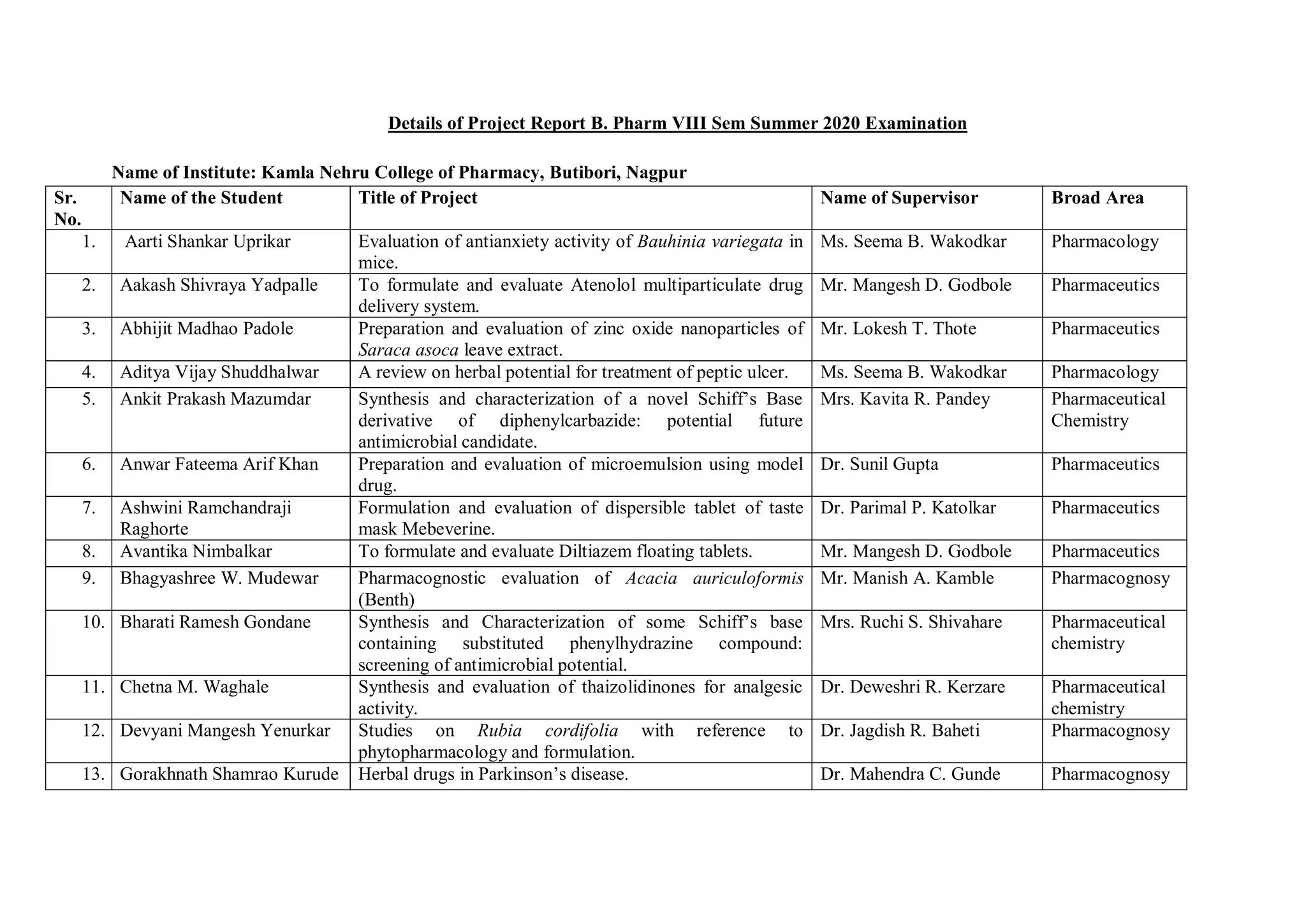
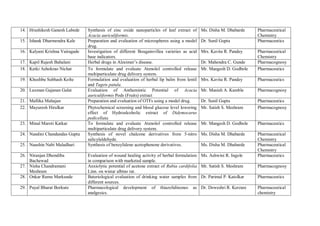
This document provides details of projects being conducted by students of the B. Pharm VIII semester at Kamla Nehru College of Pharmacy in Nagpur, India during the summer of 2020. It lists 66 student projects, each with the student name, project title, and name of the supervising faculty member. The projects cover various areas of pharmacy including pharmaceutics, pharmacognosy, pharmacology, and pharmaceutical chemistry. They involve activities such as synthesis of drug derivatives, herbal drug formulation and evaluation, and determination of bioactivities.
![65. Vaishanavi Umakant
Nimbalkar
In vitro anti-inflammatory evaluation of Acacia
auriculiformis.
Mr. Manish A. Kamble Pharmacognosy
66. Vaishnavi Giridhar Vaidya Design, synthesis and characterization of 2-[(E)-4-[(Z)-
phenylmethylidene]aminophenyl)diazinyl]naphathalen-1-ol
Mrs. Kavita R. Pandey Pharmaceutical
Chemistry
67. Vikas Madhavrao Rathod Formulation and evaluation of herbal tablet for cancer. Dr. Mahendra C. Gunde Pharmaceutics
68. Vishvajit Rajeshwar Shelke Evaluation of polyherbal antidiabetic suspension. Mr. Pradeep S. Raghatate Pharmaceutics
69. Vrushali Subhash Pachpor Artificial intelligence robotics and computational fluid
dynamics.
Dr. Pravinkumar B. Suruse Pharmaceutics
Date: 09-03-20 Name & Signature of Principal with Seal](https://image.slidesharecdn.com/details-of-projects-b-pharm-and-m-pharm-final-year-230516095317-250be41b/85/Details-of-Projects-B-Pharm-and-M-Pharm-Final-Year-pdf-5-320.jpg)