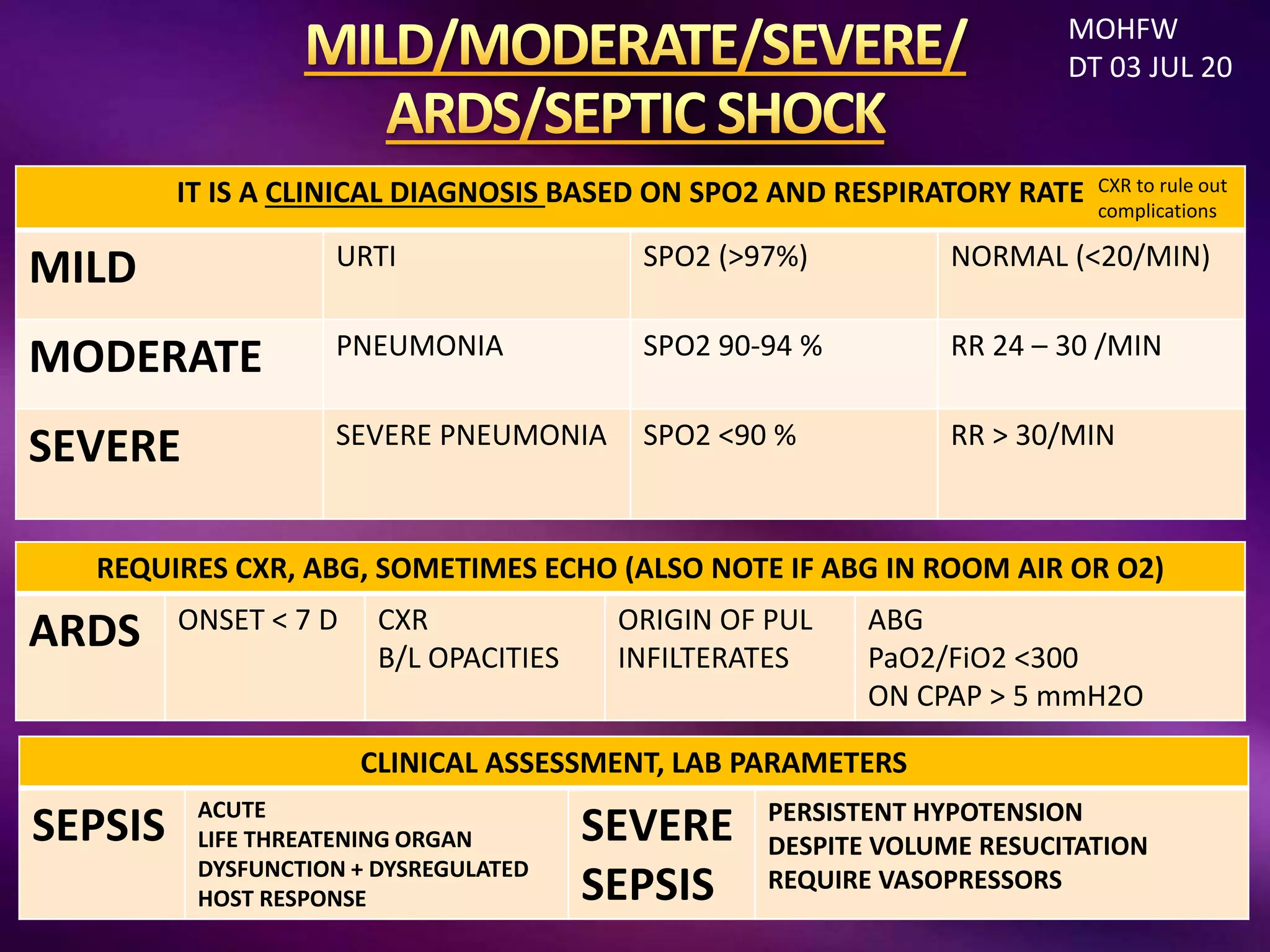
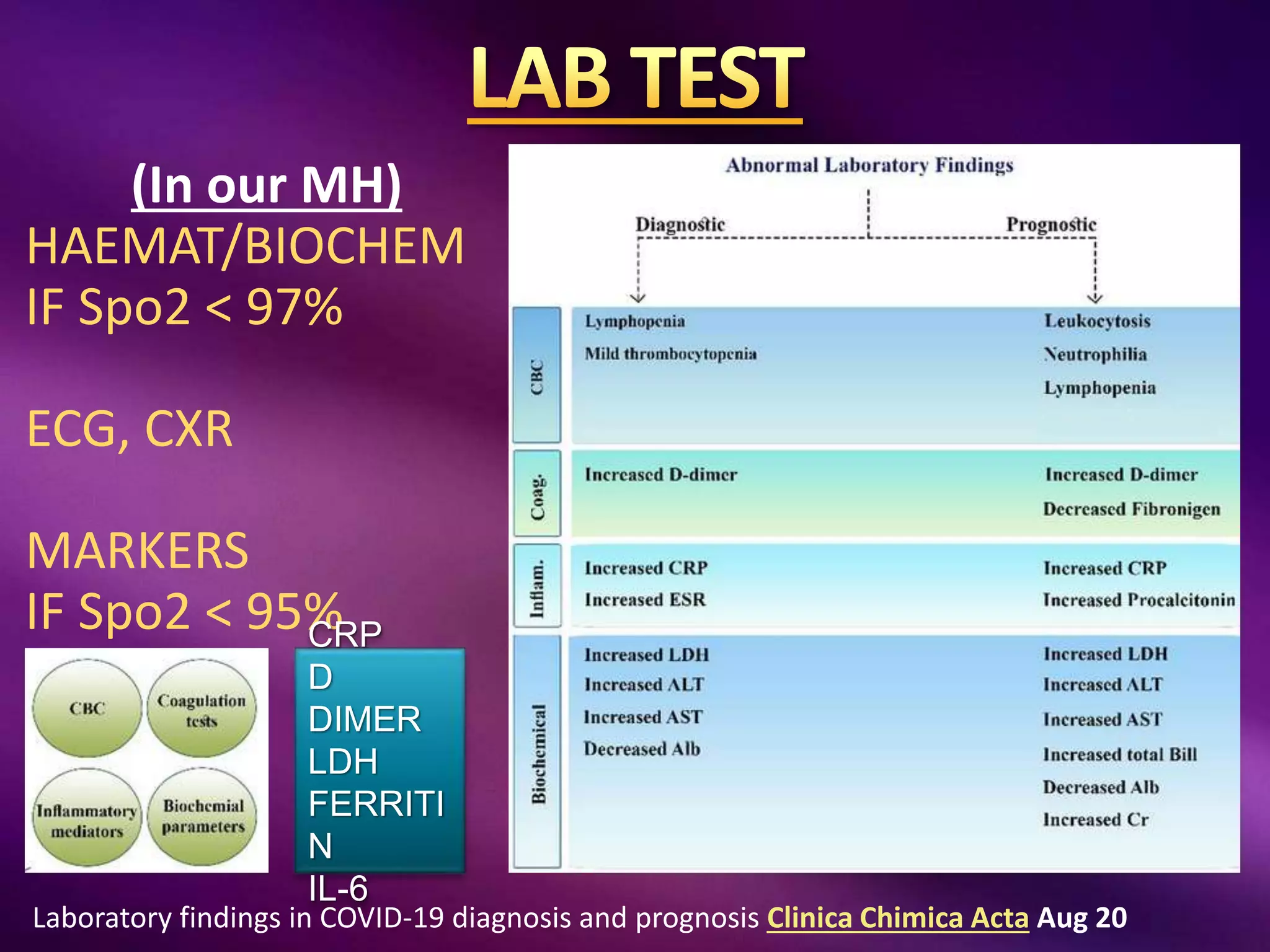
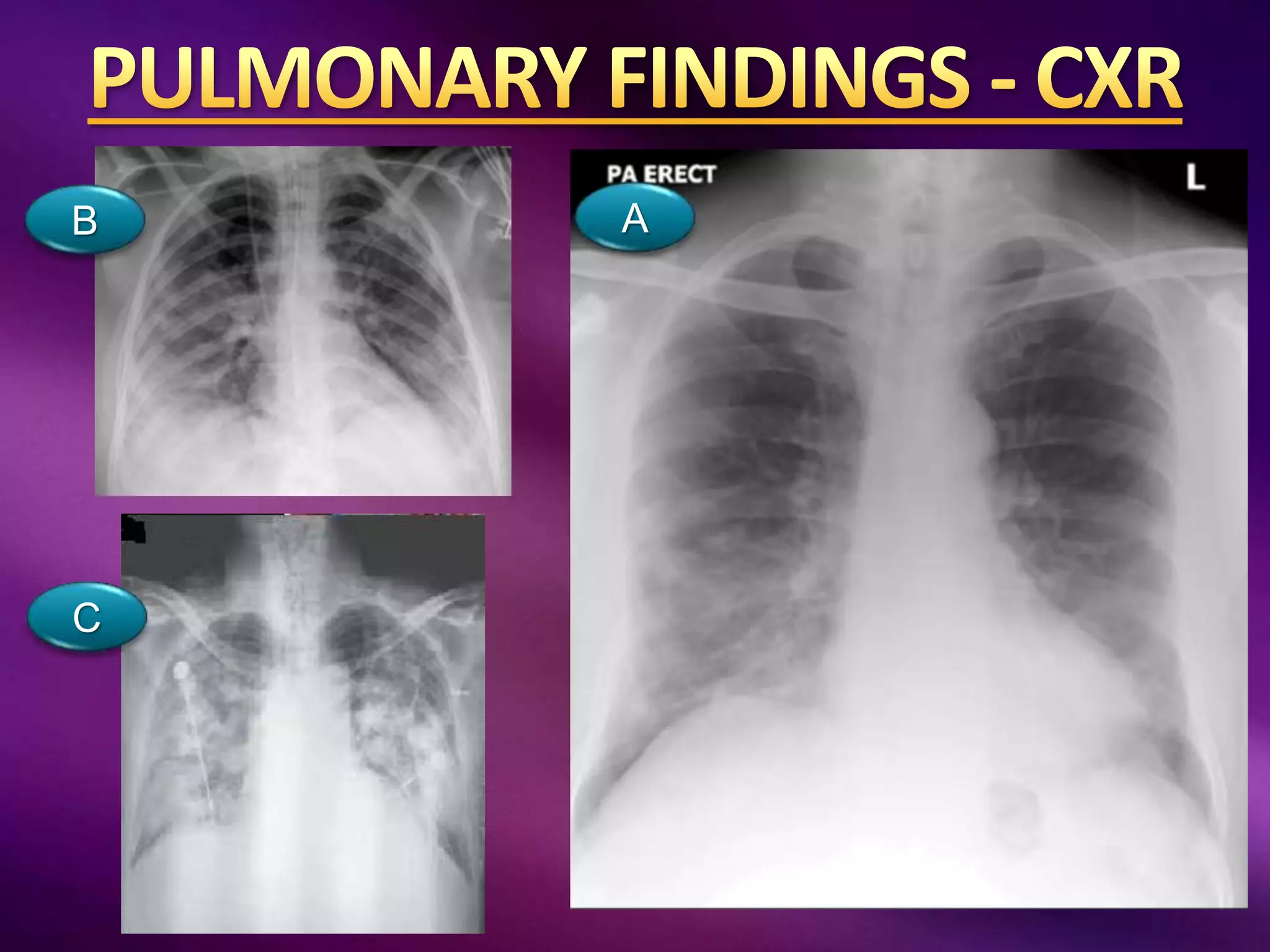
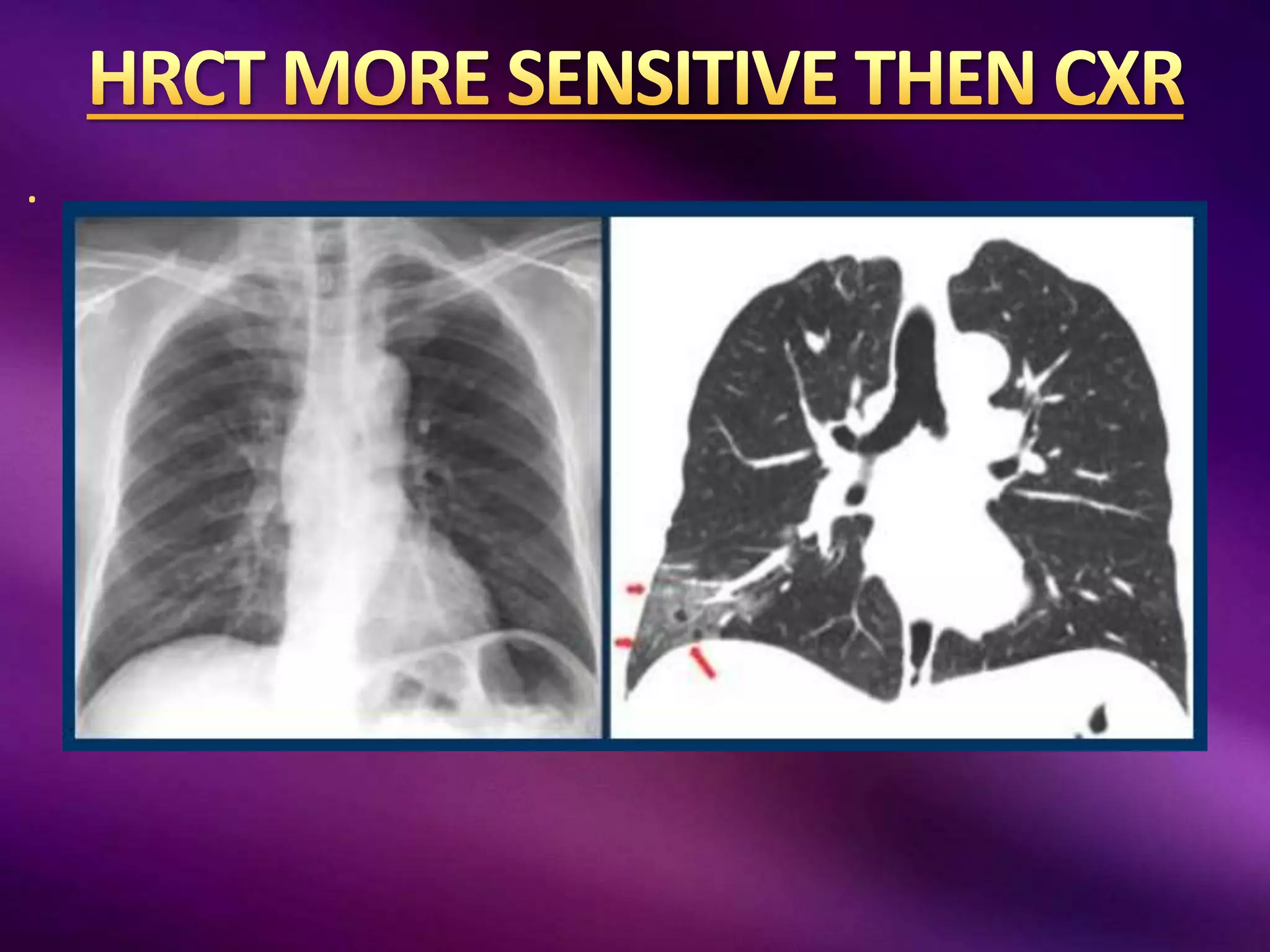
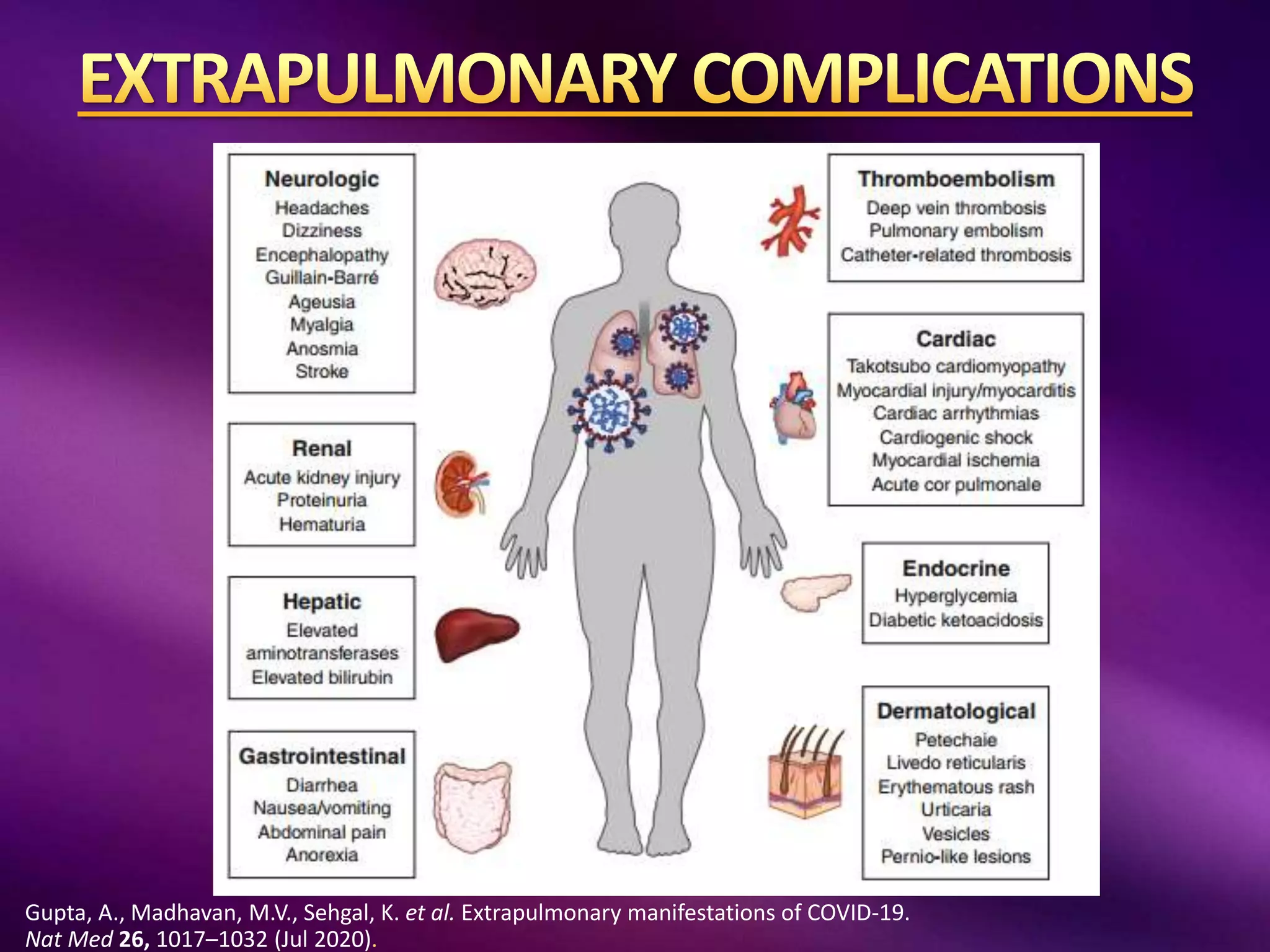
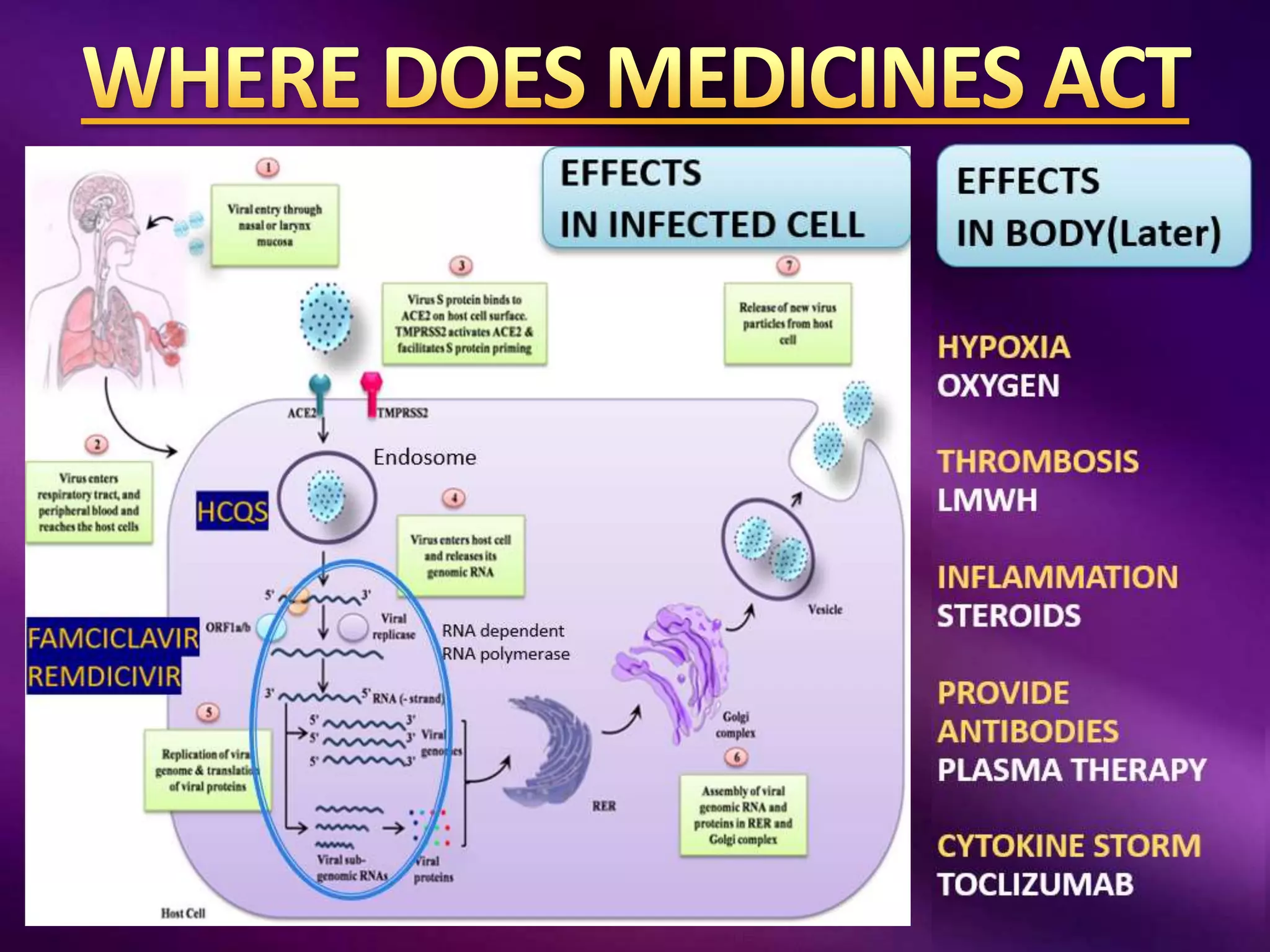
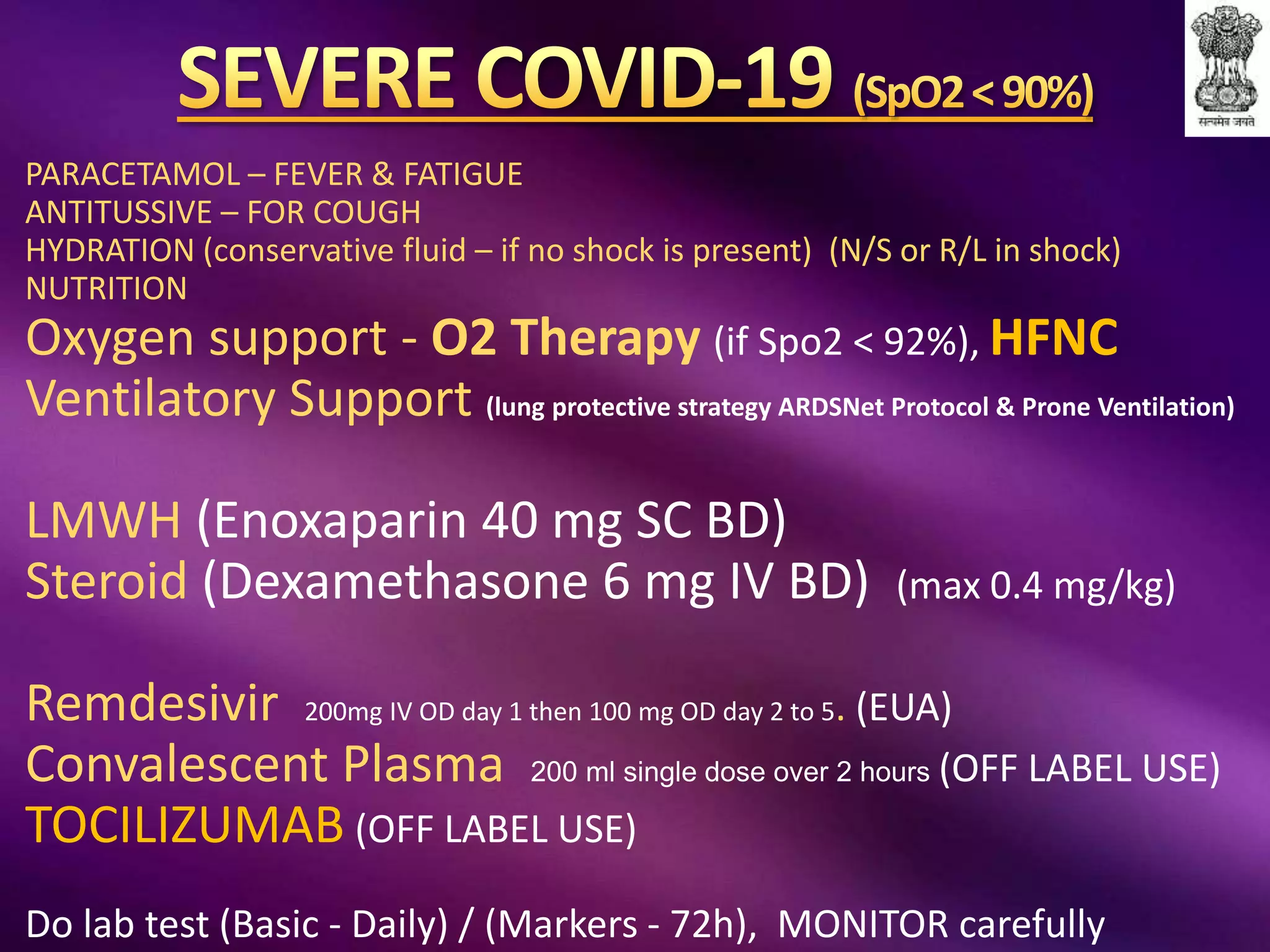
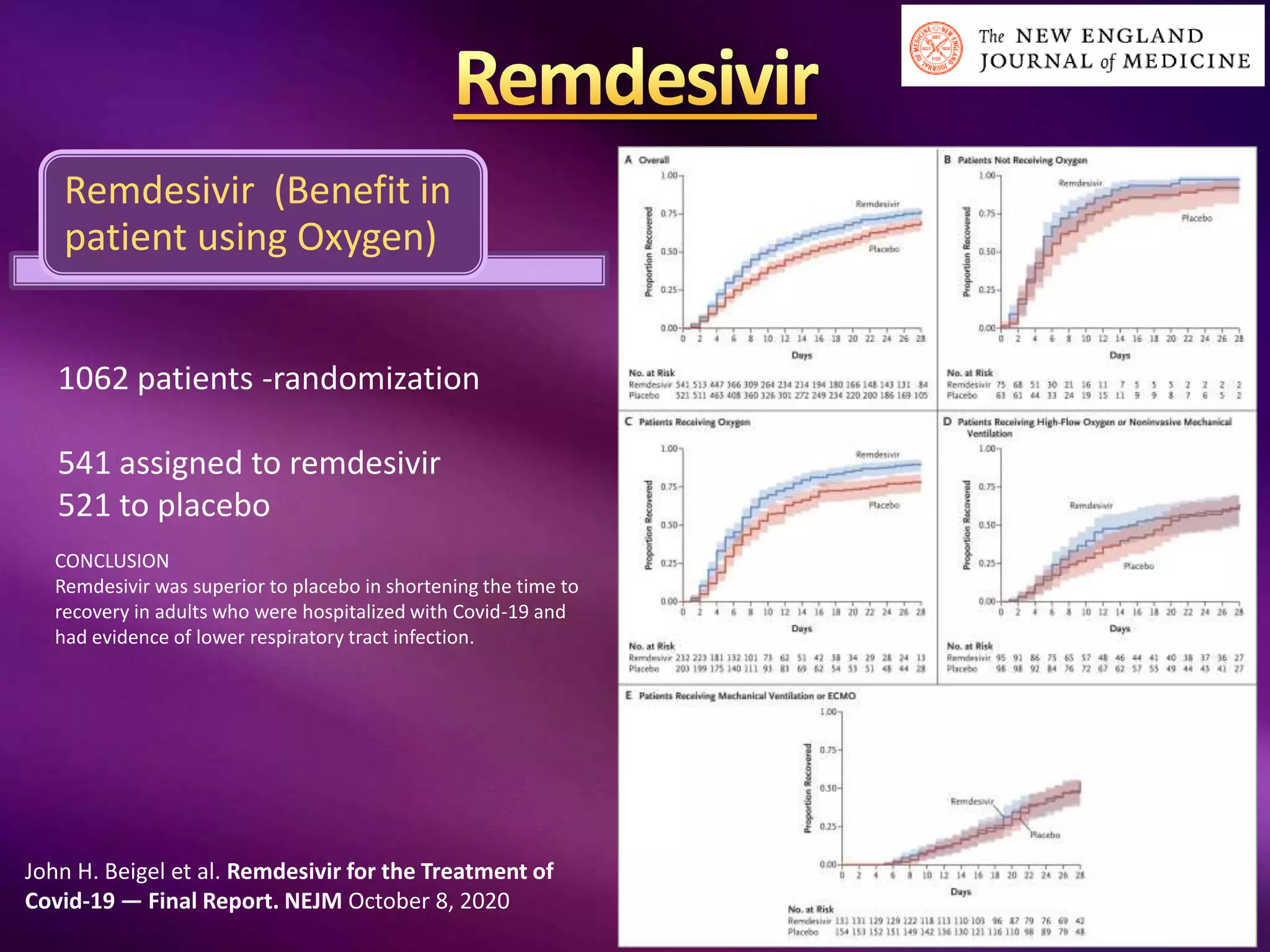
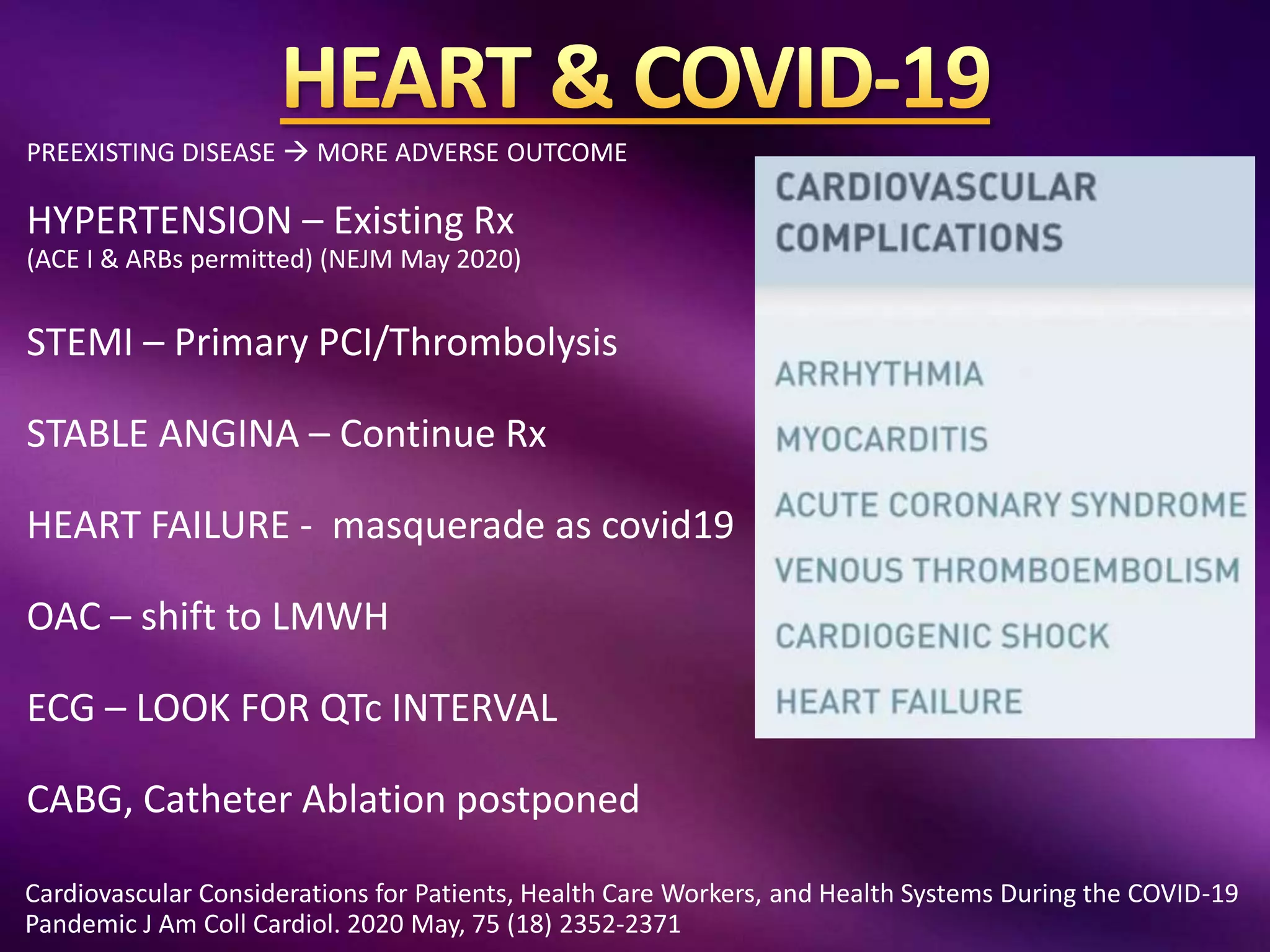
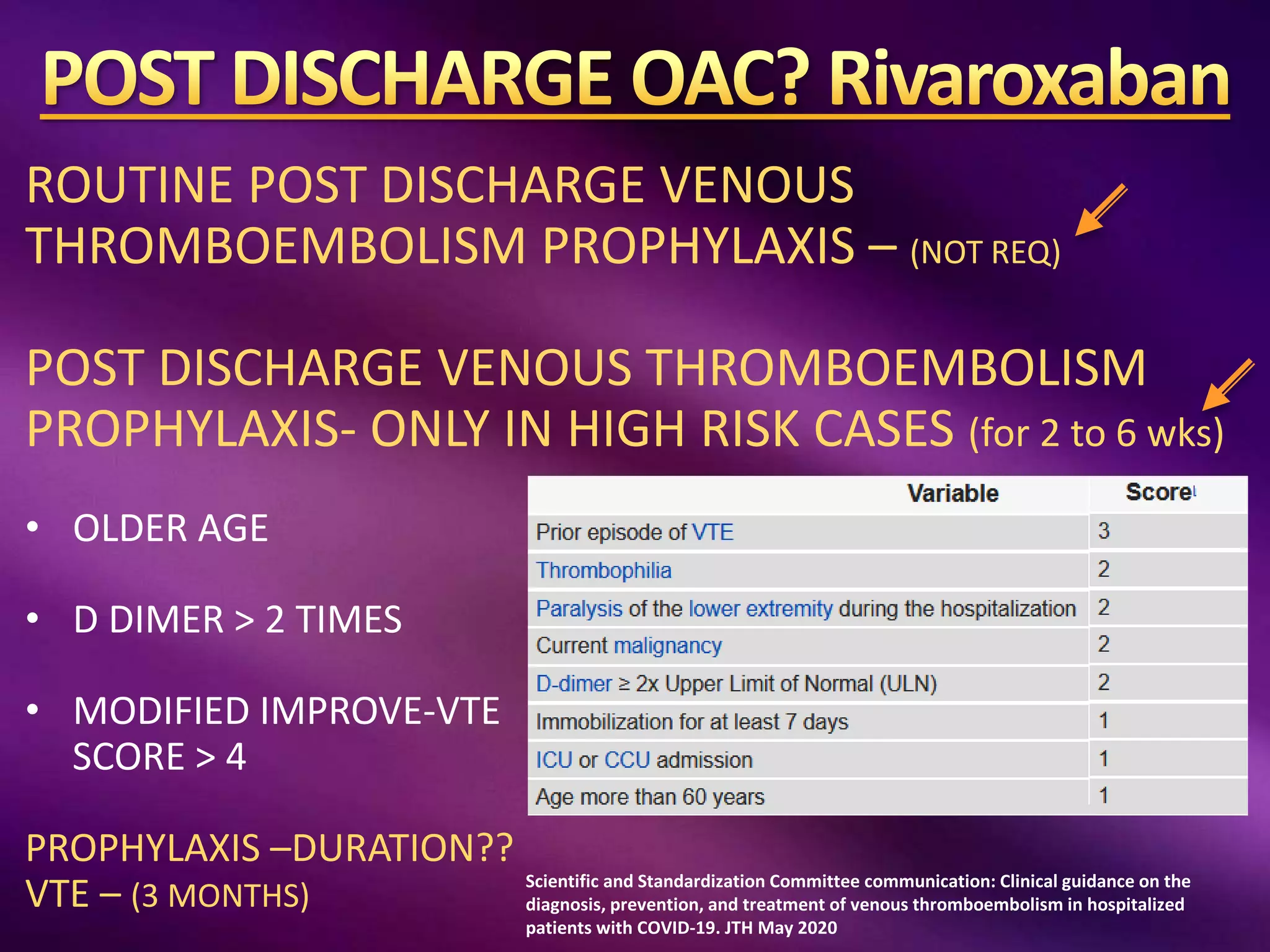
This document outlines guidelines for the diagnosis and management of COVID-19, detailing clinical assessment parameters, treatment options, and recommendations for monitoring. It emphasizes the importance of various factors such as respiratory rate, oxygen saturation, and laboratory findings in evaluating patient risk and treatment response. Additionally, it addresses mental health considerations, potential drug interactions, and best practices for preventing further complications in patients with COVID-19.