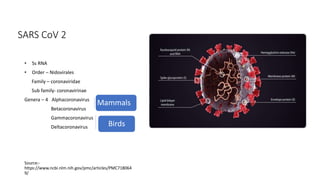
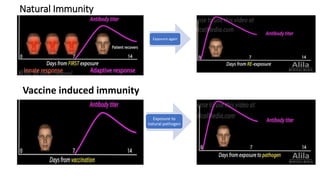
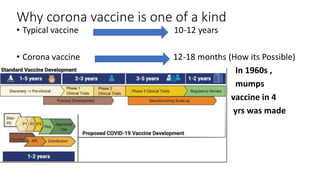
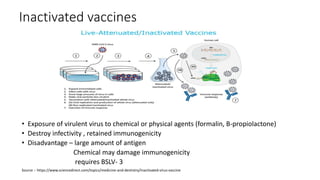
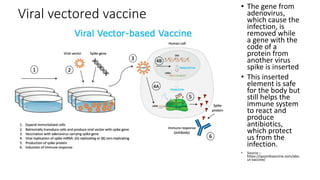
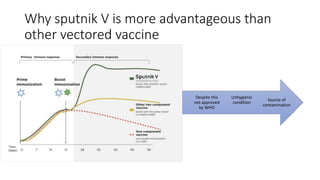
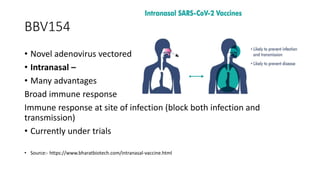
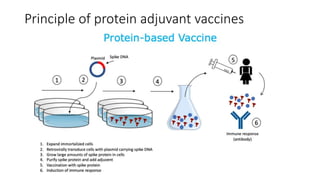
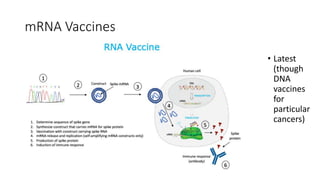
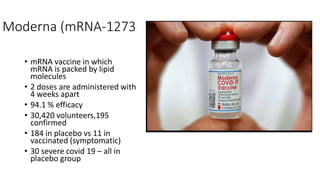
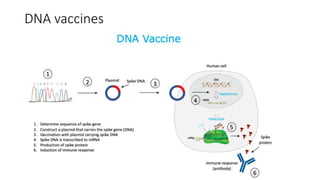
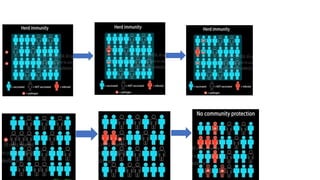
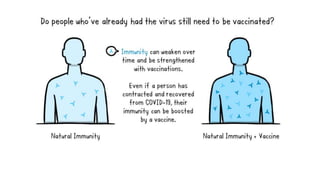
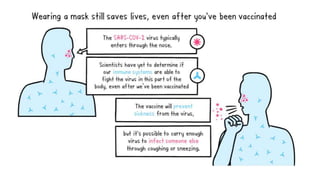
The document summarizes different aspects of COVID-19 vaccines, including how they work, types of vaccines, and specific vaccines like Covaxin, Pfizer, Moderna, and more. It discusses inactivated vaccines like Covaxin and CoronaVac, viral vector vaccines like AstraZeneca and Sputnik V, protein subunit vaccines like Novavax, and nucleic acid vaccines such as Pfizer and Moderna. Each vaccine is described in 1-2 sentences focusing on technology, efficacy, and development process. The document also covers general topics like vaccine development, immunity, and adjuvants that enhance vaccine effectiveness.