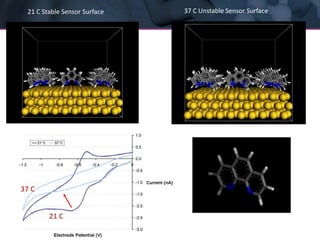
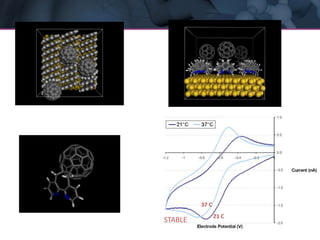
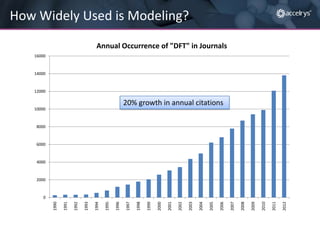
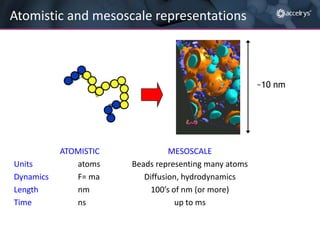
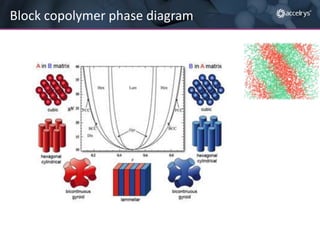
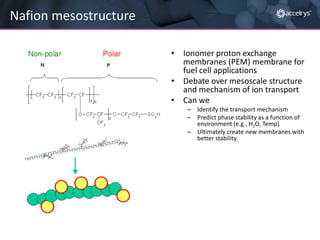
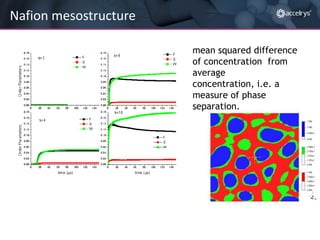
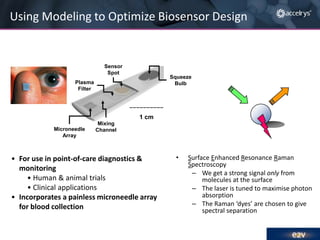
The presentation discusses the role of computational modeling in the chemicals industry, highlighting its growing importance in various sectors such as pharmaceuticals and aerospace. Successful modeling requires knowledge of relevant tools, effective communication with experimentalists, and an understanding of complex systems, like lithium-ion batteries. Examples of corporate challenges and solutions illustrate the practical applications of modeling to accelerate development and improve product performance.


















![Modeling Dye-Surface Interactions
Silver [111]
Surface
GM19 Dye
Oxide Trilayer
H2O “Bridge”
Bulk Silver
Dye-Surface Interaction](https://image.slidesharecdn.com/cornellcompchemexternal-131016113226-phpapp01/85/Cornell-Computational-Chemistry-Seminar-29-320.jpg)