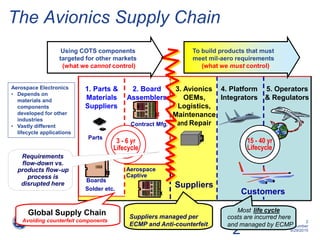
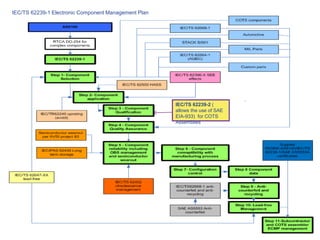
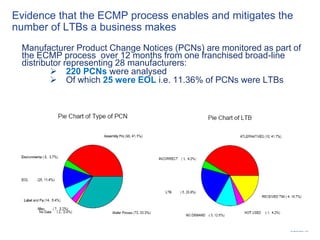
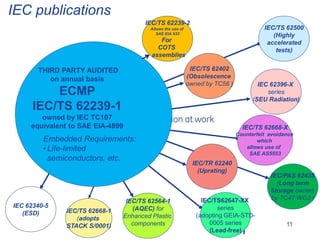
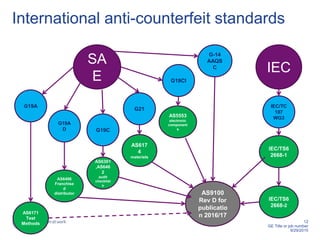
This document discusses electronic component management plans (ECMP) and obsolescence management in the avionics supply chain. It notes that 57% of reported counterfeit parts involve obsolete or end-of-life parts. The document outlines the 11 step ECMP process defined in IEC/TS 62239-1, including component selection, qualification, and obsolescence management. It provides an example where monitoring of manufacturer product change notices through the ECMP process allowed alternatives to be found for 40% of last-time buys. The document also discusses related standards like IEC/TS 62239-2 for COTS assemblies and third-party audit schemes through organizations like IECQ.