chemical Reactions DS
•Download as PPTX, PDF•
0 likes•200 views
Elements are pure chemical substances consisting of one type of atom, while compounds consist of two or more elements chemically bonded together. Atoms are the basic units of matter, containing a nucleus of protons and neutrons surrounded by electrons. Metals readily lose electrons to form positive ions surrounded by delocalized electrons, giving them high conductivity. Non-metals have more varied properties. The periodic table organizes the elements and displays recurring trends in their properties based on atomic structure. Chemical reactions lead to the transformation of substances and can be identified by types such as synthesis, where elements combine to form compounds, or decomposition, where compounds break down.
Report
Share
Report
Share
Recommended
The Elements

This document provides an overview of key chemistry concepts including:
1. Elements are substances that cannot be broken down further through chemical means. There are around 118 known elements which combine to form all matter through atoms.
2. Atoms are composed of protons, neutrons, and electrons. Atoms combine to form elements listed in the Periodic Table. Metals readily lose electrons to form positive ions surrounded by delocalized electrons.
3. Non-metals have differing physical and chemical properties from metals. The Periodic Table displays the elements and illustrates trends in their properties. Compounds consist of a fixed ratio of elements held together by chemical bonds.
Chemical Reactions JQ

The periodic table organizes all known elements and contains information about atoms and molecules. It arranges metals, non-metals, and metalloids using elemental symbols. Elements are composed of atoms with a specific number of protons. Compounds consist of two or more elements in a fixed ratio that are bonded together. Chemical reactions transform one set of substances into another, either spontaneously or through external energy sources, while physical changes only alter the state of matter.
Chemical Reactions TG

Elements are made up of atoms that contain protons, neutrons and electrons. Atoms combine to form compounds made of two or more elements that can be separated through chemical reactions. Chemical reactions involve elements interacting to form new compounds, which can sometimes produce byproducts like gases. A chemical change alters the substances chemically while a physical change only modifies the shape or form. Reaction rates can be affected by factors like temperature, with heat speeding them up and cold slowing them down.
Chemical Reactions EL

Elements are substances made of only one type of atom. Atoms contain protons, neutrons and electrons. Metals are elements that conduct heat and electricity, while non-metals have different properties and are usually gases or brittle solids. The periodic table displays all the known chemical elements and their properties in an organized format. Compounds are formed when two or more elements chemically combine, and chemical reactions involve transformations between substances that cannot be reversed.
Chemical Reactions SB

The document defines key chemistry concepts such as atoms, elements, compounds, and molecules. Atoms are the fundamental units of matter made up of protons, neutrons, and electrons. Elements are pure substances composed of only one type of atom, while compounds contain two or more elements bonded together. The document also discusses chemical and physical changes, factors that affect reaction rates, and provides examples of metals and nonmetals.
chemical Reactions DM

Elements are pure substances made of one type of atom, while atoms are composed of protons, neutrons, and electrons. Metals are elements or alloys that conduct heat and electricity well, whereas nonmetals are usually gases or weak solids that lack metallic properties. A compound is formed when two or more elements chemically bond together in fixed proportions, and a chemical reaction transforms one set of chemicals into another through processes like producing heat, light, or precipitate.
Chemical Reactions CD

The document discusses several key chemistry concepts:
- Atoms are made up of protons, neutrons, and electrons. An element is made of a single type of atom.
- Compounds are made of two or more elements chemically bonded together. Chemical reactions involve breaking and forming bonds to change one substance into another.
- Physical changes alter a substance's properties like shape or state but not its chemical makeup. Chemical reactions alter the substance's chemical identity and atomic/molecular structure.
U2L6 - Interactions of Atoms

This document discusses chemical bonding and interactions between atoms. It explains that during chemical changes, atoms are rearranged and bonds are formed and broken, but atoms are neither created nor destroyed. Chemical equations are used to represent chemical reactions, with reactants on the left and products on the right. Balanced equations show the conservation of mass with equal numbers and kinds of atoms on both sides. Bohr models represent atoms with electrons in energy levels around the nucleus and show how valence electrons participate in bonding to achieve stable configurations.
Recommended
The Elements

This document provides an overview of key chemistry concepts including:
1. Elements are substances that cannot be broken down further through chemical means. There are around 118 known elements which combine to form all matter through atoms.
2. Atoms are composed of protons, neutrons, and electrons. Atoms combine to form elements listed in the Periodic Table. Metals readily lose electrons to form positive ions surrounded by delocalized electrons.
3. Non-metals have differing physical and chemical properties from metals. The Periodic Table displays the elements and illustrates trends in their properties. Compounds consist of a fixed ratio of elements held together by chemical bonds.
Chemical Reactions JQ

The periodic table organizes all known elements and contains information about atoms and molecules. It arranges metals, non-metals, and metalloids using elemental symbols. Elements are composed of atoms with a specific number of protons. Compounds consist of two or more elements in a fixed ratio that are bonded together. Chemical reactions transform one set of substances into another, either spontaneously or through external energy sources, while physical changes only alter the state of matter.
Chemical Reactions TG

Elements are made up of atoms that contain protons, neutrons and electrons. Atoms combine to form compounds made of two or more elements that can be separated through chemical reactions. Chemical reactions involve elements interacting to form new compounds, which can sometimes produce byproducts like gases. A chemical change alters the substances chemically while a physical change only modifies the shape or form. Reaction rates can be affected by factors like temperature, with heat speeding them up and cold slowing them down.
Chemical Reactions EL

Elements are substances made of only one type of atom. Atoms contain protons, neutrons and electrons. Metals are elements that conduct heat and electricity, while non-metals have different properties and are usually gases or brittle solids. The periodic table displays all the known chemical elements and their properties in an organized format. Compounds are formed when two or more elements chemically combine, and chemical reactions involve transformations between substances that cannot be reversed.
Chemical Reactions SB

The document defines key chemistry concepts such as atoms, elements, compounds, and molecules. Atoms are the fundamental units of matter made up of protons, neutrons, and electrons. Elements are pure substances composed of only one type of atom, while compounds contain two or more elements bonded together. The document also discusses chemical and physical changes, factors that affect reaction rates, and provides examples of metals and nonmetals.
chemical Reactions DM

Elements are pure substances made of one type of atom, while atoms are composed of protons, neutrons, and electrons. Metals are elements or alloys that conduct heat and electricity well, whereas nonmetals are usually gases or weak solids that lack metallic properties. A compound is formed when two or more elements chemically bond together in fixed proportions, and a chemical reaction transforms one set of chemicals into another through processes like producing heat, light, or precipitate.
Chemical Reactions CD

The document discusses several key chemistry concepts:
- Atoms are made up of protons, neutrons, and electrons. An element is made of a single type of atom.
- Compounds are made of two or more elements chemically bonded together. Chemical reactions involve breaking and forming bonds to change one substance into another.
- Physical changes alter a substance's properties like shape or state but not its chemical makeup. Chemical reactions alter the substance's chemical identity and atomic/molecular structure.
U2L6 - Interactions of Atoms

This document discusses chemical bonding and interactions between atoms. It explains that during chemical changes, atoms are rearranged and bonds are formed and broken, but atoms are neither created nor destroyed. Chemical equations are used to represent chemical reactions, with reactants on the left and products on the right. Balanced equations show the conservation of mass with equal numbers and kinds of atoms on both sides. Bohr models represent atoms with electrons in energy levels around the nucleus and show how valence electrons participate in bonding to achieve stable configurations.
nature of bonding of chemistry

- Organic chemistry involves carbon-based compounds, with hydrogen usually being the second most common element. Other common elements are oxygen, nitrogen, sulfur, and halogens.
- Most bonds in organic compounds are covalent, formed through shared electron pairs between atoms. Whether bonds are ionic or covalent depends on the electro-negativity difference between bonded atoms.
- Polar covalent bonds form when there is some electro-negativity difference between atoms, causing the electron cloud to shift slightly towards the more electronegative atom. This gives the atoms partial positive or negative charges.
Ch10

The document discusses the formation of chemical bonds between atoms and different types of bonds including ionic bonds, metallic bonds, covalent bonds, polar bonds, and van der Waals forces. It also covers chemical reactions, states of matter, and polymerization reactions.
Chapter01

This document provides an overview of basic chemistry concepts. It begins by classifying matter as either mixtures or pure substances, with pure substances further divided into elements and compounds. Elements contain only one type of atom, while compounds contain two or more different types of atoms combined in fixed ratios. The three common states of matter - solids, liquids, and gases - are described based on how tightly or loosely the particles are packed. Key concepts like the mole, molar mass, empirical and molecular formulas are also introduced. Measurement units commonly used in chemistry like grams, meters, and moles are defined according to the International System of Units.
Presentation c.elements

The document discusses the basic concepts of chemical elements and compounds. It defines a chemical element as a substance that cannot be broken down further by chemical means, and describes elements as the basic building blocks of matter. It then discusses the structure of atoms, isotopes, electrons and their energy levels, and how chemical bonds including ionic, covalent and hydrogen bonds form between elements and allow the creation of chemical compounds. It also briefly touches on mixtures vs compounds and the different states of matter.
Atoms and Molecules

- The document discusses atoms, ions, molecules, elements, compounds, and chemical formulas and reactions.
- Atoms combine to form molecules, and molecules made of the same type of atom are elements, while molecules with different atom types are compounds.
- Chemical formulas use symbols and subscripts to represent the elements and number of each type of atom in a molecule or compound.
- Chemical reactions can release or absorb energy and are classified as exothermic or endothermic respectively.
Atomsandmolecules 100919202417-phpapp02

- The document discusses atoms, ions, molecules, elements, compounds, and chemical formulas and reactions.
- Atoms combine to form molecules, and molecules made of the same type of atom are elements, while molecules with different atom types are compounds.
- Chemical formulas use symbols and subscripts to represent the elements and number of each type of atom in a molecule or compound.
- Chemical reactions can release or absorb energy and are classified as exothermic or endothermic respectively.
Notes chemical formulas

The document discusses chemical formulas and their use in representing substances. It explains that chemical formulas use symbols to show the elements present in a compound and subscripts to indicate the number of atoms if more than one atom is present. Coefficients are also used to show the number of molecules if more than one molecule makes up the total substance. Examples of common chemical formulas are provided along with the total number of atoms and molecules they represent.
How chemical reaction occurs

1. A chemical reaction occurs when the chemical bonds in molecules are broken and new bonds are formed, creating new substances. This can happen through various processes, including atoms or ions gaining, losing, or sharing electrons to achieve stable electron configurations.
2. Examples of chemical reactions include rusting of steel which combines iron and oxygen, or the mixing of hydrogen and oxygen gases which produces an explosive reaction to form water.
3. Many chemical reactions occur as part of larger reaction sequences, such as the multiple steps that plants use in photosynthesis to ultimately produce glucose molecules from carbon dioxide and water.
Chemical Reactions MB

An element is a pure substance made of only one type of atom, distinguished by its atomic number. An atom is the smallest unit of an element, and metals are elements that conduct heat and electricity well while being malleable. Non-metals are generally poor conductors that are brittle. The periodic table displays all known elements arranged by atomic structure. Compounds are formed by two or more elements bonding together. Chemical reactions lead to new substances through breaking and forming of bonds.
A&P basic chemistry, atoms to ions, bonding, molecules v compounds, water and...

This document provides an overview of key concepts in chemistry including:
1) Matter is made up of atoms, which are the basic building blocks. Atoms can combine to form molecules or ionic compounds.
2) The periodic table describes all known elements and their properties. Atoms can gain or lose electrons to form ions and achieve stability.
3) Chemical bonds form when atoms share or transfer electrons. Ionic bonds form when an electron is transferred between atoms to form ions, while covalent bonds form when electrons are shared between atoms.
Reactions20082

The document describes different types of chemical reactions: synthesis reactions occur when two substances combine to form a compound; decomposition reactions occur when a single substance breaks down into simpler parts upon heating; single replacement reactions involve one metal displacing another from a compound; double replacement reactions involve the exchange of ions between two compounds to form new compounds. The document also notes that a chemical equation represents the reactants on the left and products on the right, and that the law of conservation of mass means the mass of reactants equals the mass of products in a chemical reaction.
Properties Of Matter Elements Compounds And Bonding

This document discusses key concepts in chemistry including the structure of atoms, elements, compounds, and chemical bonding. It explains that atoms are made up of protons, neutrons, and electrons; that elements are composed of only one type of atom; and that compounds are formed by chemical combinations of two or more elements. The three main types of chemical bonding - ionic, covalent, and polar covalent - are introduced along with examples of how they form compounds. Properties of water related to its chemical structure and hydrogen bonding are also summarized.
The structure of_matter

The document discusses the structure of matter and its chemical basis. It defines matter as anything that has weight and takes up space, including solids, liquids, and gases. All matter is composed of elements, which are made up of atoms. Atoms consist of a nucleus and electrons, and can bond together through ionic, covalent, or hydrogen bonds to form molecules and compounds. Chemical reactions form or break bonds between atoms and molecules.
Anatomy & Physiology Lecture Notes - Ch. 2 chemistry- part 1

website: http://www.am-medicine.com
Facebook page : https://www.facebook.com/pages/Am-medicine/207726329406832
Facebook group: https://www.facebook.com/groups/1409138472653811/
Changing matter

There are two types of matter changes: physical changes and chemical changes. Physical changes alter the physical properties of matter without changing its chemical composition, such as changes in state from solid to liquid to gas. Chemical changes form new chemical substances through rearrangement of atoms, such as burning or rusting. Chemical changes are identified through evidence like gas formation, color change, or energy transfer, and matter is conserved before and after the reaction according to the law of conservation of mass.
Chemistry

The document is an outline for a chemistry textbook covering topics like the composition of matter, energy, and water and solutions. It defines key terms like elements, atoms, isotopes, compounds, and bonds. It describes states of matter and energy changes during chemical reactions. It also explains the polarity and hydrogen bonding of water molecules and how this relates to properties like solubility, cohesion, adhesion and temperature moderation. Finally, it covers acids, bases, pH and buffers.
Compounds, chem formulas & covalent bonds

Compounds are substances made of two or more elements that often have different properties than their constituent elements. Atoms bond together in compounds via chemical bonds formed by sharing valence electrons, called covalent bonds. A covalent bond forms when two atoms share one or more pairs of valence electrons, with single bonds sharing one pair and double or triple bonds sharing more pairs. The sharing of electrons leads to stable covalent compounds with properties like low melting and boiling points.
Chemical Bonding

The atoms of molecules are linked together through a reaction known as chemical bonding. A chemical bond is a force that holds atoms together.
Chemical Reactions KW

This document provides an overview of key chemistry concepts including:
1) Elements are substances that cannot be broken down further through chemical means. There are around 118 known elements which combine to form all matter through atoms.
2) Atoms are composed of protons, neutrons and electrons. Atoms combine to form elements listed in the periodic table. Metals readily lose electrons to form positive ions surrounded by delocalized electrons.
3) Chemical reactions lead to the transformation of substances and can be spontaneous or non-spontaneous. They involve changes in electron motion and the formation/breaking of chemical bonds. Examples of reaction types include synthesis and decomposition.
Chemical Reactions MB

An element is a pure substance made of only one type of atom, distinguished by its atomic number. An atom is the smallest unit of an element, and metals are elements that conduct heat and electricity well while being malleable. Non-metals are generally poor conductors that are brittle. The periodic table displays all known elements arranged by atomic structure. Compounds are formed by two or more elements chemically bonding together. Chemical reactions lead to new substances through breaking and forming of bonds.
Chemical Reactions PP

An element is a pure substance that cannot be broken down further. An atom is the smallest particle of an element that retains the element's properties. The periodic table organizes the known elements based on their atomic structure and properties. Compounds are made of two or more elements chemically bonded together. Chemical reactions involve the rearrangement of atomic bonds to form new substances, while physical changes do not create new compounds.
Basic Terminologies IN Chemistry, Applications of Inorganic Chemistry

This document provides an assignment for an Inorganic Chemistry course. It includes definitions for key terms in chemistry such as atom, molecule, element, compound, and chemical bonding. It also defines different states of matter and phase changes. The document lists common laboratory equipment used in chemistry like beakers, Bunsen burners, and analytical balances. It concludes with examples of how inorganic chemistry is applied in environmental science and manufacturing fibers and plastics.
More Related Content
What's hot
nature of bonding of chemistry

- Organic chemistry involves carbon-based compounds, with hydrogen usually being the second most common element. Other common elements are oxygen, nitrogen, sulfur, and halogens.
- Most bonds in organic compounds are covalent, formed through shared electron pairs between atoms. Whether bonds are ionic or covalent depends on the electro-negativity difference between bonded atoms.
- Polar covalent bonds form when there is some electro-negativity difference between atoms, causing the electron cloud to shift slightly towards the more electronegative atom. This gives the atoms partial positive or negative charges.
Ch10

The document discusses the formation of chemical bonds between atoms and different types of bonds including ionic bonds, metallic bonds, covalent bonds, polar bonds, and van der Waals forces. It also covers chemical reactions, states of matter, and polymerization reactions.
Chapter01

This document provides an overview of basic chemistry concepts. It begins by classifying matter as either mixtures or pure substances, with pure substances further divided into elements and compounds. Elements contain only one type of atom, while compounds contain two or more different types of atoms combined in fixed ratios. The three common states of matter - solids, liquids, and gases - are described based on how tightly or loosely the particles are packed. Key concepts like the mole, molar mass, empirical and molecular formulas are also introduced. Measurement units commonly used in chemistry like grams, meters, and moles are defined according to the International System of Units.
Presentation c.elements

The document discusses the basic concepts of chemical elements and compounds. It defines a chemical element as a substance that cannot be broken down further by chemical means, and describes elements as the basic building blocks of matter. It then discusses the structure of atoms, isotopes, electrons and their energy levels, and how chemical bonds including ionic, covalent and hydrogen bonds form between elements and allow the creation of chemical compounds. It also briefly touches on mixtures vs compounds and the different states of matter.
Atoms and Molecules

- The document discusses atoms, ions, molecules, elements, compounds, and chemical formulas and reactions.
- Atoms combine to form molecules, and molecules made of the same type of atom are elements, while molecules with different atom types are compounds.
- Chemical formulas use symbols and subscripts to represent the elements and number of each type of atom in a molecule or compound.
- Chemical reactions can release or absorb energy and are classified as exothermic or endothermic respectively.
Atomsandmolecules 100919202417-phpapp02

- The document discusses atoms, ions, molecules, elements, compounds, and chemical formulas and reactions.
- Atoms combine to form molecules, and molecules made of the same type of atom are elements, while molecules with different atom types are compounds.
- Chemical formulas use symbols and subscripts to represent the elements and number of each type of atom in a molecule or compound.
- Chemical reactions can release or absorb energy and are classified as exothermic or endothermic respectively.
Notes chemical formulas

The document discusses chemical formulas and their use in representing substances. It explains that chemical formulas use symbols to show the elements present in a compound and subscripts to indicate the number of atoms if more than one atom is present. Coefficients are also used to show the number of molecules if more than one molecule makes up the total substance. Examples of common chemical formulas are provided along with the total number of atoms and molecules they represent.
How chemical reaction occurs

1. A chemical reaction occurs when the chemical bonds in molecules are broken and new bonds are formed, creating new substances. This can happen through various processes, including atoms or ions gaining, losing, or sharing electrons to achieve stable electron configurations.
2. Examples of chemical reactions include rusting of steel which combines iron and oxygen, or the mixing of hydrogen and oxygen gases which produces an explosive reaction to form water.
3. Many chemical reactions occur as part of larger reaction sequences, such as the multiple steps that plants use in photosynthesis to ultimately produce glucose molecules from carbon dioxide and water.
Chemical Reactions MB

An element is a pure substance made of only one type of atom, distinguished by its atomic number. An atom is the smallest unit of an element, and metals are elements that conduct heat and electricity well while being malleable. Non-metals are generally poor conductors that are brittle. The periodic table displays all known elements arranged by atomic structure. Compounds are formed by two or more elements bonding together. Chemical reactions lead to new substances through breaking and forming of bonds.
A&P basic chemistry, atoms to ions, bonding, molecules v compounds, water and...

This document provides an overview of key concepts in chemistry including:
1) Matter is made up of atoms, which are the basic building blocks. Atoms can combine to form molecules or ionic compounds.
2) The periodic table describes all known elements and their properties. Atoms can gain or lose electrons to form ions and achieve stability.
3) Chemical bonds form when atoms share or transfer electrons. Ionic bonds form when an electron is transferred between atoms to form ions, while covalent bonds form when electrons are shared between atoms.
Reactions20082

The document describes different types of chemical reactions: synthesis reactions occur when two substances combine to form a compound; decomposition reactions occur when a single substance breaks down into simpler parts upon heating; single replacement reactions involve one metal displacing another from a compound; double replacement reactions involve the exchange of ions between two compounds to form new compounds. The document also notes that a chemical equation represents the reactants on the left and products on the right, and that the law of conservation of mass means the mass of reactants equals the mass of products in a chemical reaction.
Properties Of Matter Elements Compounds And Bonding

This document discusses key concepts in chemistry including the structure of atoms, elements, compounds, and chemical bonding. It explains that atoms are made up of protons, neutrons, and electrons; that elements are composed of only one type of atom; and that compounds are formed by chemical combinations of two or more elements. The three main types of chemical bonding - ionic, covalent, and polar covalent - are introduced along with examples of how they form compounds. Properties of water related to its chemical structure and hydrogen bonding are also summarized.
The structure of_matter

The document discusses the structure of matter and its chemical basis. It defines matter as anything that has weight and takes up space, including solids, liquids, and gases. All matter is composed of elements, which are made up of atoms. Atoms consist of a nucleus and electrons, and can bond together through ionic, covalent, or hydrogen bonds to form molecules and compounds. Chemical reactions form or break bonds between atoms and molecules.
Anatomy & Physiology Lecture Notes - Ch. 2 chemistry- part 1

website: http://www.am-medicine.com
Facebook page : https://www.facebook.com/pages/Am-medicine/207726329406832
Facebook group: https://www.facebook.com/groups/1409138472653811/
Changing matter

There are two types of matter changes: physical changes and chemical changes. Physical changes alter the physical properties of matter without changing its chemical composition, such as changes in state from solid to liquid to gas. Chemical changes form new chemical substances through rearrangement of atoms, such as burning or rusting. Chemical changes are identified through evidence like gas formation, color change, or energy transfer, and matter is conserved before and after the reaction according to the law of conservation of mass.
Chemistry

The document is an outline for a chemistry textbook covering topics like the composition of matter, energy, and water and solutions. It defines key terms like elements, atoms, isotopes, compounds, and bonds. It describes states of matter and energy changes during chemical reactions. It also explains the polarity and hydrogen bonding of water molecules and how this relates to properties like solubility, cohesion, adhesion and temperature moderation. Finally, it covers acids, bases, pH and buffers.
Compounds, chem formulas & covalent bonds

Compounds are substances made of two or more elements that often have different properties than their constituent elements. Atoms bond together in compounds via chemical bonds formed by sharing valence electrons, called covalent bonds. A covalent bond forms when two atoms share one or more pairs of valence electrons, with single bonds sharing one pair and double or triple bonds sharing more pairs. The sharing of electrons leads to stable covalent compounds with properties like low melting and boiling points.
Chemical Bonding

The atoms of molecules are linked together through a reaction known as chemical bonding. A chemical bond is a force that holds atoms together.
What's hot (18)
A&P basic chemistry, atoms to ions, bonding, molecules v compounds, water and...

A&P basic chemistry, atoms to ions, bonding, molecules v compounds, water and...
Properties Of Matter Elements Compounds And Bonding

Properties Of Matter Elements Compounds And Bonding
Anatomy & Physiology Lecture Notes - Ch. 2 chemistry- part 1

Anatomy & Physiology Lecture Notes - Ch. 2 chemistry- part 1
Similar to chemical Reactions DS
Chemical Reactions KW

This document provides an overview of key chemistry concepts including:
1) Elements are substances that cannot be broken down further through chemical means. There are around 118 known elements which combine to form all matter through atoms.
2) Atoms are composed of protons, neutrons and electrons. Atoms combine to form elements listed in the periodic table. Metals readily lose electrons to form positive ions surrounded by delocalized electrons.
3) Chemical reactions lead to the transformation of substances and can be spontaneous or non-spontaneous. They involve changes in electron motion and the formation/breaking of chemical bonds. Examples of reaction types include synthesis and decomposition.
Chemical Reactions MB

An element is a pure substance made of only one type of atom, distinguished by its atomic number. An atom is the smallest unit of an element, and metals are elements that conduct heat and electricity well while being malleable. Non-metals are generally poor conductors that are brittle. The periodic table displays all known elements arranged by atomic structure. Compounds are formed by two or more elements chemically bonding together. Chemical reactions lead to new substances through breaking and forming of bonds.
Chemical Reactions PP

An element is a pure substance that cannot be broken down further. An atom is the smallest particle of an element that retains the element's properties. The periodic table organizes the known elements based on their atomic structure and properties. Compounds are made of two or more elements chemically bonded together. Chemical reactions involve the rearrangement of atomic bonds to form new substances, while physical changes do not create new compounds.
Basic Terminologies IN Chemistry, Applications of Inorganic Chemistry

This document provides an assignment for an Inorganic Chemistry course. It includes definitions for key terms in chemistry such as atom, molecule, element, compound, and chemical bonding. It also defines different states of matter and phase changes. The document lists common laboratory equipment used in chemistry like beakers, Bunsen burners, and analytical balances. It concludes with examples of how inorganic chemistry is applied in environmental science and manufacturing fibers and plastics.
Atomic Theory

The document discusses the classification of elements and the periodic table. It explains that elements are arranged in the periodic table according to their atomic structure and properties, with similar elements grouped together. The periodic table is divided into rows and columns, with each row representing an energy level and each column representing common chemical properties. The position of an element in the table predicts its chemical behavior and reactivity with other elements.
Practical Analytical Instrumentation in On-line Applications

This document provides an overview of basic chemistry concepts. It defines elements, compounds, and mixtures, and discusses atomic structure and the periodic table. The periodic table is described as arranging the elements in order of atomic number and classifying them into groups with similar properties. Key groups discussed are the alkali metals, alkaline earth metals, noble gases, and halogens. The document also covers the formation of ions through gaining or losing electrons, and different bonding types including ionic and covalent.
Chapter 2

The document introduces basic chemistry concepts such as the composition of matter, elements and their symbols, atomic structure, bonds between atoms including ionic and covalent, states of matter, chemical reactions and energy, and acid-base solutions including pH and buffers. It defines terms like elements, atoms, protons, neutrons, electrons, compounds, molecules, and provides examples of chemical reactions and bonds between different types of atoms.
Elements and compounds

This document provides an overview of elements, compounds, and how they relate. It defines elements as pure substances made of single atom types, while compounds are formed when two or more different elements bond together. Elements are represented by symbols on the periodic table and have distinct properties based on their atomic structure. Compounds have new properties and are represented by formulas showing the elements present and their ratios. The document explains how ionic and covalent bonds form compounds from elements and provides examples of common elements, compounds, and how to determine compound formulas from their constituent ions.
Chapters 2&3

This document provides a summary of basic chemistry concepts covered in chapters 2 and 3 of a unit 1 notes document. It discusses the following key points in 3 sentences:
Matter is anything that takes up space and has specific physical and chemical properties. Atoms are the basic building blocks of matter and consist of a nucleus with protons and neutrons surrounded by electrons. Compounds are formed when two or more elements chemically combine, resulting in new substances with different properties than the individual elements.
Elements, Compounds, Mixtures

This document discusses the classification of matter into elements, compounds, and mixtures. It defines each term and provides examples. Elements are pure substances made of only one type of atom that cannot be broken down further. Compounds are pure substances composed of two or more elements chemically bonded together in fixed ratios to form new substances with different properties from the original elements. Mixtures are physical combinations of elements or compounds that are not chemically bonded and can be separated by physical means such as filtration or evaporation.
Atomic Theory

The document discusses the periodic table and classification of elements. It explains that (1) elements are arranged in the periodic table based on their atomic structure and chemical properties, which repeat periodically. (2) Each element is identified by its atomic number and atomic mass. (3) Elements are classified as metals, nonmetals, and metalloids based on their location in the periodic table and tendency to gain or lose electrons.
Atoms, elements, and compounds notes

Atoms are the basic building blocks of matter and are made up of protons, neutrons, and electrons. Elements are pure substances made of only one type of atom, such as sodium (Na) or oxygen (O). Compounds are formed when two or more elements chemically combine, resulting in a substance with totally new properties compared to the original elements. The periodic table provides information about each element's atomic number, chemical symbol, and atomic mass.
chemical Reactions RP

Atoms are the basic building blocks of matter and can join to form molecules and objects. Elements are substances made of only one type of atom, which are classified based on their number of protons. Metals are good conductors of heat and electricity, are generally denser than other substances, and are malleable and ductile. Non-metals have properties unlike metals and are usually gases. The periodic table arranges elements in order of atomic mass to show their properties. Compounds contain two or more elements chemically bonded together. Chemical reactions transform substances into different chemicals through chemical processes. Physical changes involve changes in phase like ice melting into water.
Periodic table

The document provides information about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are arranged based on their atomic number and can be grouped into families that show similar traits. The periodic table provides a way to predict chemical behaviors and allows scientists to identify unknown elements based on trends in atomic structure.
Periodic table

The document summarizes key concepts about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are classified into families based on their valence electrons and include metals, non-metals, metalloids and noble gases. The periodic table provides a way to predict an element's properties and reactivity.
Chapter2

This chapter introduces fundamental concepts in chemistry including atoms, molecules, ions and chemical formulas. It discusses atomic theory and how experiments led to the discovery that atoms consist of electrons, protons and neutrons. The chapter also introduces the periodic table and explains how elements are arranged based on atomic structure and properties. It describes how atoms can form ions and molecules, and how ionic and molecular compounds are named using chemical formulas.
Chemistry

This document provides an overview of key concepts in earth/environmental science chemistry including:
- Matter is anything that has mass and takes up space. Density measures the ratio of mass to volume.
- Temperature is measured in Kelvin, Celsius, and Fahrenheit scales. States of matter include solids, liquids, gases, and plasma.
- Atoms are the basic units that make up elements. The nucleus contains protons and neutrons. Electrons surround the nucleus.
- The periodic table organizes elements and shows their properties. Elements bond through ionic or covalent bonds to form compounds.
Chapt02 chemistry

The document summarizes key concepts about the nature of atoms and molecules, including:
- Atoms are composed of protons, neutrons, and electrons. The number of protons determines the element.
- Electrons can be gained or lost, creating ions with positive or negative charges.
- Elements are arranged in the periodic table based on their atomic structure.
- Chemical bonds, including ionic and covalent bonds, determine how atoms combine to form molecules.
The structure of_matter

This document discusses the structure and composition of matter. It explains that all matter is made up of elements, which are composed of atoms. Atoms contain protons, neutrons, and electrons. Elements can bond together through ionic bonds, covalent bonds, or hydrogen bonds to form molecules or compounds. Some important compounds are electrolytes, acids, bases, and salts. Chemical reactions cause bonds to form or break as atoms rearrange. The human body relies on many chemical processes and requires maintaining a narrow pH range in the blood for survival.
Chemistry chapter 3 number2

The document discusses the key concepts of elements, atoms, ions, and the periodic table. It covers early atomic theories proposed by scientists like Dalton, Thomson, and Rutherford. Key discoveries included that atoms are made up of a tiny, dense nucleus surrounded by electrons. The periodic table organizes elements by increasing atomic number and groups elements with similar properties together. Elements can form ions by gaining or losing electrons to become charged particles like cations and anions. Ionic compounds are formed from the attraction between oppositely charged ions.
Similar to chemical Reactions DS (20)
Basic Terminologies IN Chemistry, Applications of Inorganic Chemistry

Basic Terminologies IN Chemistry, Applications of Inorganic Chemistry
Practical Analytical Instrumentation in On-line Applications

Practical Analytical Instrumentation in On-line Applications
chemical Reactions DS
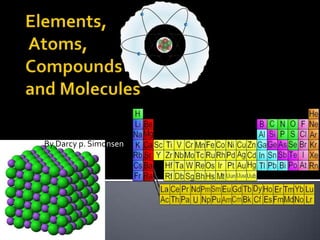
- 1. Elements, Atoms, Compounds and Molecules By Darcy p. Simonsen
- 2. Element A chemical element is a pure chemical substance consisting of one type of atom distinguished by its atomic number. Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen. In total, 117 elements have been observed as of 2008, of which 94 occur naturally on Earth
- 3. Atom The atom is a basic unit of matter consisting of a dense, central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons.
- 4. metals a metal is an element, compound, or alloy characterized by high electrical conductivity . In a metal, atoms readily lose electrons to form positive ions; those ions are surrounded by delocalized electrons, which are responsible for the conductivity.
- 5. Non metals non-metal is a term used in chemistry when classifying the chemical elements. On the basis of their general physical and chemical properties, every element in the periodic table can be termed either a metal or a nonmetal.
- 6. The periodiс table of the chemical elementsis a tabulardisplay of the chemical elements. Although precursors to this table exist, its invention is generally credited to Russian chemist Dmitri Mendeleev in 1869, who intended the table Periodic table
- 7. Compounds A chemical compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions and that have a unique and defined chemicalstructure.
- 8. Chemical reactions A chemical reaction is a process that leads to the transformation of one set of chemical substances toanother. There are three things that speed up chemical reactions one of them are:
- 9. chemical and physical changes 1. A physical change is reversible, a chemical change is not. For example, the freezing of water would be a physical change because it can be reversed, whereas the burning of wood is a chemical change - you can't 'unburn' it.
- 10. Identifying chemical reactions There are three types of chemical reactions, here are two: Synthesis A synthesis reaction is when there is a combination of two or more substances and a compound results. An example A + B --> AB Decomposition Decomposition is roughly the opposite of synthesis it is a breaking down of one molecule into two (or more) smaller molecules or elements. Sample decomposition reactions could include: 2 H2O + electricity -> 2 H2 + O2
- 11. make a reaction faster use a catalyst breaking a solid into something smaller (to increase its surface area) make the solution more concentrated, increase the temperature.
- 12. Bibliography Wikipedia the free encyclopaedia Google images What is the difference between a physical change and a chemical...