Centrifuge
- 1. CENTRIFUGES
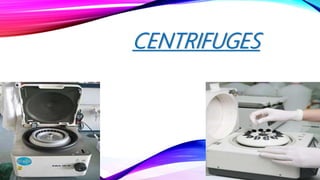
- 2. DEFINITION • A laboratory centrifuge • is laboratory equipment, • driven by a motor, • spins liquid samples at high speed. • There are various types of centrifuges, depending on the size and the sample capacity. • They vary widely in speed and capacity • work by the sedimentation principle, where the centripetal acceleration is used to separate substances of greater and lesser density. • comprise a rotor containing two, four, six, or many more numbered wells within which the samples containing centrifuge tips may be placed.
- 3. HISTORY • English military engineer Benjamin Robins (1707-1751) invented a whirling arm apparatus to determine drag. • In 1864, Antonin Prandl invented the first dairy centrifuge in order to separate cream from milk. • In 1879, Gustaf de Laval demonstrated the first continuous centrifugal separator, making its commercial application feasible.
- 4. PRINCIPLE OF CENTRIFUGE A centrifuge is a piece of equipment, generally driven by an electric motor, that puts an object in rotation around a fixed axis, applying a force perpendicular to the axis to separate substances of different densities. Tubes in the centrifuge are tilted so centrifugal force can pull denser substances towards the bottom of the tube. Relative Centrifugal Force (RCF) measures acceleration applied to the sample RCF=(r*ω2)/g r=distance from center or motor to sample ω= angular velocity (radians/ second) g=9.8m/s2
- 5. OPERATION • Increasing the effective gravitational force will more rapidly and completely cause the precipitate to gather on the bottom of the tube as a "pellet". • The remaining solution is called the "supernate" or "supernatant". • The supernatant liquid is then either • decanted from the tube without disturbing the pellet, or • withdrawn with a Pasteur pipette. • The rate of centrifugation is specified by the acceleration applied to the sample, typically measured in revolutions per minute (RPM) or relative centrifugal force (RCF).
- 6. • The particles' settling velocity in centrifugation is a function of their • size and shape, • centrifugal acceleration, • the volume fraction of solids present, • the density difference between the particle and the liquid, and the viscosity.
- 7. ROTOR OBJECTIVES Generally spoken, there are two main types of rotors: • Fixed-angle rotor • The rotor (mainly made of aluminum) is very compact. • There are boreholes with a specific angle (like 45°) within the rotor. • These boreholes are used for the sample tubes.
- 8. •Swing-out rotor (= horizontal rotor) •The rotor looks like a cross with bucket. •Within these buckets, different tubes can be centrifuged. •For a safe centrifugation, a specific adapter for every tube shape is mandatory.
- 9. TYPES OF CENTRIFUGES • Centrifuges are generally divided into 3 categories based on their maximum attainable speed: 1. "Low-speed": max ~5 x 103 rpm. 2. "High-speed": max ~2 x 104 rpm. 3. "Ultracentrifuges": max ~105 rpm Others Variable Temperatures Vacuum Capabilities
- 10. TYPES OF CENTRIFUGE There are at least five types of centrifuge: • preparative centrifuge • analytical centrifuge • angle fixed centrifuge • swing head centrifuge • hematocrit centrifuge
- 17. CENTRIFUGE TUBES • Glass centrifuge tubes can be used with most solvents, but tend to be more expensive. They can be cleaned like other laboratory glassware, and can be sterilized by autoclaving. • Plastic centrifuge tubes, especially micro-centrifuge tubes tend to be less expensive. Water is preferred when plastic centrifuge tubes are used. They are more difficult to clean thoroughly, and are usually inexpensive enough to be considered disposable
- 22. PROCEDURE Place tubes in centrifuge. Always use correct size tubes to prevent tube damage. Always using a counterbalance: By mass NOT volume Put the tubes opposite each other in the centrifuge. If you have more than two tubes, only the ones opposite each other have to be equal in mass. If centrifuge has variable speeds, enter RPM. Close lid. Turn timer on or press start. Remove the tubes carefully after the centrifuge has completely stopped spinning to prevent remixing.
- 25. •Result of unbalanced centrifuge
- 26. PREVENTIVE MAINTENANCE • Lubricate and clean motor. • Clean case. • Inspect power cords and plugs. • Inspect controls and switches. • Ensure appropriate menu settings for proper use. • Ensure tightness of rotor. • Check lights and indicators. • Verify that alarms are operating properly. • Ensure safety switch is functioning. • If refrigerated, ensure temperature reading is working. • Replace/repair gaskets, seals, and vacuum pump (if applicable).
- 27. COMMON FAILURES • Machine will not start. • Power supply is faulty. • Safety switch or timer not working properly. • Motor not working properly. • Motor making noise. • Rotor is broken. • Lid will not open or close.
- 28. APPLICATIONS • Separate solids from solution • Used during urinalysis to separate disease identifying components • Separate blood into plasma and cellular components • Remove cellular elements from blood to provide cell free serum or plasma • Conc. Cellular elements for microscopy • Remove protein precipitate from analytic sample • Isolation of macromolecules such as DNA rna proteins or lipids • Use in haematology lab for PCV Coagulation Studies • Blood Bank For Transfer Of Plasma After Donation Of blood
- 29. USED IN • Chemistry • Biology • Biochemistry • Microbiology • Hematology • Blood Bank
