Embed presentation
Download to read offline




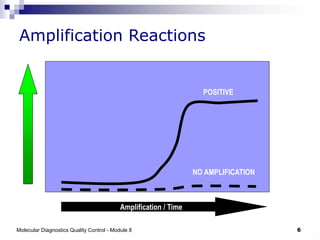


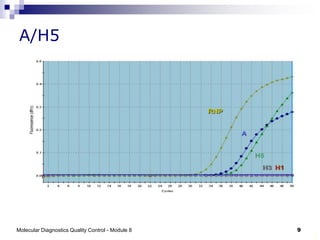

The document discusses quality control for molecular diagnostics including maintaining nucleic acid integrity during sample collection and transport, establishing contamination control through unidirectional workflow, and using positive and negative controls. It also describes real-time PCR processes, showing amplification curves for positive and negative samples, and examples for influenza A subtyping. A risk of in-house PCR is mentioned as using non-optimized primers and controls requiring cautious result interpretation.








