Cassidy Wang Poster Final
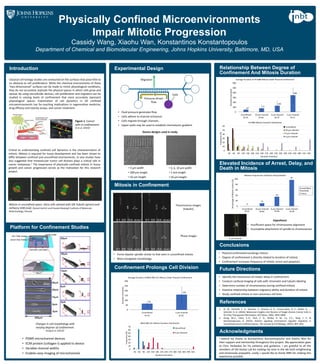
- 1. Physically Confined Microenvironments Impair Mitotic Progression Cassidy Wang, Xiaohu Wan, Konstantinos Konstantopoulos Department of Chemical and Biomolecular Engineering, Johns Hopkins University, Baltimore, MD, USA 0 10 20 30 40 50 60 30 60 90 120 150 180 210 240 270 300 330 360 390 420 450 480 510 540 Frequency (%) Duration (minutes) HT1080 Mitosis Duration Distribution Unconfined 10 μm channel 6 μm channel 3 μm channel Introduction Conclusions Elevated Incidence of Arrest, Delay, and Death in Mitosis Mitosis in Confinement Future Directions Acknowledgments I extend my thanks to Konstantinos Konstantopoulos and Xiaohu Wan for their support and mentorship throughout this project. My appreciation goes to Chris Yankaskas for his patience and guidance. I am grateful to all the members of the Kostas Lab for making my time in the lab both enlightening and immensely enjoyable. Lastly, I would like to thank INBT for making this experience possible. References Critical to understanding confined cell dynamics is the characterization of mitosis. Mitosis is required for tissue development and has been shown to differ between confined and unconfined environments. In vivo studies have also suggested that intravascular tumor cell division plays a critical role in cancer metastasis.1 The importance of physically confined mitosis in tissue growth and cancer progression serves as the motivation for this research project. Platform for Confinement Studies • PDMS microchannel devices • ECM protein (collagen-I) applied to device • Variable channel widths • Enables easy imaging of microchannels Confinement Prolongs Cell Division Relationship Between Degree of Confinement And Mitosis Duration • Physical confinement prolongs mitosis • Degree of confinement is directly related to duration of mitosis • Confinement increases frequency of mitotic arrest and apoptosis • Identify the mechanism of mitotic delay in confinement • Conduct confocal imaging of cells with chromatin and tubulin labeling • Determine number of chromosomes during confined mitosis • Examine relationship between migratory ability and duration of mitosis • Study confined mitosis in non-cancerous cell lines • Forms bipolar spindle similar to that seen in unconfined mitosis • More elongated morphology 1. Xi, W., Schmidt, C. K., Sanchez, S., Gracias, D. H., Carazo-Salas, R. E., Butler, R., ... Schmidt, O. G. (2016). Molecular Insights into Division of Single Human Cancer Cells in On-Chip Transparent Microtubes. ACS Nano, 10(6), 5835-5846. 2. Hung, W.-C., Chen, S.-H., Paul, C. D., Stroka, K. M., Lo, Y.-C., Yang, J. T., & Konstantopoulos, K. (2013). Distinct signaling mechanisms regulate migration in unconfined versus confined spaces. The Journal of Cell Biology, 202(5), 807–824. Experimental Design Classical cell biology studies are conducted on flat surfaces that pose little to no obstacle to cell proliferation. While the chemical environments of these “two-dimensional” surfaces can be made to mimic physiological conditions, they do not accurately replicate the physical spaces in which cells grow and spread. By using microfluidic devices, cell proliferation and migration can be studied in varying levels of confinement that more accurately represent physiological spaces. Examination of cell dynamics in 3D confined microenvironments has far-reaching implications in regenerative medicine, drug efficacy and toxicity assays, and cancer treatment. Figure 1. Cancer cells in confinement. Xi et al. (2016)1 0 10 20 30 40 50 60 70 30 60 90 120 150 180 210 240 270 300 330 360 390 420 Frequency (%) Duration (minutes) MDA-MB-231 Mitosis Duration Distribution Unconfined 3 μm channel Mitosis in unconfined space. HeLa cells stained with SiR-Tubulin (green) and mCherry H2B (red). (Daniel Gerlich and Claudia Blaukopf, Institute of Molecular Biotechnology, Vienna) • Fluid pressure generates flow • Cells adhere to channel entrances • Cells migrate through channels • Upper wells may be used to establish chemotactic gradient Changes in cell morphology with varying degrees of confinement. Hung et al. (2013)2 Cells Pressure-driven flow • 3, 6, 10 μm width • 1 mm length • 10 μm height • 3 μm width • 200 μm length • 10 μm height Fluorescence images (tubulin) Phase images 65 158 0 50 100 150 200 250 300 Unconfined 3 μm channel Duration (minutes) Average Duration of MDA-MB-231 Mitosis Under Physical Confinement N=74 N=25 6 μm channel 47 77 130 348 0 100 200 300 400 500 600 Unconfined 10 μm channel 6 μm channel 3 μm channel Duration (minutes) Average Duration of HT1080 Mitosis Under Physical Confinement N=53 N=62 N=37 N=11 Hypothesis • Insufficient space for chromosome alignment • Incomplete attachment of spindle to chromosomes 0 11 35 97 0 20 40 60 80 100 Unconfined 10 μm channel 6 μm channel 3 μm channel Percentage of Mitotic Events (%) Relative Frequencies of Mitotic Delays/Deaths Arrest/delay threshold: 3 hours N=53 N=66 N=46 N=30 Migration Device designs used in study
