This document provides instructions and reminders for a chemistry class. It includes:
1) A list of materials and assignments needed for class, including biochemical calculations, an updated lab log, and graphs to be created.
2) Reminders about upcoming tests and progress notices.
3) Background information on the most common elements that make up living things.
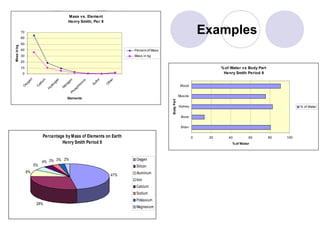
4) Directions for creating three graphs analyzing body composition and the distribution of elements on Earth.
5) Examples of the completed graphs.
6) A task list of assignments.
7) Instructions to complete homework by the next class period.