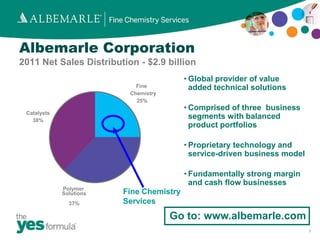
Albemarle Corporation is a global specialty chemicals company with three business segments and over 40 years of experience in continuous reactions. They offer custom manufacturing capabilities across 5 commercial scale multipurpose plants, with equipment ranging from bench to commercial scale. Their capabilities include complex, hazardous, and highly exothermic continuous reactions for applications such as agrochemicals, flame retardants, and personal care intermediates.