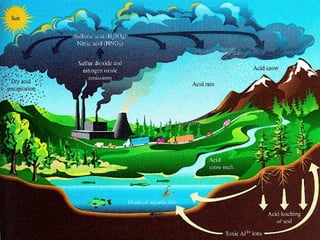
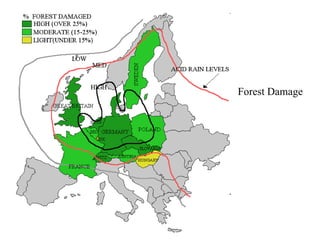
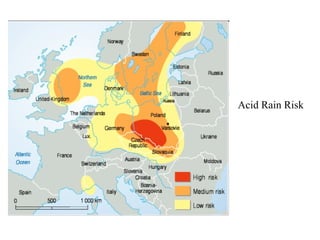
This document discusses acid rain, including its causes, effects, and real world examples. Acid rain is caused by sulfur dioxide and nitrogen oxides released into the air from burning fossil fuels. These gases react with water and other chemicals in the atmosphere to form acidic compounds. Acid rain can damage monuments by causing them to lose their appearance over time. It can also destroy natural habitats and affect plants and animal life. Some countries are more impacted by acid rain than others, even when they do not produce large amounts of the pollutants themselves, because acid rain can travel long distances in clouds.