9. quantum_numbers.energy levels, examplespdf
•
0 likes•2 views
energy sublevels, azimuthal, and more
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
Similar to 9. quantum_numbers.energy levels, examplespdf
Similar to 9. quantum_numbers.energy levels, examplespdf (20)
95electrons in the same orbital have different rus values .docx

95electrons in the same orbital have different rus values .docx
Quantum Numbers and Atomic Orbitals By solving t.pdf

Quantum Numbers and Atomic Orbitals By solving t.pdf
More from JenniferEbascoVicent
More from JenniferEbascoVicent (10)
1. Properties of Matter (solid, Liquid, and gases).ppt

1. Properties of Matter (solid, Liquid, and gases).ppt
10. Evidence of Evolution.pptx Fossils, comparative

10. Evidence of Evolution.pptx Fossils, comparative
Continental Drift.pptx explanation earth's interior

Continental Drift.pptx explanation earth's interior
Government and bussiness - economy and the environment

Government and bussiness - economy and the environment
Recently uploaded
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Recently uploaded (20)
FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry

FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry
Role of AI in seed science Predictive modelling and Beyond.pptx

Role of AI in seed science Predictive modelling and Beyond.pptx
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Human & Veterinary Respiratory Physilogy_DR.E.Muralinath_Associate Professor....

Human & Veterinary Respiratory Physilogy_DR.E.Muralinath_Associate Professor....
FAIRSpectra - Enabling the FAIRification of Analytical Science

FAIRSpectra - Enabling the FAIRification of Analytical Science
Module for Grade 9 for Asynchronous/Distance learning

Module for Grade 9 for Asynchronous/Distance learning
Climate Change Impacts on Terrestrial and Aquatic Ecosystems.pptx

Climate Change Impacts on Terrestrial and Aquatic Ecosystems.pptx
POGONATUM : morphology, anatomy, reproduction etc.

POGONATUM : morphology, anatomy, reproduction etc.
Genetics and epigenetics of ADHD and comorbid conditions

Genetics and epigenetics of ADHD and comorbid conditions
PATNA CALL GIRLS 8617370543 LOW PRICE ESCORT SERVICE

PATNA CALL GIRLS 8617370543 LOW PRICE ESCORT SERVICE
GBSN - Biochemistry (Unit 2) Basic concept of organic chemistry 

GBSN - Biochemistry (Unit 2) Basic concept of organic chemistry
THE ROLE OF BIOTECHNOLOGY IN THE ECONOMIC UPLIFT.pptx

THE ROLE OF BIOTECHNOLOGY IN THE ECONOMIC UPLIFT.pptx
Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b

Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b
Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds

Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds
9. quantum_numbers.energy levels, examplespdf
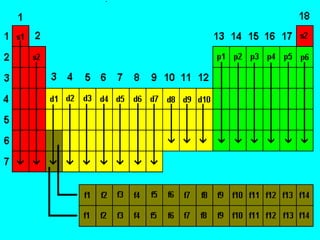
- 2. Energy Levels Energy Levels (7) Sublevels (4) s p d f 1 orbital 3 orbitals 5 orbitals 7 orbitals 2 e- 6 e- 10 e- 14 e-
- 3. QUANTUM NUMBERS Set of 4 numbers used to describe the electrons in terms of : a. Distance from the nucleus b. Shape of the orbitals c. Orientation in space d. Direction of electron spin
- 6. PRINCIPAL QUANTUM NUMBER, n - Refers to the main energy levels - Related to the average distance of the electron from the nucleus - Can only have integral values of n = 1, 2, 3, 4 etc.
- 10. AZIMUTHAL or SECONDARY QUANTUM NUMBER, l - Defines the shape of the orbital. - Also refers to the energy sublevels
- 13. MAGNETIC QUANTUM NUMBER, ml Describes the orientation of the orbitals in space. The range of magnetic quantum numbers defined by the formula ml = 2l + 1 and takes the integer value between –l to +l including 0
- 17. SPIN QUANTUM NUMBER, ms - differentiates how two electrons behave under a magnetic field. - Can only have two possible values +½ and -½
- 21. Example. What are the possible quantum numbers for the outermost electron of oxygen? SOLUTION: Identify the final orbital occupied by the electron. 1s2 2s2 2p4 2 p4 n = l = ml = ms = 2 1 -1, 0, +1 +½ , -½
- 22. What are the possible quantum numbers for the outermost electron of chromium? SOLUTION: Identify the final orbital occupied by the electron. 1s2 2s2 2p6 3s2 3p6 4s1 3d5 3 d5 n = l = ml = ms = 3 2 -2, -1, 0, +1, +2 +½ , -½
- 24. Electron Population Distribution or Electron Orbital Notation Show the electron population distribution for 15P. P has 15 electrons. Its electron configuration is 1s2 2s2 2p6 3s2 3p3. The electron population distribution for P is: 1s 2s 2p 3s 3p