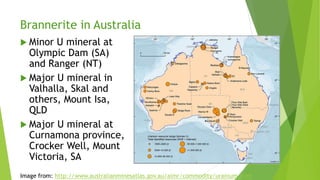
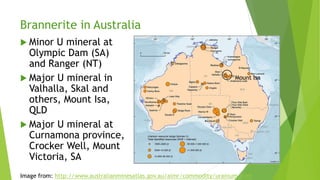
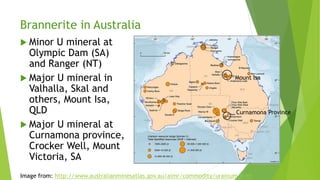
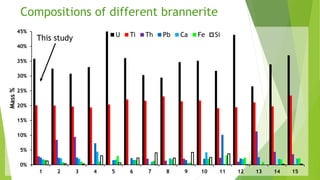
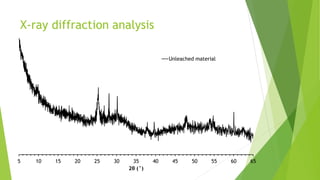
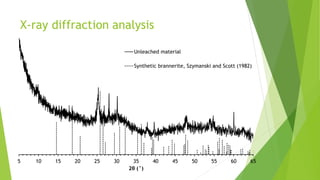
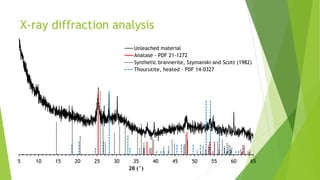
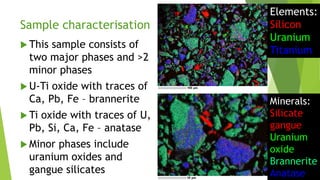
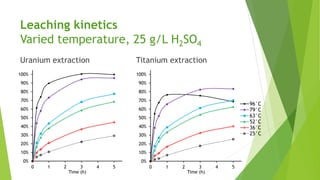
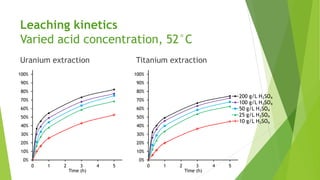
Brannerite is a uranium-titanium mineral that requires aggressive leaching conditions to dissolve. Experiments were conducted to characterize brannerite samples from Australia and study how temperature and acid concentration impact leaching kinetics. At lower temperatures and acidity, brannerite dissolved via a reaction releasing uranium oxide and titanium dioxide. At higher temperatures, a new reaction mechanism was observed involving uranium and titanium sulfate complexes that hydrolyzed to release uranium and form anatase. Dissolution was strongly influenced by temperature and extraction of uranium generally exceeded titanium. The results provide new insights into brannerite leaching reactions and conditions.