Cytological methods
- 1. CYTOLOGICAL METHODS Prof. Ichha Purak Department of Botany Ranchi Women’s College, Ranchi 11/14/2021 Cytological Methods 1
- 2. METHODS OF SLIDE PREPARATION FOR CYTOLOGICAL STUDIES A) Plant mitosis (Onion,Garlic,Lens,Vicia,Pea) B) Mitosis in Charophytes (Chara,Nitella,Tolypella ) C) Plant Meiosis (Onion,Rhoeo discolor ) 11/14/2021 Cytological Methods 2
- 3. A. Plant mitosis (Onion,Garlic,Lens,Vicia,Pea) STEPS : Selection of material Root tip cutting Pretreatment of root tips Fixation of root tip Storage of root tips Staining Temporary slide preparation Tapping Squashing and Sealing Making the slide permanent Observation 11/14/2021 Cytological Methods 3
- 4. SELECTION OF MATERIAL Mitosis occurs in all meristematic cells so can be worked out from root tips, shoot tips, leaf tips etc . Among these root tips are the most actively growing parts and are devoid of pigments ,so can be used to study mitosis. For the demonstration of different stages of mitosis, root tips of Allium cepa are the most ideal material. Root tips can be obtained easily from bulbs (Onion and Garlic - whole or cloves ) / germinating seeds (Lens,Vicia,Pea,Gram, Hordeum /Onion,Groundnut etc.) 11/14/2021 Cytological Methods 4
- 5. Method of growing onion/garlic root tips from bulbs Old roots from bulb of onion or garlic are cut and removed by sharp knife Bulbs are placed inverted on wide mouth bottle or specimen tube or ice tray having water or on a plate having wet sand Left for 1-2 -6-8 days, if temperature is high ,covered by poly bag to keep moist White coloured root tips emerge out in bunches in 2-4 days 11/14/2021 Cytological Methods 5
- 7. Methods of growing root tips from seeds of Lens culinaris / Vicia faba / Gram/Pea /Allium cepa /Trigonella/Groundnut Fresh and healthy seeds are taken a) The seeds are washed and soaked in water for one day and then seeds are spread under the layer of soil or in petri plates with filter papers or cotton or sand in plate. Or seeds are tied up in wet cloth . Seeds are kept moist. b) Generally seeds germinate in 3-4 days. 11/14/2021 Cytological Methods 7 Lentil Root tips Green gram root tips
- 8. Cutting the Root tips and Pretreatment When root tips are about ½ to 1 cm long they are cut with sharp blade or scissor. Cut root tips are then subjected to pretreatment For it cut and washed root tips are kept in saturated solution of para di chloro benzene (Pretreating Agent) for 1/2-1-2 hours. Pretreatment softens the tissue and clears cytoplasm and helps in better staining of chromosomes Paradichloro benzene - It is a white crystalline organic substance sparingly soluble in water. Besides PDCB there are several other pre-treating agents. 8 hydroxyquinoline and Oxyquinoline Colchicine Bromonapthalene Cycloheximide Chloral hydrate Coumarin 11/14/2021 Cytological Methods 8
- 9. FIXATION OF ROOT TIPS After Pretreatment, root tips are fixed in any of the fixative agent FIXATIVE AGENTS : 1) Carnoy’s I Fixative (1:3 acetic acid & alcohol) with few drops of Ferric Chloride ( Aceto alcohol) 2) Carnoy’s II Fixative (4:3:1 absolute alcohol & chloroform & acetic acid) 3) Ethyl alcohol 4) Formaldehyde 5) Acetic acid 6) Chromic acid 7) Potassium Dichromate 8) Mercuric chloride Carnoy’s I Fixative is generally used for root tip mitosis . Root tips after pre-treatment are transferred to fixative for about 24 hours. 11/14/2021 Cytological Methods 9
- 10. STORAGE OF ROOT TIPS The fixed root tips can be stored for future use in Preservative 70% Ethyl alcohol acts as good preservative Preparation by using formula – S1V1= S2V2 S- Strength V- Volume 70 cc Absolute alcohol + 30 cc DW = 100 cc 70% Alcohol 77.7 cc 90% Alcohol + 22.3 cc DW = 100 cc 70% Alcohol 11/14/2021 Cytological Methods 10
- 11. STAINING The chromosomes are colour less so stained to make visible under microscope . Different Kinds of staining reagent can be used a) Acetocarmine and b) Aceto orecin ( Prepared solution of 1% 25ml pack available ) c) Feulgen stain d) Toluidine Blue, 0.05% solution available in 100ml & 500ml packs In our laboratory generally Acetocarmine or Aceto- orecin are used to stain root tips, Meiotic slide preparation ,antheridia of Charophytes, algal materials as Rhizoclonium,Oedogonium ,Cladophora, Chaetophorales and other material as Pteridophytes and Gymnosperms . 11/14/2021 Cytological Methods 11
- 12. 1-2 gms of carmine/orecin is dissolved in 100 cc of 45% glacial acetic acid in conical flask. The mixture is then placed on tripod stand with wire gauze, heated for about 15 minutes with the help of burner . While heating the mixture is continuously stirred and then cooled at room temperature Filtered and stored in dark coloured bottle. The preparation is confirmed by blotting paper test. For staining root tips are taken out of preservative Placed in a watch glass. Acetocarmine or Acetoorecin solution is added A few drops of N HCL may be added, which helps in dissolving middle lamella and cells are separated easily. The material is gently heated over spirit lamp for about 5- 15 minutes or in oven at 60 ℃ or hot plate For good staining red hot needle can be inserted into acetocarmine Aceto-orecin solution while heating over spirit lamp By heating root tips become ready for preparing cytological slides PREPARATION OF ACETOCARMINE / ACETO- ORECIN ( 1-2 %) 11/14/2021 Cytological Methods 12
- 13. Preparation of temporary cytological slide by Squash Method Requirements : Watch glass, spirit lamp, Slides, Cover slips ,Needle, Blotting paper,Brush,Tapper ,Scissor, Blade, cutex or wax 45% acetic acid in dropping bottle, Acetocarmine solution 11/14/2021 Cytological Methods 13
- 14. Root cap Mitotic region cell elongation region Root tip On it a stained root tip is placed ,if root tip is long it is made short by cutting with razor blade. For good separation,less material should be taken. In the centre of a clean slide few drops of 45% acetic acid is added . On it a clean coverslip is placed with the help of needle. Care should be taken that bubbles are not introduced while adding coverslip. 11/14/2021 Cytological Methods 14
- 15. The slide with material is slightly heated over spirit lamp. The heated slide is kept in filter paper folds. The slide is then gently pressed with needle tip or gently tapped with flat tapper to spread the heated material. (Tapper must have flat surface, may be a glass rod, brush end etc.) The spread material appears as spray painting 11/14/2021 Cytological Methods 15
- 16. Observation of cytological slide under microscope The slide for observation is placed on stage of compound binolular / trinocular microscope below the objective lens Different stages of division are located under microscope, first at low power magnification, attempts are made to see whether cells of root tip are separated and in metaphase plates chromosomes are separated or not. If not, few drops of 45% Acetic Acid from the sides of coverslip are added, again gently heated over spirit lamp and tapped . Again observed under microscope to locate stages of mitotic division. During observing under microscope ,care should be taken that slide does not dries up, for this 45% AA is regularly added from sides of cover slip. This also helps in removing the excess stain taken by the cytoplasm and enhances visibility of chromosomes ,develops contrast. 11/14/2021 Cytological Methods 16
- 17. Squashing and Temporary sealing of slide After proper separation has been done the slide is placed in filter paper folds on Plain table surface. The slide is pressed or squashed by right hand’s thumb, to bring the material and chromosomes in one plane. Again 45% AA from sides of cover glass is added , left for 2 minutes, the slide is sealed by applying cutex or melted wax on sides of cover glass with help of brush. This temporary slide can observed for 2-3 days or even a week under microscope for making diagrams ,taking photographs or Karyotyping purpose. These slides can be kept in refrigerator. 11/14/2021 Cytological Methods 17
- 18. MAKING THE SLIDE PERMANENT Wax or cutex sealed slide can not be kept for many days due to precipitation of stain. It is therefore essential to make the slide permanent by dehydrating solvents and finally mounting in euperol or DPX. 3 staining troughs are required for turning temporary slide to permanent slide Steps : 2 Alternative Methods 1. Cutex or wax is removed with the help of blade 2. Slide is dipped in first staining trough containing 45% Acetic acid or 70% Alcohol 3. When coverslip separates from the slide, the coverslip and slide both are transferred separately by forceps into second trough containing 45% acetic acid and Butanol in 1:1 Ratio or 90% alcohol 4. Then finally in one minute both slide and coverslip are transferred to third staining trough containing Butanol or Absolute ethyl alcohol 5. Slide and coverslip with the help of forceps are carefully taken out of the trough,placed on filter paper. 11/14/2021 Cytological Methods 18
- 19. Precautions As both slide and cover slip contain stained cytological material ,care should be taken to remember the side on which material is present while transferring them 1st staining trough to second staining trough. Slide and coverslip should only be dipped for one minute because if left for longer time the material may be washed out Microphotographs of well separated metaphase plates and different stages of mitosis can be taken by Nikon Digital Camera and Trinocular microscope with the help of Adaptor or Trinocular microscope with LED or Monitor attachment 11/14/2021 Cytological Methods 19 The slide is mounted with euperol or DPX using a new coverslip and coverslip having cytological material is also mounted using fresh slide with euperol or DPX So from one temporary slide two permanent slides are formed .
- 22. 11/14/2021 Cytological Methods 22 Photographs taken by Nikon digital camera Antheridial filaments of Chara showing prophase of Mitosis
- 23. 11/14/2021 Cytological Methods 23 Observation under low power of Microscope Observation under high power of Microscope
- 24. QUOTATION ATRI SALES CORPORATION SCIENTIFIC CHEMICALS GLASSWARES SURVEYING ENGG., AGRICULTURE & EDUCATIONAL FURNITURE 13 GOVINDPURA,RANJEET,ROAD,AHMEDNAGAR. PH.NO – 0241 -2324672 GST no. 27AWAPA6132H1ZZ Sr no. PARTICULARS UNIT RATE 1 Microscope with LIVE LED screen Make : SPARK x Model : LED08 13.5" led display & 360 degree screen rotation, with one set of eyepiece 10xwf and objectives 5x,10x,40x,100x provided with extra compatible software for image capturing.Optical magnification of 50x to 1000x, Led light source,Coaxial coarse & fine focus system with focusing handwheel, Double layer mechanical stage. per 110000.00 NOTE : GST 18% extra as applicable. Discount 20%. Packing Forwarding & delivery charges extra. For - ATRI SALES CORPORATION TO, Prof (Dr) Ichha Purak. Ref no:- ASC /1255 DATE – 14/11/2019 Your ref no – 11/14/2021 Cytological Methods 24
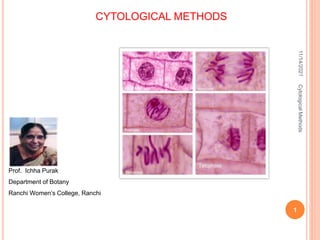
- 25. 11/14/2021 Cytological Methods 25 Different stages of Mitosis in Allium cepa prophase Metaphase Early Anaphase Late Anaphase Telophase
- 27. B. METHODS OF CYTOLOGICAL SLIDE PREPARATION OF CHARA AND NITELLA AND OTHER CHAROPHYTES (almost same as onion root tip mitosis) For cytological studies antheridia/ Globules are selected and stages of mitosis are observed in the antheridial filaments (mostly show synchronous stages) The iron alum aceto-carmine squash technique as proposed by Godward (1948) is employed for cytological preparations. Using pretreatment with PDCB, Fixation in 1:3 aceto alcohol,preservation in 70% C2H5OH,staining with 1.5% Acetocarmine solution Steps are same as Onion Root tip mitosis ,differs only in selection of material,Time of fixation and Staining and Preparation of slide 11/14/2021 Cytological Methods 27
- 28. FIXATION : After pretreatment material is transferred to fixative . As fixative Carnoy’s I is used which is a mixture of one part of glacial acetic acid and three parts of absolute ethyl alcohol with few drops of ferric chloride solution. The materials can be fixed at the site of collection as well as in the laboratory from cultures. The material is left in fixative for 24 hours. Time of Fixation : Material for cytological studies is generally fixed in the morning hours i.e. 9 A M to 11.30 A M. 12.30 P.M. 11/14/2021 Cytological Methods 28
- 29. For staining antheridia, the apical portions of Chara and Nitella containing Nucule and globule are heated in 1.5 % acetocarmine in watch glass or wide test tube for about half an hour. Some stained reproductive branchlets are transferred to 45% acetic acid After that 3-4 antheridia are selected under dissecting microscope with the help of sharp and pointed needle, inserted into antheridia,are picked up and placed in middle of a clean slide with few drops of 45% Acetic acid and then coverslip is gently added. After that slide is gently warmed and is then kept in folds of blotting paper. With the help of tapper the slide is tapped gently so that cells get separated and can be observed for stages of mitotic division. During tapping excess stain of cytoplasm is removed by adding 45% acetic acid from the sides of cover slip repeatedly. Rest of the processes are same as that of Onion root tip mitosis 11/14/2021 Cytological Methods 29
- 30. 11/14/2021 Cytological Methods 30 Picking antheridia of Chara with the help of needles under dissecting microscope
- 33. PLATE SHOWING PROPHASE ,METAPHASE ,ANAPHASE & TELOPHASE ANAPHASE PROPHASE METAPHASE Chara fibrosa 11/14/2021 Cytological Methods 33 METAPHASE 4 CELLS OF ANTHERIDIAL FILAMENT SHOWING METAPHASE Chara fibrosa ( n=56 )
- 34. C.METHOD OF CYTOLOGICAL SLIDE PREPARATION TO STUDY PLANT MEIOSIS (Onion,Rhoeo discolor etc. ) Selection of Material In higher plants meiosis occurs in pollen mother cells which are present in anthers and also in megaspore mother cell (only one ) which lies in ovule. Anthers contain many pollen mother cells (PMC) which can be very easily exposed by bursting anthers. So anthers are excellent source of meiotic material. Flower buds of different sizes are collected in morning hours and are fixed in acetic-alcohol (1:3) for about 24 hours The buds are preserved in 70% alcohol Buds of Rhoeo discolor Buds of Ixora coccinia 11/14/2021 Cytological Methods 34
- 35. For staining buds are dissected to procure anthers After that 1 or 2 anthers are placed in the centre of a clean slide with few drops of 1.5% acetocarmine and a coverglass is placed on the material. The slide is gently heated over a spirit lamp flame . The slide is kept between folds of a blotting paper and a gentle pressure is applied over the cover glass to induce uniform spreading of pollen mother cells (PMCs) and to remove excess stain (Tapping not needed) The cover glass edges are sealed with cutex or wax. s11/14/2021 Cytological Methods 35
- 36. The slides then can be observed under microscope to locate different stages of meiosis I and II As prophase 1 sub stages leptotene,zygotene,pachytene,diplotene and diakinesis (Bivalents ), Anaphase 1 ,Telophase 1 and Metaphase 2 and Anaphase 2 and Telophase 2 . Chromosomal abnormalities can also be located as meiotic anaphasic bridge, translocation heterozygote in Rhoeo discolor anthers, micronuclei formation and chromatin clumping Microphotographs can be taken with the help of trinocular microscope having attachments as adapter and digital camera in of place of eyepiece 11/14/2021 Cytological Methods 36
- 37. 11/14/2021 Cytological Methods 37 Meiosis I Onion Meiosis II Onion Translocation Heterozygote In Rhoeo discolor Tetrads (Meiosis II) in Carica papaya Hordeum vulgare 2n=14, 7 bivalents SOME MEIOTIC STAGES
- 38. REFERENCES Chromosome Techniques Theory and Practice Third Edition Arun Kumar Sharma and Archana Sharma .Aditya Books ,New Delhi College Botany Practical Volume-1 S C Santra, T P Chatterjee and A P Das. New Central Book Agency 1989 Godward, M B E 1948. The iron-alum acetocarmine method for algae. Nature 161 11/14/2021 Cytological Methods 38
