Conjugate A B Pairs - Buffers.ppt
•Download as PPT, PDF•
0 likes•40 views
Conjugate A B Pairs
Report
Share
Report
Share
Recommended
More Related Content
Similar to Conjugate A B Pairs - Buffers.ppt
Similar to Conjugate A B Pairs - Buffers.ppt (20)
ACID BASE BALANCE AND RELATED DISORDERS(Dr.M PRIYANKA)

ACID BASE BALANCE AND RELATED DISORDERS(Dr.M PRIYANKA)
Physicochemical principles and buffers (MUSHTAQ AHMED)

Physicochemical principles and buffers (MUSHTAQ AHMED)
Apchemunit14presentationpart1 120226122440-phpapp02

Apchemunit14presentationpart1 120226122440-phpapp02
Lec9 level4-debiologicalbuffer-130202064553-phpapp01

Lec9 level4-debiologicalbuffer-130202064553-phpapp01
More from Mr. El-Sayed Ramadan
More from Mr. El-Sayed Ramadan (20)
Vocabulary Workshop Level G Unit 1 Presentation.pptx

Vocabulary Workshop Level G Unit 1 Presentation.pptx
Grade 6 unit 1 genre study 1 (narrative nonfiction) target vocabulary

Grade 6 unit 1 genre study 1 (narrative nonfiction) target vocabulary
Cursive Letters Worksheets - Cursive A (elafree.com)

Cursive Letters Worksheets - Cursive A (elafree.com)
Agreement subject and verb, pronoun and antecedent chapter review chapter r...

Agreement subject and verb, pronoun and antecedent chapter review chapter r...
Agreement subject and verb, pronoun and antecedent chapter review

Agreement subject and verb, pronoun and antecedent chapter review
Recently uploaded
KSHARA STURA .pptx---KSHARA KARMA THERAPY (CAUSTIC THERAPY)————IMP.OF KSHARA ...

KSHARA STURA .pptx---KSHARA KARMA THERAPY (CAUSTIC THERAPY)————IMP.OF KSHARA ...M56BOOKSTORE PRODUCT/SERVICE
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR9953056974 Low Rate Call Girls In Saket, Delhi NCR
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
Recently uploaded (20)
TataKelola dan KamSiber Kecerdasan Buatan v022.pdf

TataKelola dan KamSiber Kecerdasan Buatan v022.pdf
Framing an Appropriate Research Question 6b9b26d93da94caf993c038d9efcdedb.pdf

Framing an Appropriate Research Question 6b9b26d93da94caf993c038d9efcdedb.pdf
EPANDING THE CONTENT OF AN OUTLINE using notes.pptx

EPANDING THE CONTENT OF AN OUTLINE using notes.pptx
KSHARA STURA .pptx---KSHARA KARMA THERAPY (CAUSTIC THERAPY)————IMP.OF KSHARA ...

KSHARA STURA .pptx---KSHARA KARMA THERAPY (CAUSTIC THERAPY)————IMP.OF KSHARA ...
Enzyme, Pharmaceutical Aids, Miscellaneous Last Part of Chapter no 5th.pdf

Enzyme, Pharmaceutical Aids, Miscellaneous Last Part of Chapter no 5th.pdf
internship ppt on smartinternz platform as salesforce developer

internship ppt on smartinternz platform as salesforce developer
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Hierarchy of management that covers different levels of management

Hierarchy of management that covers different levels of management
Conjugate A B Pairs - Buffers.ppt
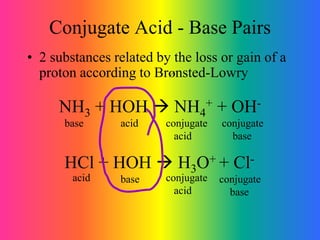
- 1. Conjugate Acid - Base Pairs • 2 substances related by the loss or gain of a proton according to BrØnsted-Lowry NH3 + HOH NH4 + + OH- base conjugate acid acid conjugate base HCl + HOH H3O+ + Cl- base conjugate base acid conjugate acid
- 2. • Acts either as an acid or a base • Water & NH3 . . . . . –Sometimes accepts an H+ –Sometimes donates an H+ Amphiprotic
- 3. • NH4 + + OH- NH3 + H2O • CO3 2- + H2O HCO3 - + OH- • HPO4 -2 + H2O PO4 -3 + H3O+ Identify the conjugate pairs
- 4. What is the conjugate Base for…..? HOH NH3 HCO3 - Acid for…..? HOH NH3 HCO3 - When finding the conj base the given substance acts like an acid and vice versa
- 5. Buffers • A buffer is a mixture of chemicals that make a solution resist a change of pH – pH remains relatively constant when adding an acid or base • A buffer is either a solution of a weak acid and one of its salts or a weak base and one of its salts – The salt cation is only a spectator ion and is not involved in the reaction
- 6. Buffer Capacity • There comes a point when the buffer is “used up” “reached its capacity” • It can no longer take H+ ions (or OH-) out of solution and the pH begins to change
- 7. • This is important when the system is fragile – Bloodstream pH is between 7.3 - 7.5 – If < 6.9 or > 7.7 the person dies • Buffer aids in maintaining homeostasis of the individual • The buffer species in the blood is carbonic acid / hydrogen carbonate ion H2CO3 / HCO3 -
- 8. • If the blood is too basic (alkaline), the reaction decreases the amount of OH- in the bloodstream H2CO3 + OH- HOH + HCO3 - • If excess H+ is in the blood, the rxn decreases it H+ + HCO3 - H2CO3
- 9. Practice • Write an equation to show the addition of an acid and a base to the following buffer systems NH4 + / NH3 CH3COOH / CH3COO- H2PO4 - / HPO4 -
- 10. THE END