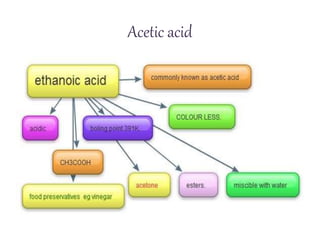
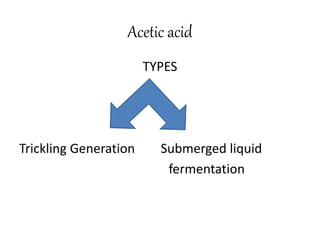
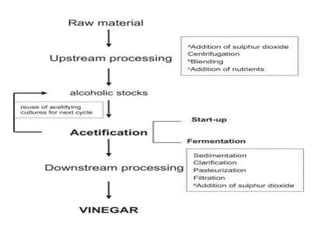
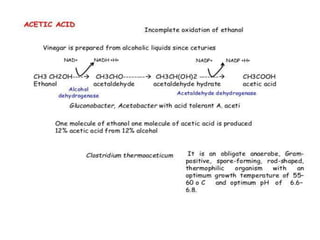
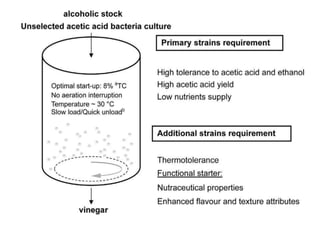
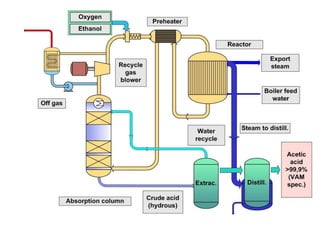
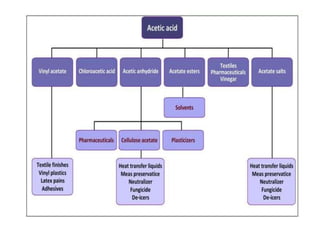
This document discusses the production of acetic acid using microbes. It begins by providing background on acetic acid, noting that it is also known as ethanoic acid and is the main component of vinegar. It then describes acetic acid bacteria, the gram-negative bacteria that oxidize ethanol to produce acetic acid. The document outlines two methods for producing acetic acid - trickling generation using wood chips as a substrate, and submerged liquid fermentation using ethanol and Acetobacter aceti in a silicon tube. It states that submerged liquid fermentation is more efficient, producing a purer form of acetic acid in a shorter time period. Finally, some common uses of acetic acid are mentioned.