Atoms and subatomic particles
•Download as PPTX, PDF•
0 likes•101 views
Science 8
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Bond Formation (Ionic, Covalent, and Metallic Bonding)

Bond Formation (Ionic, Covalent, and Metallic Bonding)
Similar to Atoms and subatomic particles
Similar to Atoms and subatomic particles (20)
Subatomic Particle. proton number atomic number etc

Subatomic Particle. proton number atomic number etc
More from YhanzieCapilitan
More from YhanzieCapilitan (20)
Recently uploaded
Mehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
Recently uploaded (20)
Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...

Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...
Interdisciplinary_Insights_Data_Collection_Methods.pptx

Interdisciplinary_Insights_Data_Collection_Methods.pptx
Sensory_Experience_and_Emotional_Resonance_in_Gabriel_Okaras_The_Piano_and_Th...

Sensory_Experience_and_Emotional_Resonance_in_Gabriel_Okaras_The_Piano_and_Th...
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx

Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx
Salient Features of India constitution especially power and functions

Salient Features of India constitution especially power and functions
ICT Role in 21st Century Education & its Challenges.pptx

ICT Role in 21st Century Education & its Challenges.pptx
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Atoms and subatomic particles
- 3. Atom • is the fundamental unit of element. • Is atom indivisible? • NO!
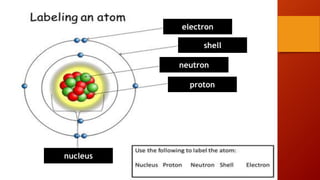
- 4. Structure of Atom Positively charged No charges Negatively charged
- 5. Subatomic particles Charge Mass (g) ~size (m) Coulumb Charge unit electron -1.6022 x 10-19 -1 9.10939 x 10-28 Not known exactly (~1 x 10-18) proton +1.6022 x 10-19 +1 1.67262 x 10-24 1 x 10-15 neutron 0 0 1.67493 x 10-24 1 x 10-15
- 6. • Elements differ in the number of protons, electrons, and neutrons. • Identity of an element is based on the number of protons equates to the atomic number. EXAMPLE: Atomic number = number of protons For neutral atom: Number of protons = number of electrons Number of electrons = 11
- 7. Elements are generally represented by a nuclide symbol: • Mass number = number of protons + number of neutrons • Number of neutrons = mass number – atomic number N = A - Z
- 8. What is the difference between mass number and atomic number? ELEMENTS Na K Mg Al N Mass number 23 24 14 Atomic Mass Identify the atomic mass and mass number of the following elements:
- 9. ELEMENTS Na K Mg Al N Mass number 23 39 24 27 14 Atomic Mass 22.9898 39.0983 24.305 26.9815 14.0067