Pharmaceutical Freeze Drying Technology Conference
•
2 likes•57 views
This document summarizes a pharmaceutical freeze drying technology conference that took place in London in 2018. The conference featured presentations on optimizing freeze drying cycles through both established and new methods. Specific topics included using vial impedance spectroscopy for cycle development, evaluating stability of freeze dried vaccines through water activity measurement and modeling, and comparing quality-by-design strategies for spray drying versus lyophilization. Over 60 industry professionals from Europe and various other countries attended and provided positive feedback on the high quality presentations and selection of relevant topics covered over the two-day event.
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (12)
MDC Connects: The challenges of accessing clinical samples

MDC Connects: The challenges of accessing clinical samples
ChemBio Hub engagement in Oxford university - Lessons learnt

ChemBio Hub engagement in Oxford university - Lessons learnt
Global Discovery & Developement Innovation Forum 29016

Global Discovery & Developement Innovation Forum 29016
Similar to Pharmaceutical Freeze Drying Technology Conference
Similar to Pharmaceutical Freeze Drying Technology Conference (20)
SMi Group's 6th annual Pharmaceutical Freeze Drying Technology 2018 conference

SMi Group's 6th annual Pharmaceutical Freeze Drying Technology 2018 conference
2016 05 technology roadmapping update for u mass (1)

2016 05 technology roadmapping update for u mass (1)
SMi Group's 4th annual Lyophilisation Europe 2016 conference

SMi Group's 4th annual Lyophilisation Europe 2016 conference
The study of scope and implementation of lean aspects

The study of scope and implementation of lean aspects
The study of scope and implementation of lean aspects

The study of scope and implementation of lean aspects
Maize - Presentation for Discussion with Donors and Partners - June 2013

Maize - Presentation for Discussion with Donors and Partners - June 2013
Maize - Presentation for Discussion with Donors and Partners - June 2013

Maize - Presentation for Discussion with Donors and Partners - June 2013
SMi Group's Pharmaceutical Freeze Drying Technology 2017 

SMi Group's Pharmaceutical Freeze Drying Technology 2017
Waste Plastic Pyrolysis Manufacturing Plant Project Report.pptx

Waste Plastic Pyrolysis Manufacturing Plant Project Report.pptx
CBINet 11 Life Sciences Finance and Accounting Congress 2015

CBINet 11 Life Sciences Finance and Accounting Congress 2015
Plastic Pyrolysis Manufacturing Plant Project Report.pptx

Plastic Pyrolysis Manufacturing Plant Project Report.pptx
How Sustainability management and the GRI structure can benefit your company

How Sustainability management and the GRI structure can benefit your company
Recently uploaded
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptxNandakishor Bhaurao Deshmukh
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...Nistarini College, Purulia (W.B) India
Recently uploaded (20)
Neurodevelopmental disorders according to the dsm 5 tr

Neurodevelopmental disorders according to the dsm 5 tr
Scheme-of-Work-Science-Stage-4 cambridge science.docx

Scheme-of-Work-Science-Stage-4 cambridge science.docx
Call Us ≽ 9953322196 ≼ Call Girls In Mukherjee Nagar(Delhi) |

Call Us ≽ 9953322196 ≼ Call Girls In Mukherjee Nagar(Delhi) |
insect anatomy and insect body wall and their physiology

insect anatomy and insect body wall and their physiology
LIGHT-PHENOMENA-BY-CABUALDIONALDOPANOGANCADIENTE-CONDEZA (1).pptx

LIGHT-PHENOMENA-BY-CABUALDIONALDOPANOGANCADIENTE-CONDEZA (1).pptx
Call Girls In Nihal Vihar Delhi ❤️8860477959 Looking Escorts In 24/7 Delhi NCR

Call Girls In Nihal Vihar Delhi ❤️8860477959 Looking Escorts In 24/7 Delhi NCR
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx
Engler and Prantl system of classification in plant taxonomy

Engler and Prantl system of classification in plant taxonomy
Call Girls in Hauz Khas Delhi 💯Call Us 🔝9953322196🔝 💯Escort.

Call Girls in Hauz Khas Delhi 💯Call Us 🔝9953322196🔝 💯Escort.
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...
Pharmaceutical Freeze Drying Technology Conference
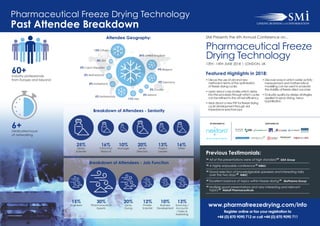
- 1. Pharmaceutical Freeze Drying Technology Past Attendee Breakdown Featured Highlights in 2018: • Discuss the use of old and new methods in terms of the optimisation of freeze drying cycles • Learn about case studies which delve into the processes through which cycles can be refined to the utmost efficiency • Hear about a new PAT for freeze drying cycle development through vial impedance spectroscopy • Discover ways in which water activity measurement and mathematical modelling can be used to evaluate the stability of freeze dried vaccines • Evaluate quality-by-design strategies, applied to spray drying, versus lyophilisation. SMi Presents the 6th Annual Conference on... 13TH - 14TH JUNE 2018 | LONDON, UK Pharmaceutical Freeze Drying Technology 60+Industry professionals from Europe and beyond SPONSORED BY SUPPORTED BY 6+Dedicated hours of networking Attendee Geography: Breakdown of Attendees - Seniority 25% Senior Scientist 16% CEO/VPs/ Head of 10% Manager 20% Senior Directors 13% Project Leader 16% Other Breakdown of Attendees - Job Function 15% Engineers 30% Pharmaceutical Experts 20% Spray Drying 12% Protein Scientist 10% Business Development 13% Sourcing / Accounts / Sales & Marketing www.pharmafreezedrying.com/info Register online or fax your registration to +44 (0) 870 9090 712 or call +44 (0) 870 9090 711 Previous Testimonials: “ All of the presentations were of high standard ” GEA Group “ A highly enjoyable conference ” NIBSC “ Good selection of knowledgeable speakers and interesting talks over the two days ” NIBSC “ Excellent balance of topics within freeze-drying ” BioPharma Group “ Multiple good presentations and very interesting and relevant topics ” Abbott Pharmaceuticals 36%36% United KingdomUnited Kingdom 11% Italy 3%3% Ireland 3% USA 2% Czech RepublicCzech Republic 5% NetherlandsNetherlands 4% Switzerland 9% Belgium 5% NetherlandsNetherlands 3% CroatiaCroatia 9% Germany 13% OthersOthers