Pharma manufacturing facility maintenance
•
0 likes•137 views
Using sound science and risk based approaches is indispensable in prediction and maintaining certainty within emerging regulatory frames.
Report
Share
Report
Share
Download to read offline
Recommended
EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...

EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...A.T.E. Private Limited
WHO Guidance on Preparation of Laboratory Information File

WHO Guidance on Preparation of Laboratory Information FileGMP EDUCATION : Not for Profit Organization
Who Guidance on Process Validation for Non Serile Pharmaceuticals

Who Guidance on Process Validation for Non Serile PharmaceuticalsGMP EDUCATION : Not for Profit Organization
More Related Content
What's hot
EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...

EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...A.T.E. Private Limited
WHO Guidance on Preparation of Laboratory Information File

WHO Guidance on Preparation of Laboratory Information FileGMP EDUCATION : Not for Profit Organization
Who Guidance on Process Validation for Non Serile Pharmaceuticals

Who Guidance on Process Validation for Non Serile PharmaceuticalsGMP EDUCATION : Not for Profit Organization
What's hot (20)
EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...

EnviroCOLA® – A digital transformation of wastewater treatment and recycling ...
How to Prepare for an FDA Inspection and Respond to FDA 483's / Warning Letters

How to Prepare for an FDA Inspection and Respond to FDA 483's / Warning Letters
WHO Guidance on Preparation of Laboratory Information File

WHO Guidance on Preparation of Laboratory Information File
Item 3. Internal quality control - Quality Control Charts

Item 3. Internal quality control - Quality Control Charts
New hire orientation good documentation practice presentation

New hire orientation good documentation practice presentation
Who Guidance on Process Validation for Non Serile Pharmaceuticals

Who Guidance on Process Validation for Non Serile Pharmaceuticals
Similar to Pharma manufacturing facility maintenance
US FDA Process Validation Stage 3: Continued Process Verification

US FDA Process Validation Stage 3: Continued Process VerificationGMP EDUCATION : Not for Profit Organization
Similar to Pharma manufacturing facility maintenance (20)
Data integrity Presentation@GCC Regulatory Summit April-2017

Data integrity Presentation@GCC Regulatory Summit April-2017
US FDA Process Validation Stage 3: Continued Process Verification

US FDA Process Validation Stage 3: Continued Process Verification
More from Obaid Ali / Roohi B. Obaid
More from Obaid Ali / Roohi B. Obaid (20)
A Strategic Talk on Biologicals & Biotechnological Products

A Strategic Talk on Biologicals & Biotechnological Products
Centre for Quality Sciences-Information File-2024.pdf

Centre for Quality Sciences-Information File-2024.pdf
Demonstrate your QMS during Inspection-30 Sep 2023.pdf

Demonstrate your QMS during Inspection-30 Sep 2023.pdf
Track, Trace & Understand Science of Life from Real World Evidence.pdf

Track, Trace & Understand Science of Life from Real World Evidence.pdf
120th Training Session - Unfortunate Wave of DEG Tragedy.pdf

120th Training Session - Unfortunate Wave of DEG Tragedy.pdf
Recently uploaded
Rohini Sector 16 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 16 Call Girls Delhi 9999965857 @Sabina Saikh No AdvanceCall Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Call Girls Service Tilak Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SERVICE

Call Girls Service Tilak Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SERVICECall Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
sauth delhi call girls in Defence Colony🔝 9953056974 🔝 escort Service

sauth delhi call girls in Defence Colony🔝 9953056974 🔝 escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (17)
Rohini Sector 16 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 16 Call Girls Delhi 9999965857 @Sabina Saikh No Advance
Call Girls Service Tilak Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SERVICE

Call Girls Service Tilak Nagar @9999965857 Delhi 🫦 No Advance VVIP 🍎 SERVICE
CALL ON ➥8923113531 🔝Call Girls Charbagh Lucknow best sexual service

CALL ON ➥8923113531 🔝Call Girls Charbagh Lucknow best sexual service
Introduction to LPC - Facility Design And Re-Engineering

Introduction to LPC - Facility Design And Re-Engineering
crisiscommunication-presentation in crisis management.pptx

crisiscommunication-presentation in crisis management.pptx
sauth delhi call girls in Defence Colony🔝 9953056974 🔝 escort Service

sauth delhi call girls in Defence Colony🔝 9953056974 🔝 escort Service
LPC Operations Review PowerPoint | Operations Review

LPC Operations Review PowerPoint | Operations Review
Becoming an Inclusive Leader - Bernadette Thompson

Becoming an Inclusive Leader - Bernadette Thompson
Pooja Mehta 9167673311, Trusted Call Girls In NAVI MUMBAI Cash On Payment , V...

Pooja Mehta 9167673311, Trusted Call Girls In NAVI MUMBAI Cash On Payment , V...
VIP Kolkata Call Girl Rajarhat 👉 8250192130 Available With Room

VIP Kolkata Call Girl Rajarhat 👉 8250192130 Available With Room
LPC Warehouse Management System For Clients In The Business Sector

LPC Warehouse Management System For Clients In The Business Sector
ANIn Gurugram April 2024 |Can Agile and AI work together? by Pramodkumar Shri...

ANIn Gurugram April 2024 |Can Agile and AI work together? by Pramodkumar Shri...
Pharma manufacturing facility maintenance
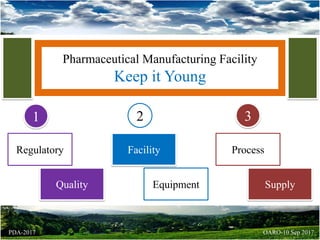
- 1. Pharmaceutical Manufacturing Facility Keep it Young Regulatory Quality Facility Equipment Process Supply 1 2 3 OARO-10 Sep 2017PDA-2017
- 2. Pharmaceutical Manufacturing Facility Keep it Young Regulatory Quality 1 OARO-10 Sep 2017PDA-2017 Review & monitor • Recalls & Lot Acceptance Rates, Trend data & keep an eye on • Increase in Deviations, OOS, CAPAs, complaints, audit observations Trace and track increase in • Change in Process/ equipment/ facility OOS: Out of Specification (Results), CAPA: Corrective & Preventive Action
- 3. Pharmaceutical Manufacturing Facility Keep it Young Facility Equipment 2 OARO-10 Sep 2017PDA-2017 WHEN Finishing attracts Parts are not available Technology is obsolete INCREASE Maintenance & Downtime Contamination issues Environmental Monitoring
- 4. Pharmaceutical Manufacturing Facility Keep it Young Process Supply 3 OARO-10 Sep 2017PDA-2017 Materials • Supplier is lethargic or losing interest Capacity • Increase in market demand Change • ↓ Process yield • ↑ deviations • ↑ Downtime • ↑ Failures • ↑ Variability • ↑ Rework
