CH%206-3%20SN2%20Reaction-III.pptx
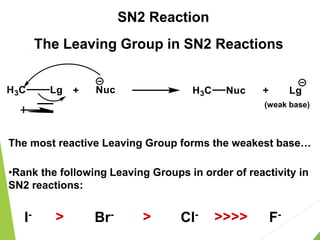
- 1. The Leaving Group in SN2 Reactions The most reactive Leaving Group forms the weakest base… I- Br- Cl- F- <<<< < < •Rank the following Leaving Groups in order of reactivity in SN2 reactions: (weak base) + SN2 Reaction
- 2. The Nucleophile in SN2 Reactions •The chemical reactivity of the nucleophile is referred to as its “NUCLEOPHILICITY”. Several factors effect the reactivity of the nucleophile. (1) Solvent effects the rate of SN2 reactions. The typical solvents for SN2 reactions are “polar aprotic” solvents (DMSO, acetone, DMF): •Polar “aprotic” solvents bind the positive cation, leaving a “naked anion” which is a good nucleophile. •For NaCl in acetone:
- 3. “Nucleophilicity” in SN2 Reactions (Effect of the nucleophile on reaction rate) F- NH2 - HO- •Rank the following in order of their Nucleophilicity (1 = most…3 = least): Cl- Br- I- (2) Base Strength. The strongest (more reactive) nucleophile is typically the stronger base. 2 1 3 3 1 2 1 2 3
- 4. “Nucleophilicity” in SN2 Reactions H2O H2S H2Se •EXCEPTION: For neutral molecules from same column, Nucleophilicity depends on the size of the central atom. The larger atom is more nucleophilic. The trend is opposite the expected base strength! < <
- 5. “Nucleophilicity” in SN2 Reactions •Large “bulky” bases are poor nucleophiles…..WHY? (3) Steric effects (nucleophile size). Which is the more reactive nucleophile: CH 3 C CH 3 O CH 3 CH 3 CH 2 O Better nucleophile
- 6. Re-cap: The SN2 Substitution Reaction •A bimolecular, concerted mechanism - R Lg N uc R R - •Nucleophile: best Nuc is usually a small, strong base, or large neutral atom, in polar-aprotic solvent (acetone). •Energy Diagram: minimum of one step (one T.S.) •“Pentavalent Carbon” in the T.S.; crowded T.S. •Nucleophile “attacks” 180o opposite the Leaving Group. •Stereospecific reaction; inversion of configuration •Substrate structure: methyl > 1o > 2o >>> 3o (NR by SN2) •Leaving groups: best leaving groups form a weak base