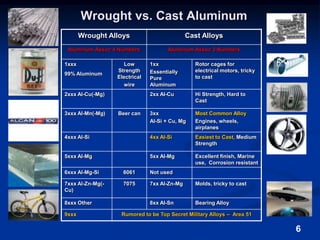
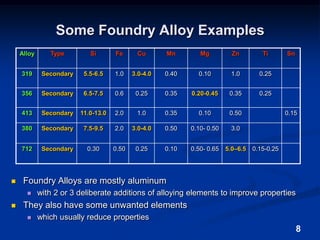
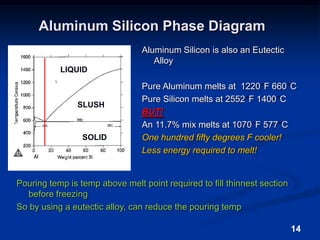
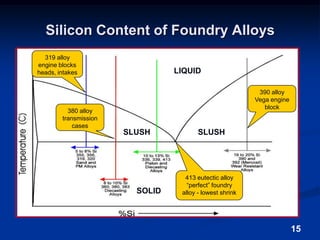
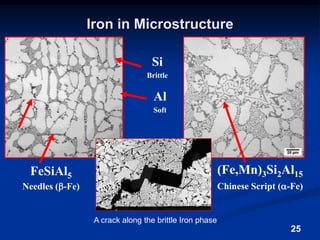
This document discusses the basics of aluminum alloys used in metalcasting. It explains that cast alloys have different compositions than wrought alloys and are designed specifically for casting. Common alloying elements like silicon, magnesium, copper, and zinc are added to aluminum to improve properties like fluidity, strength, and corrosion resistance. Unwanted elements like iron can make alloys more brittle. Gases like hydrogen and oxygen must also be controlled to prevent defects. Grain refiners and modifiers can further improve mechanical properties of cast aluminum alloys.