CML Chronic Myeloid Leukemia.pdf
•
0 likes•28 views
Chronic Myeloid Leukemia-Causing Event—How the BCR-ABL1 Cancer-Causing Gene (Oncogene) Is Formed. Mr.Naveed Shaikh, Quality Manager Molecular Laboratory, Divinations Lifecare Pvt Ltd Pune
Report
Share
Report
Share
Download to read offline
Recommended
all u want to knew about CMLChronic Myloid Leukemia overview (CML)

Chronic Myloid Leukemia overview (CML)National Cancer Institute, Cairo University - Children's Cancer Hospital - Egypt 57357
Recommended
all u want to knew about CMLChronic Myloid Leukemia overview (CML)

Chronic Myloid Leukemia overview (CML)National Cancer Institute, Cairo University - Children's Cancer Hospital - Egypt 57357
More Related Content
Similar to CML Chronic Myeloid Leukemia.pdf
Similar to CML Chronic Myeloid Leukemia.pdf (20)
New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...

New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...
New Directions in Targeted Therapeutic Approaches for Older Adults with Mantl...

New Directions in Targeted Therapeutic Approaches for Older Adults with Mantl...
Recently uploaded
Test Bank -Medical-Surgical Nursing Concepts for Interprofessional Collaborative Care 11th edition (All chapters complete 1 - 74, Question and Answers with Rationales).Test Bank -Medical-Surgical Nursing Concepts for Interprofessional Collaborat...

Test Bank -Medical-Surgical Nursing Concepts for Interprofessional Collaborat...rightmanforbloodline
TEST BANK For Little and Falace's Dental Management of the Medically Compromised Patient, 10th Edition by Craig Miller, Verified Chapters 1 - 30, Complete Newest VersionTEST BANK For Little and Falace's Dental Management of the Medically Compromi...

TEST BANK For Little and Falace's Dental Management of the Medically Compromi...rightmanforbloodline
Mtp kit in kuwait௹+918133066128....) @abortion pills for sale in Kuwait City ✒Abortion CLINIC In Kuwait ?Kuwait pills +918133066128௵) safe Abortion Pills for sale in Salmiya, Kuwait city,Farwaniya-cytotec pills for sale in Kuwait city. Kuwait pills +918133066128WHERE I CAN BUY ABORTION PILLS IN KUWAIT, CYTOTEC 200MG PILLS AVAILABLE IN KUWAIT, MIFEPRISTONE & MISOPROSTOL MTP KIT FOR SALE IN KUWAIT. Whatsapp:+Abortion Pills For Sale In Mahboula-abortion pills in Mahboula-abortion pills in Kuwait City- .Kuwait pills +918133066128)))abortion pills for sale in Mahboula …Mtp Kit On Sale Kuwait pills +918133066128mifepristone Tablets available in Kuwait?Zahra Kuwait pills +918133066128Buy Abortion Pills Cytotec Misoprostol 200mcg Pills Brances and now offering services in Sharjah, Abu Dhabi, Dubai, **))))Abortion Pills For Sale In Ras Al-Khaimah(((online Cytotec Available In Al Madam))) Cytotec Available In muscat, Cytotec 200 Mcg In Zayed City, hatta,Cytotec Pills௵+ __}Kuwait pills +918133066128}— ABORTION IN UAE (DUBAI, SHARJAH, AJMAN, UMM AL QUWAIN, ...UAE-ABORTION PILLS AVAILABLE IN DUBAI/ABUDHABI-where can i buy abortion pillsCytotec Pills௵+ __}Kuwait pills +918133066128}}}/Where can I buy abortion pills in KUWAIT , KUWAIT CITY, HAWALLY, KUWAIT, AL JAHRA, MANGAF , AHMADI, FAHAHEEL, In KUWAIT ... pills for sale in dubai mall and where anyone can buy abortion pills in Abu Dhabi, Dubai, Sharjah, Ajman, Umm Al Quwain, Ras Al Khaimah ... Abortion pills in Dubai, Abu Dhabi, Sharjah, Ajman, Fujairah, Ras Al Khaimah, Umm Al Quwain…Buy Mifepristone and Misoprostol Cytotec , Mtp KitABORTION PILLS _ABORTION PILLS FOR SALE IN ABU DHABI, DUBAI, AJMAN, FUJUIRAH, RAS AL KHAIMAH, SHARJAH & UMM AL QUWAIN, UAE ❤ Medical Abortion pills in ... ABU DHABI, ABORTION PILLS FOR SALE ----- Dubai, Sharjah, Abu dhabi, Ajman, Alain, Fujairah, Ras Al Khaimah FUJAIRAH, AL AIN, RAS AL KHAIMAMedical Abortion pills in Dubai, Abu Dhabi, Sharjah, Al Ain, Ajman, RAK City, Ras Al Khaimah, Fujairah, Dubai, Qatar, Bahrain, Saudi Arabia, Oman, ...Where I Can Buy Abortion Pills In Al ain where can i buy abortion pills in #Dubai, Exclusive Abortion pills for sale in Dubai ... Abortion Pills For Sale In Rak City, in Doha, Kuwait.௵ Kuwait pills +918133066128₩ Abortion Pills For Sale In Doha, Kuwait,CYTOTEC PILLS AVAILABLE Abortion in Doha, ꧁ @ ꧂ ☆ Abortion Pills For Sale In Ivory park,Rabie Ridge,Phomolong. ] Abortion Pills For Sale In Ivory Park, Abortion Pills+918133066128In Ivory Park, Abortion Clinic In Ivory Park,Termination Pills In Ivory Park,. *)][(Abortion Pills For Sale In Tembisa Winnie Mandela Ivory Park Ebony Park Esangweni Oakmoor Swazi Inn Whats'app...In Ra al Khaimah,safe termination pills for sale in Ras Al Khaimah. | Dubai.. @Kuwait pills +918133066128Abortion Pills For Sale In Kuwait, Buy Cytotec Pills In Kuwait.Cytotec Pills௵ __}Kuwait pills +918133066128}}}/Where cAbortion pills Buy Farwaniya (+918133066128) Cytotec 200mg tablets Al AHMEDI

Abortion pills Buy Farwaniya (+918133066128) Cytotec 200mg tablets Al AHMEDIAbortion pills in Kuwait Cytotec pills in Kuwait
Spesialis Penggugur Kandungan 081466799220 Jual Obat Aborsi Cytotec Gastrul Peluntur Janin 1 2 3 4 5 6 7 Bulan, Metode Cara Aborsi Menggugurkan Kandungan Aman Cepat & Tuntas 100% Berhasil, Obat Aborsi, Obat Penggugur Kandungan, Jual Obat Misotrol, Cytotec, dan Cyprostol, Cyrux Pendorong Janin, Pil Aborsi, Pil Cytotec Aborsi, Jual Obat Penggugur Kandungan, Klinik Konsultasi, Obat Telat Bulan, Obat Pelancar Haid, Jual Cytotec Asli, Cara Menggugurkan Kandungan, Penjual Obat Aborsi Terpercaya, Obat Aborsi Ampuh, Obat Aborsi Manjur, Obat Aborsi Tuntas, Obat Aborsi Aman, Obat Aborsi Asli, Obat Aborsi Bergaransi, Obat Aborsi 6 Bulan, Obat Aborsi 5 Bulan, Obat Aborsi 4 Bulan, Obat Aborsi 3 Bulan, Obat Aborsi 2 Bulan, Obat Aborsi 1 Bulan, Apotik Jual Obat Aborsi, Apotik Jual Cytotec, Cara Penggunaan Obat Aborsi Cytotec, Klinik Aborsi, Pil Penggugur Kandungan, Cara Pakai Cytotec, Cara Pemakian Cyrux Pendorong Janin
KAMI SIAP BANTU ANDA YANG INGIN MENGGUGURKAN KANDUNG
1.TERJAMIN BERHAS
2.TERJAMIN TUNTA
3.TERJAMIN AMA
4.TERJAMIN TANPA EFEK SAMPIN
5.TERJAMIN 100% TERBUKT
6.TERJAMIN REAL & TERPERCAY
NB: HIMBAUHAN BAGI KONSUMEN JANGAN TERPENGARUH DENGAN PENAWARAN HARGA MURAH. KARENA OBAT ASLI TIDAK DI JUAL DENGAN HARGA MURAH. PASTIKAN ANDA MEMBELI DI TEMPAT KAMI DI JAMIN ASLI & BERGARANSI CUKUP 1X PEMAKIAN JANIN LANGSUNG GUGUR 100% AMAN TANPA EFEK SAMPI
Cytotec ® adalah nama merek dagang Misoprostol yang di produksi oleh PFIZER (searle) yang di setujui US FDA. Obat cytotec botol digunakan untuk mengobati masalah yang berkaitan dengan NSAID (Non Steroidal Anti Inflammatory Drugs) atau obat anti inflamasi non steroid (AINS). Selain itu, proses cytotec menyebabkan kontraksi otot rahim yang mengarah pada penggunaannya sebagai obat penggugur kandungan untuk menginduksi rahim gugur secara bertahap dan bersih hingga TUNTA
Penjual Obat Aborsi Penggugur Kandungan Cytotec 400 mg dan Cyrux Pendorong Janin, Kami akan membantu serta memberikan panduan cara pakai / menggunakan obat cytotec mcg sampai gugur dan ama
Obat Aborsi Penggugur Kandungan Cytotec 400 mg dan Cyru
Beli Obat Cytotec Tanpa Resep Dokter hanya Pil Aborsi Asli untuk kehamilan 1 Bulan, 2 Bulan, 3 Bulan, 4 Bulan, 5 Bulan, 6 Bulan, 7 Bulan Jika Kehamilan Anda Kuat atau Bandel susah digugurkan Perlu menggunakan dosis tinggi khusus janin kuat bisa menggunakan 20 Butir Cytotec / Paket Tuntas 8 Bulan
Cytotec 400 mcg: Fungsi, Dosis Tinggi, Aturan Pakai, dan 100% Berhas
Jual Obat Aborsi Cytotec 400 Mcg Asli. Sedia Obat Penggugur Kandungan Gastrul dan Cytotec 400 mcg Asli Di Apotek
Obat Cytotec Pfizer 400 mcg adalah obat aborsi yang mengandung zat aktif misoprostol. Kandungan cytotec asli ini dapat menurunkan kadar asam lambung yang berlebihan dan mencegah kerusakan lambung yang disebabkan obat OAINS (obat antiinflamasi nonsteroid)
Fungsi Cytotec 400 m
Obat Cytotec Asli bekerja untuk haid yang tidak teratur atau untuk mengatur yang tidak diinginkanOBAT PENGGUGUR KANDUNGAN 081466799220 PIL ABORSI CYTOTEC PELUNTUR JANIN

OBAT PENGGUGUR KANDUNGAN 081466799220 PIL ABORSI CYTOTEC PELUNTUR JANINJUAL OBAT GASTRUL MISOPROSTOL 081466799220 PIL ABORSI CYTOTEC 1 2 3 4 5 6 7 BULAN TERPERCAYA
CALL GIRLS IN GOA & ESCORTS SERVICE 9316020077 Door Step Delivery We Offering You 100% Genuine Completed Body And Mind Relaxation With Happy Ending ServiCe Done By Most Attractive Charming Soft Spoken Bold Beautiful Full Cooperative Independent Escort Girls ServiCe In All Star Hotel And Home ServiCe In All Over North Goa-Baga , Calangute , Anjuna , Candolim , Arpora , Vagator , Morjim , Arambol , Mandrem , Mapusa , Siolim , Porvorim , Panaji , Miramar , Dona Paula ,Etc. Goa Also …,
I Have Extremely Beautiful Broad Minded Cute Sexy & Hot Call Girls and Escorts, We Are Located in 3* 4* 5* Hotels in GOA. Safe & Secure High Class Services Affordable Rate 100% Satisfaction, Unlimited Enjoyment. Any Time for Model/Teens Escort in GOA High Class luxury and Premium Escorts ServiCe.
★ CALL US High Class Luxury and Premium Escorts ServiCe We Provide Well Educated, Royal Class Female, High-Class Escorts Offering a Top High Class Escorts Service In the & Several Nearby All Places Of .
★ Get The High Profile, Bollywood Queens , Well Educated , Good Looking , Full Cooperative Model Services. You Can See Me at My Comfortable Hotels or I Can Visit You In hotel Our Service Available IN All SERVICE, 3/4/5 STAR HOTEL , In Call /Out Call Services.24 hrs ,
★ To Enjoy With Hot and Sexy Girls .
★ We Are Providing :-
• Models
• Vip Models
• Russian Models
• Foreigner Models
• TV Actress and Celebrities
• Receptionist
• Air Hostess
• Call Center Working Girls/Women
• Hi-Tech Co. Girls/Women
• Housewife
• Collage Going Girls.
• Travelling Escorts.
• Ramp-Models
• Foreigner And Many More.. Incall & Outcall Available…
• INDEPENDENT GIRLS / HOUSE WIFES
No Advance 931~602~0077 Goa ✂️ Call Girl , Indian Call Girl Goa For Full nig...

No Advance 931~602~0077 Goa ✂️ Call Girl , Indian Call Girl Goa For Full nig...Real Sex Provide In Goa
PEMESANAN OBAT ASLI : +6287776558899
Cara Menggugurkan Kandungan usia 1 , 2 , bulan - obat penggugur janin - cara aborsi kandungan - obat penggugur kandungan 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 bulan - bagaimana cara menggugurkan kandungan - tips Cara aborsi kandungan - trik Cara menggugurkan janin - Cara aman bagi ibu menyusui menggugurkan kandungan - klinik apotek jual obat penggugur kandungan - jamu PENGGUGUR KANDUNGAN - WAJIB TAU CARA ABORSI JANIN - GUGURKAN KANDUNGAN AMAN TANPA KURET - CARA Menggugurkan Kandungan tanpa efek samping - rekomendasi dokter obat herbal penggugur kandungan - ABORSI JANIN - aborsi kandungan - jamu herbal Penggugur kandungan - cara Menggugurkan Kandungan yang cacat - tata cara Menggugurkan Kandungan - obat penggugur kandungan di apotik kimia Farma - obat telat datang bulan - obat penggugur kandungan tuntas - obat penggugur kandungan alami - klinik aborsi janin gugurkan kandungan - ©Cytotec ™misoprostol BPOM - OBAT PENGGUGUR KANDUNGAN ®CYTOTEC - aborsi janin dengan pil ©Cytotec - ®Cytotec misoprostol® BPOM 100% - penjual obat penggugur kandungan asli - klinik jual obat aborsi janin - obat penggugur kandungan di klinik k-24 || obat penggugur ™Cytotec di apotek umum || ®CYTOTEC ASLI || obat ©Cytotec yang asli 200mcg || obat penggugur ASLI || pil Cytotec© tablet || cara gugurin kandungan || jual ®Cytotec 200mcg || dokter gugurkan kandungan || cara menggugurkan kandungan dengan cepat selesai dalam 24 jam secara alami buah buahan || usia kandungan 1_2 3_4 5_6 7_8 bulan masih bisa di gugurkan || obat penggugur kandungan ®cytotec dan gastrul || cara gugurkan pembuahan janin secara alami dan cepat || gugurkan kandungan || gugurin janin || cara Menggugurkan janin di luar nikah || contoh aborsi janin yang benar || contoh obat penggugur kandungan asli || contoh cara Menggugurkan Kandungan yang benar || telat haid || obat telat haid || Cara Alami gugurkan kehamilan || obat telat menstruasi || cara Menggugurkan janin anak haram || cara aborsi menggugurkan janin yang tidak berkembang || gugurkan kandungan dengan obat ©Cytotec || obat penggugur kandungan ™Cytotec 100% original || HARGA obat penggugur kandungan || obat telat haid 1 bulan || obat telat menstruasi 1-2 3-4 5-6 7-8 BULAN || obat telat datang bulan || cara Menggugurkan janin 1 bulan || cara Menggugurkan Kandungan yang masih 2 bulan || cara Menggugurkan Kandungan yang masih hitungan Minggu || cara Menggugurkan Kandungan yang masih usia 3 bulan || cara Menggugurkan usia kandungan 4 bulan || cara Menggugurkan janin usia 5 bulan || cara Menggugurkan kehamilan 6 Bulan
________&&&_________&&&_____________&&&_________&&&&____________
Cara Menggugurkan Kandungan Usia Janin 1 | 7 | 8 Bulan Dengan Cepat Dalam Hitungan Jam Secara Alami, Kami Siap Meneriman Pesanan Ke Seluruh Indonesia, Melputi: Ambon, Banda Aceh, Bandung, Banjarbaru, Batam, Bau-Bau, Bengkulu, Binjai, Blitar, Bontang, Cilegon, Cirebon, Depok, Gorontalo, Jakarta, Jayapura, Kendari, Kota Mobagu, Kupang, LhokseumaweObat Penggugur Kandungan Cytotec Dan Gastrul Harga Indomaret

Obat Penggugur Kandungan Cytotec Dan Gastrul Harga IndomaretCara Menggugurkan Kandungan 087776558899
Recently uploaded (20)
TIME FOR ACTION: MAY 2024 Securing A Strong Nursing Workforce for North Carolina

TIME FOR ACTION: MAY 2024 Securing A Strong Nursing Workforce for North Carolina
Cash Payment 😋 +9316020077 Goa Call Girl No Advance *Full Service

Cash Payment 😋 +9316020077 Goa Call Girl No Advance *Full Service
Test Bank -Medical-Surgical Nursing Concepts for Interprofessional Collaborat...

Test Bank -Medical-Surgical Nursing Concepts for Interprofessional Collaborat...
Test bank for community public health nursing evidence for practice 4TH editi...

Test bank for community public health nursing evidence for practice 4TH editi...
Making change happen: learning from "positive deviancts"

Making change happen: learning from "positive deviancts"
TEST BANK For Little and Falace's Dental Management of the Medically Compromi...

TEST BANK For Little and Falace's Dental Management of the Medically Compromi...
Pathways to Equality: The Role of Men and Women in Gender Equity

Pathways to Equality: The Role of Men and Women in Gender Equity
Abortion pills Buy Farwaniya (+918133066128) Cytotec 200mg tablets Al AHMEDI

Abortion pills Buy Farwaniya (+918133066128) Cytotec 200mg tablets Al AHMEDI
Goa Call Girl 931~602~0077 Call ✂️ Girl Service Vip Top Model Safe

Goa Call Girl 931~602~0077 Call ✂️ Girl Service Vip Top Model Safe
❤️ Chandigarh Call Girls Service ☎️99158-51334☎️ Escort service in Chandigarh...

❤️ Chandigarh Call Girls Service ☎️99158-51334☎️ Escort service in Chandigarh...
OBAT PENGGUGUR KANDUNGAN 081466799220 PIL ABORSI CYTOTEC PELUNTUR JANIN

OBAT PENGGUGUR KANDUNGAN 081466799220 PIL ABORSI CYTOTEC PELUNTUR JANIN
Independent Call Girl in 😋 Goa +9316020077 Goa Call Girl

Independent Call Girl in 😋 Goa +9316020077 Goa Call Girl
No Advance 931~602~0077 Goa ✂️ Call Girl , Indian Call Girl Goa For Full nig...

No Advance 931~602~0077 Goa ✂️ Call Girl , Indian Call Girl Goa For Full nig...
Obat Penggugur Kandungan Cytotec Dan Gastrul Harga Indomaret

Obat Penggugur Kandungan Cytotec Dan Gastrul Harga Indomaret
Nursing Care Plan for Surgery (Risk for Infection)

Nursing Care Plan for Surgery (Risk for Infection)
CML Chronic Myeloid Leukemia.pdf
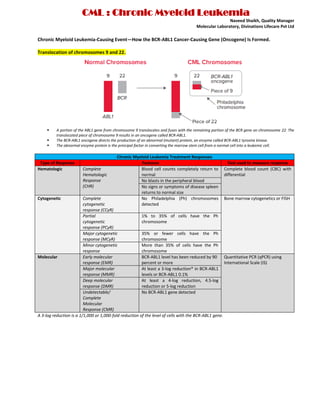
- 1. CML : Chronic Myeloid Leukemia Naveed Shaikh, Quality Manager Molecular Laboratory, Divinations Lifecare Pvt Ltd Chronic Myeloid Leukemia-Causing Event—How the BCR-ABL1 Cancer-Causing Gene (Oncogene) Is Formed. Translocation of chromosomes 9 and 22. A portion of the ABL1 gene from chromosome 9 translocates and fuses with the remaining portion of the BCR gene on chromosome 22. The translocated piece of chromosome 9 results in an oncogene called BCR-ABL1. The BCR-ABL1 oncogene directs the production of an abnormal (mutant) protein, an enzyme called BCR-ABL1 tyrosine kinase. The abnormal enzyme protein is the principal factor in converting the marrow stem cell from a normal cell into a leukemic cell. Chronic Myeloid Leukemia Treatment Responses Type of Response Features Test used to measure response Hematologic Complete Hematologic Response (CHR) Blood cell counts completely return to normal Complete blood count (CBC) with differential No blasts in the peripheral blood No signs or symptoms of disease spleen returns to normal size Cytogenetic Complete cytogenetic response (CCyR) No Philadelphia (Ph) chromosomes detected Bone marrow cytogenetics or FISH Partial cytogenetic response (PCyR) 1% to 35% of cells have the Ph chromosome Major cytogenetic response (MCyR) 35% or fewer cells have the Ph chromosome Minor cytogenetic response More than 35% of cells have the Ph chromosome Molecular Early molecular response (EMR) BCR-ABL1 level has been reduced by 90 percent or more Quantitative PCR (qPCR) using International Scale (IS) Major molecular response (MMR) At least a 3-log reduction* in BCR-ABL1 levels or BCR-ABL1 0.1% Deep molecular response (DMR) At least a 4-log reduction, 4.5-log reduction or 5-log reduction Undetectable/ Complete Molecular Response (CMR) No BCR-ABL1 gene detected A 3-log reduction is a 1/1,000 or 1,000-fold reduction of the level of cells with the BCR-ABL1 gene.
- 2. CML : Chronic Myeloid Leukemia Naveed Shaikh, Quality Manager Molecular Laboratory, Divinations Lifecare Pvt Ltd Treatment Response International Scale Republished with permission of American Society of Hematology (ASH), from ‘Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet’, Michele Baccarani, Giuseppe Saglio, John Goldman et al, 108, (6), 2006; permission conveyed through Copyright Clearance Center, Inc. Log Reduction A log reduction indicates the BCR-ABL1 level has decreased by a certain amount from the standard baseline. 1-log The BCR-ABL1 level has decreased to 10 times below the standardized baseline. This means that 10 percent of cells (10 out of every 100 cells) have the BCR-ABL1 gene. This reduction is equivalent to an early molecular response when achieved within 3 to 6 months of starting treatment. 2-log The BCR-ABL1 level has decreased to 100 times below the standardized baseline. This means that 1 percent of cells (1 out of every 100 cells) have the BCR-ABL1 gene. 3-log The BCR-ABL1 level has decreased to 1,000 times below the standardized baseline. This means that 0.1 percent of cells (1 out of every 1,000 cells) have the BCR-ABL1 gene. It is also known as a “major molecular response (MMR).” 4-log 0.01% of cells (1 out of every 10,000 cells) have the BCR-ABL1 gene. 4.5-log A “complete molecular response (CMR)” or a “deep molecular response (DMR).” Doctors may refer to this as “MR4.5.” A 4.5-log reduction indicates that 0.0032% of cells (1 out of every 32,000 cells) have the BCR-ABL1 gene. Achieving a deep molecular response is a sign of disease remission. Patients who achieve and then sustain a deep molecular response for a significant period of time may be considered candidates for discontinuing TKI therapy, under careful medical supervision. 5-log 0.001% of cells (1 out of every 100,000) have the BCR-ABL1 gene. By reaching a 5-log reduction, patients have achieved “undetectable BCR-ABL1.”
- 3. CML : Chronic Myeloid Leukemia Naveed Shaikh, Quality Manager Molecular Laboratory, Divinations Lifecare Pvt Ltd Treatment Response by Log Reduction Treatment Response Milestones and Follow-up Recommendation Guidelines TIME AFTER START OF TREATMENT BCR-ABL1 (IS) 3 months 6 months 12 months * More than 15 months >10 % YELLOW RED > 1% - >10% GREEN YELLOW RED ≤ 1% GREEN Color Code Concern Treatment Team Considerations Potential Decisions About Treatment RED TKI-resistant disease Evaluate patient compliance and drug interactions Switch to alternate TKI Consider BCR-ABL1 gene mutation testing Evaluate for allogeneic stem cell transplantation YELLOW Possible TKI resistance Evaluate patient compliance and drug interactions Switch to alternate TKI OR Consider BCR-ABL1 gene mutation testing Continue same TKI (other than imatinib) OR Consider bone marrow cytogenetic testing to assess for MCyR at 3 months or CCyR at 12 months Dose escalation of imatinib (to a max of 800 mg) AND Consider evaluation for allogeneic stem cell transplantation GREEN No concerns– treatment is working Monitor response Continue same TKI† Monitor and manage side effects as needed Adapted from NCCN Clinical Practice Guidelines in Oncology: Chronic Myeloid Leukemia; version 3.2020. Abbreviations: MCyR, major cytogenetic response; CCyR; complete cytogenetic response; TKI, tyrosine kinase inhibitor. *BCR-ABL1 0.1% at 12 months is associated with a very low probability of disease progression and a high likelihood of achieving a subsequent MR4.5, which may allow for discontinuation of TKI therapy. †Discontinuation of TKI with careful monitoring is possible in selected patients.
