Stereochemistry
•
2 likes•36 views
The aim in this lecture is to study 1. isomer 2. Enantiomers 3. Chiral carbon atom 4. Optical activity 5. Mesocompound 6.
Report
Share
Report
Share
Download to read offline
Recommended
STEREOISOMERISM IN NATURAL PRODUCTSConcept of stereoisomerism of natural products

Concept of stereoisomerism of natural productsMAHAJAN DHANRAJ,R C PATEL INSTITUTE OF PHARMACEUTICAL EDUCATION AND RESEARCH, SHIRPUR
Recommended
STEREOISOMERISM IN NATURAL PRODUCTSConcept of stereoisomerism of natural products

Concept of stereoisomerism of natural productsMAHAJAN DHANRAJ,R C PATEL INSTITUTE OF PHARMACEUTICAL EDUCATION AND RESEARCH, SHIRPUR
More Related Content
What's hot
What's hot (19)
Applied Chapter 3.1 : Substances are Made of Atoms

Applied Chapter 3.1 : Substances are Made of Atoms
Constructing organic compounds lab (1) modfied no molecular kits

Constructing organic compounds lab (1) modfied no molecular kits
Isomerism an introduction ,Geometrical Isomerism Syn Anti isomerism E-Z Isome...

Isomerism an introduction ,Geometrical Isomerism Syn Anti isomerism E-Z Isome...
Similar to Stereochemistry
Similar to Stereochemistry (20)
Practical Analytical Instrumentation in On-line Applications

Practical Analytical Instrumentation in On-line Applications
Recently uploaded
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
God is a creative God Gen 1:1. All that He created was “good”, could also be translated “beautiful”. God created man in His own image Gen 1:27. Maths helps us discover the beauty that God has created in His world and, in turn, create beautiful designs to serve and enrich the lives of others.
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Recently uploaded (20)
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
Role Of Transgenic Animal In Target Validation-1.pptx

Role Of Transgenic Animal In Target Validation-1.pptx
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
Seal of Good Local Governance (SGLG) 2024Final.pptx

Seal of Good Local Governance (SGLG) 2024Final.pptx
Stereochemistry
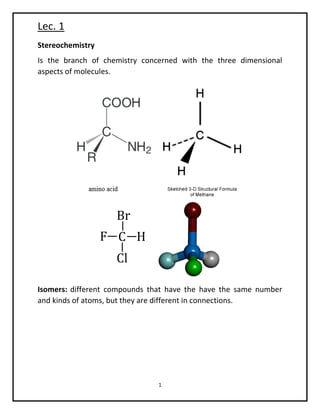
- 1. Lec. 1 1 Stereochemistry Is the branch of chemistry concerned with the three dimensional aspects of molecules. Isomers: different compounds that have the have the same number and kinds of atoms, but they are different in connections.
- 2. Lec. 1 2
- 3. Lec. 1 3 Enantiomers: are stereoisomers that are mirror images but cannot be superimposed.
- 4. Lec. 1 4 Enantiomers have identical physical properties but different in biological activity. One enantiomer of a drug can be considered as a cure for a disease while another could be very toxic. D-amino acids are biologically not active, while L-amino acids are active. D-sugars are biologically active, while L- sugars are not. Chiral carbon atom Tetrahedral carbon atom connected to four different groups is called chiral carbon, it has asymmetrical carbon. Example
- 5. Lec. 1 5 (a) (b) D-alanine called a chiral molecule. It has mirror image and they are superimposed. Number of isomers (I)=2n I=number of isomers n= number of chiral atoms In alanine n=1, I=21 =2
- 7. Lec. 1 7 Example Give me all isomer of ribose C5H10O5 Optical activity All biological compounds such as sugars, amino acids and drugs with chiral carbon atoms can rotate the polarized light to the right or to the left.
- 8. Lec. 1 8 Mesocompound: A molecule that contains chiral centers but optically inactive. It contains internal plane of symmetry:
- 9. Lec. 1 9 Smiley face has an internal mirror plane of symmetry
- 10. Lec. 1 10 Example for cyclic enantiomers and diastereomers.
- 11. Lec. 1 11 Stereoisomers of carbon compounds Configuration: The arrangement in space of the atoms of a molecule. Chiral carbon atom: a carbon atom bonded to for different atom or groups. Optical activity: all chiral molecules have the ability to rotate plane- polarized light. Polarized light: light in which all rays vibrate in a single plane.
- 12. Lec. 1 12 Any substance that will rotate plane polarized light is said to be optically active. A compound that rotates plane-polarized light to the right is called dextrorotatory. If the rotation is to the left, the compound is called levorotatory.These compound are called optical isomers. A mixture of equal amounts of the two enantiomers is called a racemic mixture. Such a mixture is optically inactive. Diastereomers: Stereoisomers that are optically active but are not mirror images.
- 13. Lec. 1 13 Meso stereoisomers: A molecule that contains chiral centers but is optically inactive.