Mass spectroscopy
- 2. Mass spectroscopy is one of the primary spectroscopic methods for molecular analysis available to organic chemist. It is a microanalytical technique requiring only a few nanomoles of the sample to obtain characteristic information pertaining to the structure and molecular weight of analyte. It involves the production and separation of ionised molecules and their ionic decompositon product and finally measurement of the relative abundance of different the ions produced. It is, thus a destructive technique in that the sample is consumed during analysis.
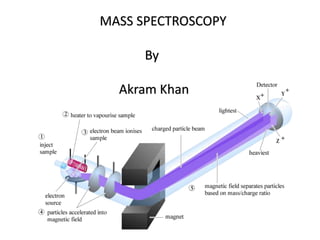
- 3. Principle Mass spectroscopy is the most accurate method for determining the molecular mass of the compound and its elemental composition. In this technique, molecules are bombarded with a beam of energetic electrons. The molecules are ionised and broken up into many fragments, some of which are positive ions. Each kind of ion has a particular ratio of mass to charge, i.e. m/e ratio(value). For most ions, the charge is one and thus, m/e ratio is simply the molecular mass of the ion.
- 6. cc
- 7. solid Directly into the chamber with low vapour pressure by means of probe. e.g. steroids, carbohydrates Vaporized with the help of heat inlet system. e.g. sugars , amino acids Gas Directly inleted & vaporized Liquid
- 10. CHEMICAL IONIZATION Reactive gases bombards with electron beam Reactive species are formed which colloid with sample molecules +vely charged ions are produced 1 0
- 11. Fast atoms of Xe/Ar collides with neutral gas atoms Transferof energy to gas atoms High energy gas atoms directed towards sample Energy transfer to sample Fast atom bombardment
- 14. + - + + - + - + + +- Analyte Matrix Laser + + + + -+ - + + + + Laser is bombarded with Matrix containing thesample Energy transfer to sample Expansion of matrix
- 15. +ve ions at the end of chamber Bombarded with accelerating potential(2-8kV) Deflection inmagnetic field according to velocity & m/e 1 5 Analyzers are used to deflect ions based upon m/e ratio. MASS ANALYSER
- 16. SINGLE FOCUSING MAGNETIC ANALYSER
- 17. DOUBLE FOCUSING MAGNETIC ANALYSER
- 18. Magneticfield Ions of same mass number are separated Less sensitivity(10-1) Magnetic field & electric field Ions of same mass no.& same velocity are separated Highly sensitive(10-12) 18
- 20. sensitive. 1000 times greater than faraday cup. Least sensitive. Cup will capture the ion. Most sensitive. Used for high resolution. Photomult iplier detector Photogra phic detectors Faraday Cup detector 20 DETECTORS
- 22. Identification of fragmentation pattern Impurity detection As LC-MS in analysis ofproteins 22 Identification of an unknown compound Molecular weight , formula &elemental composition determination Identification of an unknown compound APPLICATIONS
- 23. SPECTRA
