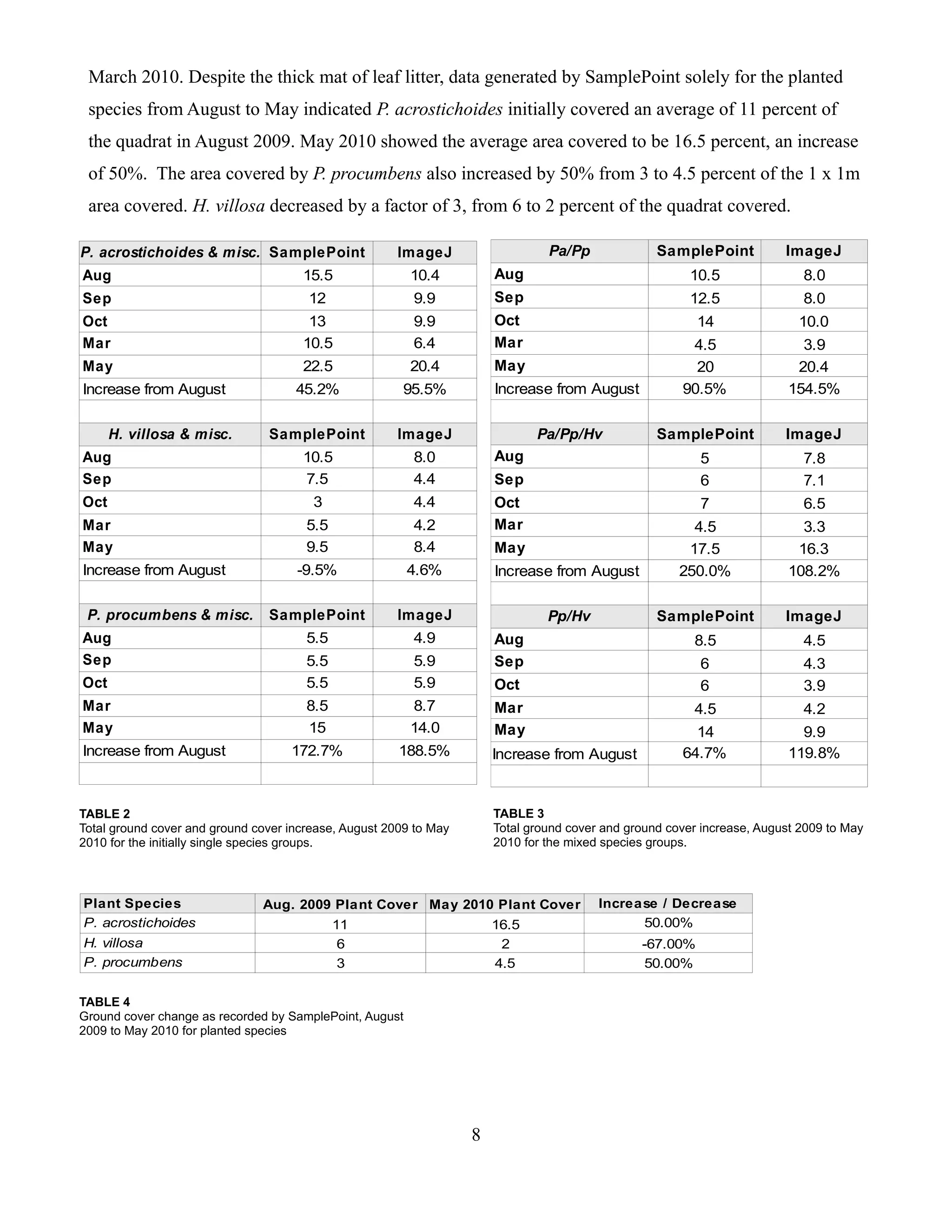
This project evaluated the potential of three plant species - Heuchera villosa, Polystichum acrostichoides, and Pachysandra procumbens - for ground cover on a shaded north-facing slope at the Maymont Estate in Richmond, Virginia. Digital photos of test plots with different species combinations were taken over time. Two software programs - SamplePoint and ImageJ - were used to quantify ground cover from the images. SamplePoint uses point sampling while ImageJ analyzes pixel colors. Results from the two methods were similar. SamplePoint is easier to use but ImageJ has potential with proper algorithms. P. acrostichoides showed the best growth over time, increasing from 11% to