IA on effect of different transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
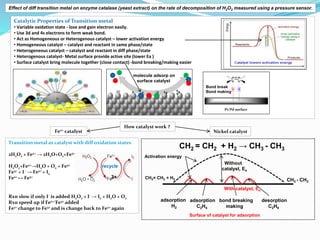
- 1. Pt/Pd surface Catalytic Properties of Transition metal • Variable oxidation state - lose and gain electron easily. • Use 3d and 4s electrons to form weak bond. • Act as Homogeneous or Heterogenous catalyst – lower activation energy • Homogeneous catalyst – catalyst and reactant in same phase/state • Heterogeneous catalyst – catalyst and reactant in diff phase/state • Heterogenous catalyst- Metal surface provide active site (lower Ea ) • Surface catalyst bring molecule together (close contact) -bond breaking/making easier Transition metal as catalyst with diff oxidation states 2H2O2 + Fe2+ → 2H2O+O2+Fe3+ H2O2+Fe2+→H2O + O2 + Fe3+ Fe3+ + I - → Fe2+ + I2 Fe2+ ↔ Fe3+ Rxn slow if only I- is added H2O2 + I- → I2 + H2O + O2 Rxn speed up if Fe2+/Fe3+ added Fe2+ change to Fe3+ and is change back to Fe2+ again recycle molecule adsorp on surface catalyst Pt/Pd surface Bond break Bond making 3+ CH2 = CH2 + H2 → CH3 - CH3 Nickel catalyst Without catalyst, Ea CH2= CH2 + H2 CH3 - CH3 Surface of catalyst for adsorption With catalyst, Ea adsorption H2 adsorption C2H4 bond breaking making desorption C2H6 Fe2+ catalyst How catalyst work ? Activation energy Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
- 2. Across period Cr - 4s13d5 • half filled more stable Cu - 4s13d10 • fully filled more stable Ca 4s2 K 4s1 Transition metal have partially fill 3d orbital • 3d and 4s electron can be lost easily • electron fill from 4s first then 3d • electron lost from 4s first then 3d • 3d and 4s energy level close together (similar in energy) Filling electron- 4s level lower, fill first Losing electron- 4s higher, lose first 3d 4s Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
- 3. Centrifuge to spin down the extract Catalase extract (clear solution used) Yeast, pressure sensor, H2O2 Diff transition metal were used to determine which will inhibit enzyme catalase Pb2+, Cu2+, Ni2+ 1% (w/v) of solution was prepared. Amt added from 0.003 – 0.004 mol. 5% (v/v) H2O2 used. Pressure sensor to measure O2 released. Reaction mechanism Procedure: 5g of yeast powder added to 50ml water (10%) – centrifuge to collect extract 1ml catalase extract added to 1 ml of transition metal solution Solution added to 5ml of 5% H2O2 Rate was measured – change of pressure over time. Hydrogen peroxide decomposition – O2 production 2H2O2→ 2H2O + O2 Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
- 4. 0 1 2 3 4 Ni2+ Pb2+ Cu2+ +ve control Rate of decomposition Type transition metals Type transition metals vs rate of decomposition Metal chosen as they will not act as catalyst to break down H2O2 Negative control - only Ni2+, Pb2+, Cu2+ Positive control – only catalase without any transition metal. Pressure increase - due to enzyme catalase, and not transition metal. Transition metal Rate/kPas-1 % reduction in rate Ni2+ 2.725 (3.39-2.725/3.39) x 100% = 20% Pb2+ 1.438 (3.39-1.438/3.39) x 100% = 58% Cu2+ 1.000 (3.39-1.0/3.39) x 100% = 70% +ve control 3.390 -ve control No change Rate measured as pressure change over time. Pb and Cu metal, used for further investigation as they are strong inhibitor for enzyme catalase Diff transition metal were used. Rate measured as change of pressure over time. Ni2+, Pb2+, Cu2+ Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
- 5. Method 1 Method 2 Time Time Volume Pressure • Rate = Δ vol O2 over time • Volume recorded • Rate = Δ pressure O2 over time • Pressure recorded Procedure 2H2O2 → O2 + 2H2O Rxn: H2O2 with diff (catalyst) measured using TWO diff methods • 2H2O2 → O2 + 2H2O (H2O2 limiting, KI excess) • Pipette 1ml 1.0M KI to 20ml of 1.5% H2O2 • Vol O2 released recorded at 1 min interval • Repeated using 3% H2O2 conc Time/m Vol O2 (H2O2 1.5%) Vol O2 (H2O2 3.0%) 0 0.0 0.0 1 8.5 14.0 2 15.0 26.5 3 21.0 34.0 4 26.0 39.0 Volume O2 Time 3 % 1.5 % Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
- 6. • 2H2O2 → O2 + 2H2O (H2O2 limiting, KI excess) • Pipette 1ml 1.0M KI to 20ml of 1.5% H2O2 • Pressure O2 released recorded at 1 min interval • Repeat using 3% H2O2 conc Method 1 Method 2 Time Time Volume Pressure • Rate = Δ vol O2 over time • Volume recorded • Rate = Δ pressure O2 over time • Pressure recorded Procedure 2H2O2 → O2 + 2H2O Time 3 % 1.5 % Time/m Pressure O2 (H2O2 1.5%) Pressure O2 (H2O2 3%) 0 101.3 101.3 1 102.4 103.4 2 103.5 105.6 3 110.3 115.2 4 113.5 118.2 Pressure O2 Rxn: H2O2 with diff (catalyst) measured using TWO diff methods Effect of diff transition metal on enzyme catalase (yeast extract) on the rate of decomposition of H2O2 measured using a pressure sensor.
